Abstract
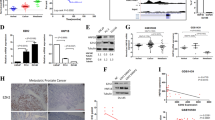
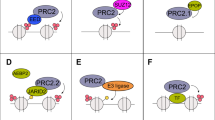
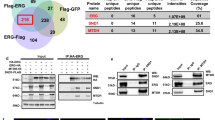
Prostate cancer is a leading cause of cancer-related death in males and is second only to lung cancer. Although effective surgical and radiation treatments exist for clinically localized prostate cancer, metastatic prostate cancer remains essentially incurable. Here we show, through gene expression profiling1, that the polycomb group protein enhancer of zeste homolog 2 (EZH2)2,3 is overexpressed in hormone-refractory, metastatic prostate cancer. Small interfering RNA (siRNA) duplexes4 targeted against EZH2 reduce the amounts of EZH2 protein present in prostate cells and also inhibit cell proliferation in vitro. Ectopic expression of EZH2 in prostate cells induces transcriptional repression of a specific cohort of genes. Gene silencing mediated by EZH2 requires the SET domain and is attenuated by inhibiting histone deacetylase activity. Amounts of both EZH2 messenger RNA and EZH2 protein are increased in metastatic prostate cancer; in addition, clinically localized prostate cancers that express higher concentrations of EZH2 show a poorer prognosis. Thus, dysregulated expression of EZH2 may be involved in the progression of prostate cancer, as well as being a marker that distinguishes indolent prostate cancer from those at risk of lethal progression.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Dhanasekaran, S. M. et al. Delineation of prognostic biomarkers in prostate cancer. Nature 412, 822–826 (2001)
Laible, G. et al. Mammalian homologues of the Polycomb-group gene Enhancer of zeste mediate gene silencing in Drosophila heterochromatin and at S. cerevisiae telomeres. EMBO J. 16, 3219–3232 (1997)
Satijn, D. P. & Otte, A. P. Polycomb group protein complexes: do different complexes regulate distinct target genes? Biochim. Biophys. Acta 1447, 1–16 (1999)
Elbashir, S. M. et al. Duplexes of 21-nucleotide RNAs mediate RNA interference in cultured mammalian cells. Nature 411, 494–498 (2001)
Jacobs, J. J. & van Lohuizen, M. Cellular memory of transcriptional states by Polycomb-group proteins. Semin. Cell. Dev. Biol. 10, 227–235 (1999)
Francis, N. J. & Kingston, R. E. Mechanisms of transcriptional memory. Nature Rev. Mol. Cell Biol. 2, 409–421 (2001)
Jacobs, J. J., Kieboom, K., Marino, S., DePinho, R. A. & van Lohuizen, M. The oncogene and Polycomb-group gene bmi-1 regulates cell proliferation and senescence through the ink4a locus. Nature 397, 164–168 (1999)
Jacobs, J. J. et al. Bmi-1 collaborates with c-Myc in tumorigenesis by inhibiting c-Myc-induced apoptosis via INK4a/ARF. Genes Dev. 13, 2678–2690 (1999)
Mahmoudi, T. & Verrijzer, C. P. Chromatin silencing and activation by Polycomb and trithorax group proteins. Oncogene 20, 3055–3066 (2001)
LaJeunesse, D. & Shearn, A. E(z): a polycomb group gene or a trithorax group gene? Development 122, 2189–2197 (1996)
Raaphorst, F. M. et al. Coexpression of BMI-1 and EZH2 polycomb group genes in Reed-Sternberg cells of Hodgkin's disease. Am. J. Pathol. 157, 709–715 (2000)
van Lohuizen, M. et al. Identification of cooperating oncogenes in Eµ–myc transgenic mice by provirus tagging. Cell 65, 737–752 (1991)
Haupt, Y., Alexander, W. S., Barri, G., Klinken, S. P. & Adams, J. M. Novel zinc finger gene implicated as myc collaborator by retrovirally accelerated lymphomagenesis in Eµ–myc transgenic mice. Cell 65, 753–763 (1991)
Jenuwein, T., Laible, G., Dorn, R. & Reuter, G. SET domain proteins modulate chromatin domains in eu- and heterochromatin. Cell. Mol. Life Sci. 54, 80–93 (1998)
Tusher, V. G., Tibshirani, R. & Chu, G. Significance analysis of microarrays applied to the ionizing radiation response. Proc. Natl Acad. Sci. USA 98, 5116–5121 (2001)
van Lohuizen, M. et al. Interaction of mouse polycomb-group (Pc-G) proteins Enx1 and Enx2 with Eed: indication for separate Pc-G complexes. Mol. Cell. Biol. 18, 3572–3579 (1998)
Sewalt, R. G. et al. Characterization of interactions between the mammalian polycomb-group proteins Enx1/EZH2 and EED suggests the existence of different mammalian polycomb-group protein complexes. Mol. Cell. Biol. 18, 3586–3595 (1998)
