Abstract
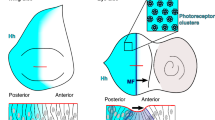
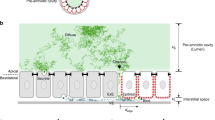
Developmental patterning relies on morphogen gradients, which generally involve feedback loops to buffer against perturbations caused by fluctuations in gene dosage and expression1. Although many gene components involved in such feedback loops have been identified, how they work together to generate a robust pattern remains unclear. Here we study the network of extracellular proteins that patterns the dorsal region of the Drosophila embryo by establishing a graded activation of the bone morphogenic protein (BMP) pathway. We find that the BMP activation gradient itself is robust to changes in gene dosage. Computational search for networks that support robustness shows that transport of the BMP class ligands (Scw and Dpp) into the dorsal midline by the BMP inhibitor Sog is the key event in this patterning process. The mechanism underlying robustness relies on the ability to store an excess of signalling molecules in a restricted spatial domain where Sog is largely absent. It requires extensive diffusion of the BMP–Sog complexes, coupled with restricted diffusion of the free ligands. We show experimentally that Dpp is widely diffusible in the presence of Sog but tightly localized in its absence, thus validating a central prediction of our theoretical study.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Freeman, M. Feedback control of intercellular signalling in development. Nature 408, 313–319 (2000)
Podos, S. D. & Ferguson, E. L. Morphogen gradients: new insights from DPP. Trends. Genet. 15, 396–402 (1999)
Raftery, L. A. & Sutherland, D. J. TGF-β family signal transduction in Drosophila development: from Mad to Smads. Dev. Biol. 210, 251–268 (1999)
Stein, D., Roth, S., Vogelsang, E. & Nusslein-Volhard, C. The polarity of the dorsoventral axis in the Drosophila embryo is defined by an extracellular signal. Cell 65, 725–735 (1991)
DeRobertis, E. M. & Sasai, Y. A common plan for dorsoventral patterning in Bilateria. Nature 380, 37–40 (1996)
Nguyen, M., Park, S., Marques, G. & Arora, K. Interpretation of a BMP activity gradient in Drosophila embryos depends on synergistic signaling by two type I receptors, SAX and TKV. Cell 95, 495–506 (1998)
Arora, K., Levine, M. S. & O'Connor, M. B. The screw gene encodes a ubiquitously expressed member of the TGF-β family required for specification of dorsal cell fates in the Drosophila embryo. Genes Dev. 8, 2588–2601 (1994)
Mason, E. D., Williams, S., Grotendorst, G. R. & Marsh, J. L. Combinatorial signaling by Twisted Gastrulation and Decapentaplegic. Mech. Dev. 64, 61–75 (1997)
Persson, U. et al. The L45 loop in type I receptors for TGF-β family members is a critical determinant in specifying Smad isoform activation. FEBS Lett. 434, 83–87 (1998)
Tanimoto, H., Itoh, S., ten Dijke, P. & Tabata, T. Hedgehog creates a gradient of DPP activity in Drosophila wing imaginal discs. Mol. Cell 5, 59–71 (2000)
Dorfman, R. & Shilo, B.-Z. Biphasic activation of the BMP pathway in the Drosophila embryonic dorsal region. Development 128, 965–972 (2001)
Wharton, K. A., Ray, R. P. & Gelbart, W. M. An activity gradient of decapentaplegic is necessary for the specification of dorsal pattern elements in the Drosophila embryo. Development 117, 807–822 (1993)
Srinivasan, S., Rashka, K. E. & Bier, E. Creation of a Sog morphogen gradient in the Drosophila embryo. Dev. Cell 2, 91–101 (2002)
Neul, J. L. & Ferguson, E. L. Spatially restricted activation of the SAX receptor by SCW modulates DPP/TKV signaling in Drosophila dorsal–ventral patterning. Cell 95, 483–494 (1998)
Ross, J. J. et al. Twisted gastrulation is a conserved extracellular BMP antagonist. Nature 410, 479–483 (2001)
Marques, G. et al. Production of a DPP activity gradient in the early Drosophila embryo through the opposing actions of the SOG and TLD proteins. Cell 91, 417–426 (1997)
Ashe, H. L., Mannervik, M. & Levine, M. Dpp signaling thresholds in the dorsal ectoderm of the Drosophila embryo. Development 127, 3305–3312 (2000)
Barkai, N. & Leibler, S. Robustness in simple biochemical networks. Nature 387, 913–917 (1997)
Barkai, N. & Leibler, S. Circadian clocks limited by noise. Nature 403, 267–268 (2000)
von Dassow, G., Meir, E., Munro, E. M. & Odell, G. M. The segment polarity network is a robust developmental module. Nature 406, 188–192 (2000)
Hartwell, L. H., Hopfield, J. J., Leibler, S. & Murray, A. W. From molecular to modular biology. Nature 402, C47–C52 (1999)
Kosman, D. & Small, S. Concentration-dependent patterning by an ectopic expression domain of the Drosophila gap gene knirps. Development 124, 1343–1354 (1997)
Swaminathan, R., Hoang, C. P. & Verkman, A. S. Photobleaching recovery and anisotropy decay of green fluorescent protein GFP–S65T in solution and cells: cytoplasmic viscosity probed by green fluorescent protein translational and rotational diffusion. Biophys. J. 72, 1900–1907 (1997)
Acknowledgements
We thank P. ten Dijke for the pMad antibody; L. Marsh, M. O'Connor, S. Roth, D. St. Johnston and the Umea and Bloomington Fly Centers for strains; and S. Leibler and S. Roth for comments and criticism. This work was funded by the Israel Science Foundation (B-Z.S.) and the Israel Science Foundation and the Minerva Foundation (N.B). H.A. is a Lister Institute Research Fellow. B-Z.S. is the incumbent of the Hilda and Cecil Lewis professorial chair in Molecular Genetics. N.B. is the incumbent of the Soretta and Henry Shapiro career development chair.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Supplementary information
Rights and permissions
About this article
Cite this article
Eldar, A., Dorfman, R., Weiss, D. et al. Robustness of the BMP morphogen gradient in Drosophila embryonic patterning. Nature 419, 304–308 (2002). https://doi.org/10.1038/nature01061
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature01061
This article is cited by
-
Highly conserved and extremely evolvable: BMP signalling in secondary axis patterning of Cnidaria and Bilateria
Development Genes and Evolution (2024)
-
Universal structures for adaptation in biochemical reaction networks
Nature Communications (2023)
-
Flagellar dynamics reveal fluctuations and kinetic limit in the Escherichia coli chemotaxis network
Scientific Reports (2023)
-
Early radial positional information in the cochlea is optimized by a precise linear BMP gradient and enhanced by SOX2
Scientific Reports (2023)
-
Generation of extracellular morphogen gradients: the case for diffusion
Nature Reviews Genetics (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.