Rubin, M. A. et al. Rapid (‘warm’) autopsy study for procurement of metastatic prostate cancer. Clin. Cancer Res. 6, 1038–1045 (2000)
Perrone, E. E. et al. Tissue microarray assessment of prostate cancer tumour proliferation in African-American and white men. J. Natl Cancer Inst. 92, 937–939 (2000)
Webber, M. M., Bello, D., Kleinman, H. K. & Hoffman, M. P. Acinar differentiation by non-malignant immortalized human prostatic epithelial cells and its loss by malignant cells. Carcinogenesis 18, 1225–1231 (1997)
Bello, D., Webber, M. M., Kleinman, H. K., Wartinger, D. D. & Rhim, J. S. Androgen responsive adult human prostatic epithelial cell lines immortalized by human papillomavirus 18. Carcinogenesis 18, 1215–1223 (1997)
Littlewood, T. D., Hancock, D. C., Danielian, P. S., Parker, M. G. & Evan, G. I. A modified oestrogen receptor ligand-binding domain as an improved switch for the regulation of heterologous proteins. Nucleic Acids Res. 23, 1686–1690 (1995)
Juin, P., Hueber, A. O., Littlewood, T. & Evan, G. c-Myc induced sensitization to apoptosis is mediated through cytochrome c release. Genes Dev. 13, 1367–1381 (1999)
van der Vlag, J. & Otte, A. P. Transcriptional repression mediated by the human polycomb-group protein EED involves histone deacetylation. Nature Genet. 23, 474–478 (1999)
Eisen, M. B., Spellman, P. T., Brown, P. O. & Botstein, D. Cluster analysis and display of genome-wide expression patterns. Proc. Natl Acad. Sci. USA 95, 14863–14868 (1998)
Manley, S., Mucci, N. R., De Marzo, A. M. & Rubin, M. A. Relational database structure to manage high-density tissue microarray data and images for pathology studies focusing on clinical outcome: the prostate specialized program of research excellence model. Am. J. Pathol. 159, 837–843 (2001)
Bova, G. S. et al. Web-based tissue microarray image data analysis: initial validation testing through prostate cancer Gleason grading. Hum. Pathol. 32, 417–427 (2001)
Kumar-Sinha, C., Varambally, S., Sreekumar, A. & Chinnaiyan, A. M. Molecular cross-talk between the TRAIL and interferon signaling pathways. J. Biol. Chem. 277, 575–585 (2001)
Acknowledgements
This study was made possible by tissues donated by patients with metastatic prostate cancer enrolled in the University of Michigan, Rapid Autopsy Program funded by the Specialized Program of Research Excellence (SPORE) in Prostate Cancer at the National Cancer Institute. We thank J. Wei for clinical data collection; K. Hamer for preparing polycomb antibodies; R. Kunkel for figure preparation; J. Harwood and M. LeBlanc for technical assistance; A. Menon for sequence verification; and C. Ingold and G. Tueckmantel for database assistance. A.M.C. is a Pew Foundation Scholar. This work is supported in part by grants from the NIH (A.M.C.), CaPCURE (A.M.C.) and the Michigan SPORE in Prostate Cancer (K.P., M.A.R., A.M.C. and M.G.S.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Rights and permissions
About this article
Cite this article
Varambally, S., Dhanasekaran, S., Zhou, M. et al. The polycomb group protein EZH2 is involved in progression of prostate cancer. Nature 419, 624–629 (2002). https://doi.org/10.1038/nature01075
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature01075
This article is cited by
-
CUL4B-DDB1-COP1-mediated UTX downregulation promotes colorectal cancer progression
Experimental Hematology & Oncology (2023)
-
UHRF1/UBE2L6/UBR4-mediated ubiquitination regulates EZH2 abundance and thereby melanocytic differentiation phenotypes in melanoma
Oncogene (2023)
-
Nuclear condensates of YAP fusion proteins alter transcription to drive ependymoma tumourigenesis
Nature Cell Biology (2023)
-
Lysine methylation promotes NFAT5 activation and determines temozolomide efficacy in glioblastoma
Nature Communications (2023)
-
Methylation across the central dogma in health and diseases: new therapeutic strategies
Signal Transduction and Targeted Therapy (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.