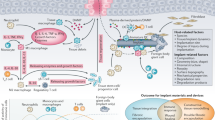
Abstract
Implantable neuroprostheses are engineered systems designed to restore or substitute function for individuals with neurological deficits or disabilities. These systems involve at least one uni- or bidirectional interface between a living neural tissue and a synthetic structure, through which information in the form of electrons, ions or photons flows. Despite a few notable exceptions, the clinical dissemination of implantable neuroprostheses remains limited, because many implants display inconsistent long-term stability and performance, and are ultimately rejected by the body. Intensive research is currently being conducted to untangle the complex interplay of failure mechanisms. In this Review, we emphasize the importance of minimizing the physical and mechanical mismatch between neural tissues and implantable interfaces. We explore possible materials solutions to design and manufacture neurointegrated prostheses, and outline their immense therapeutic potential.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Hochberg, L. R. et al. Reach and grasp by people with tetraplegia using a neurally controlled robotic arm. Nature 485, 372–375 (2012).
Bouton, C. E. et al. Restoring cortical control of functional movement in a human with quadriplegia. Nature 533, 247–250 (2016).
Barrese, J. C. et al. Failure mode analysis of silicon-based intracortical microelectrode arrays in non-human primates. J. Neural Engineer. 10, 066014 (2013).
Barrese, J. C., Aceros, J. & Donoghue, J. P. Scanning electron microscopy of chronically implanted intracortical microelectrode arrays in non-human primates. J. Neural Engineer. 13, 026003 (2016).
Sanchez, J. C., Alba, N., Batich, C. & Carney, P. R. Structural modifications in chronic microwire electrodes for cortical neuroprosthetics: a case study. IEEE Trans. Neural Syst. Rehabil. Eng. 14, 217–212 (2006).
Sankar, V. et al. Electrode impedance analysis of chronic tungsten microwire neural implants: understanding abiotic vs. biotic contributions. Front. Neuroeng. 7, http://dx.doi.org/10.3389/fneng.2014.00013 (2014).
Jorfi, M., Skousen, J. L., Weder, C. & Capadona, J. R. Progress towards biocompatible intracortical microelectrodes for neural interfacing applications. J. Neural Eng. 12, 011001 (2015).
Jeong, J.-W. et al. Soft materials in neuroengineering for hard problems in neuroscience. Neuron 86, 175–186 (2015).
Lee, J. H., Kim, H., Kim, J. H. & Lee, S.-H. Soft implantable microelectrodes for future medicine: prosthetics, neural signal recording and neuromodulation. Lab Chip 16, 959–976 (2016).
Prodanov, D. & Delbeke, J. Mechanical and biological interactions of implants with the brain and their impact on implant design. Front. Neurosci. 10, http://dx.doi.org/10.3389/fnins.2016.00011 (2016).
Jones, E. G. & Rakic, P. Radial columns in cortical architecture: it is the composition that counts. Cerebral Cortex 20, 2261–2264 (1984).
Bailey, S. A., Zidell, R. H. & Perry, R. W. Relationships between organ weight and body/brain weight in the rat: what is the best analytical endpoint? Toxicol. Pathol. 32, 448–466 (2004).
Koo, B. B. et al. Age-related effects on cortical thickness patterns of the rhesus monkey brain. Neurobiol. Aging 33, 200.e23–200.e31 (2012).
Herculano-Houzel, S. in In The Light of Evolution: Volume VI: Brain and Behavior Ch. 8 (eds Striedter, G. F., Avise, J. C. & Ayala, F. J. ) 127–148 (National Academies Press, 2013).
Tallinen, T. et al. On the growth and form of cortical convolutions. Nat. Phys. 12, 588–593 (2016).
Wagshul, M. E., Eide, P. K. & Madsen, J. R. The pulsating brain: a review of experimental and clinical studies of intracranial pulsatility. Fluids Barriers CNS 8, http://dx.doi.org/10.1186/2045-8118-8-5 (2011).
Harrison, D. E., Cailliet, R., Harrison, D. D., Troyanovich, S. J. & Harrison, S. O. A review of biomechanics of the central nervous system — Part I: spinal canal deformations resulting from changes in posture. J. Manipulative Physiol. Ther. 22, 227–234 (1999).
Bashkatov, A. N. et al. Glucose and mannitol doffusion in human dura mater. Biophys. J. 85, 3310–3318 (2003).
Galasha, F. O. et al. A new type of recording chamber with an easy-toexchange microdrive array for chronic recordings in macaque monkeys. J. Neurophysiol. 105, 3092–3105 (2011).
Minev, I. R. et al. Biomaterials. Electronic dura mater for long-term multimodal neural interfaces. Science 347, 159–163 (2015).
Nicholson, K. J. & Winkelstein, B. A. in Neural Tissue Biomechanics Ch. 10 (ed. Bilston, L. E. ) 203–229 (Springer, 2011).
Polikov, V. S., Tresco, P. A. & Reichert, W. M. Response of brain tissue to chronically implanted neural electrodes. J. Neurosci. Methods 148, 1–18 (2005).
Franze, K., Janmey, P. A. & Guck, J. Mechanics in neuronal development and repair. Annu. Rev. Biomed. Eng. 15, 227–251 (2013).
Ulrich, T. & Kumar, S. in Mechanobiology Handbook 391–411 (CRC Press, 2011).
Bernick, K. B., Prevost, T. P., Suresh, S. & Socrate, S. Biomechanics of single cortical neurons. Acta Biomater. 7, 1210–1219 (2011).
Lu, Y.-B. et al. Viscoelastic properties of individual glial cells and neurons in the CNS. Proc. Natl Acad. Sci. USA 103, 17759–17764 (2006).
Zou, S. et al. Force spectroscopy measurements show that cortical neurons exposed to excitotoxic agonists stiffen before showing evidence of bleb damage. PLoS One 8, e73499 (2013).
Grevesse, T., Dabiri, B. E., Parker, K. K. & Gabriele, S. Opposite rheological properties of neuronal microcompartments predict axonal vulnerability in brain injury. Sci. Rep. 5, 9475 (2015).
Bray, D. Mechanical tension produced by nerve cells in tissue culture. J. Cell Sci. 37, 391–410 (1979).
Dennerll, T. J., Joshi, H. C., Steel, V. L., Buxbaum, R. E. & Heidemann, S. R. Tension and compression in the cytoskeleton of PC-12 neurites. II: Quantitative measurements. J. Cell Biol. 107, 665–674 (1988).
Dennerll, T. J., Lamoureux, P., Buxbaum, R. E. & Heidemann, S. R. The cytomechanics of axonal elongation and retraction. J. Cell Biol. 109, 3073–3083 (1989).
Bernal, R., Pullarkat, P. A. & Melo, F. Mechanical properties of axons. Phys. Rev. Lett. 99, 018301 (2007).
MacDonald, R. B. et al. Müller glia provide essential tensile strength to the developing retina. J. Cell Biol. 210, 1075–1083 (2015).
O'Toole, M., Lamoureux, P. & Miller, K. E. A physical model of axonal elongation: force, viscosity, and adhesions govern the mode of outgrowth. Biophys. J. 94, 2610–2620 (2008).
Jagielska, A. et al. Mechanical environment modulates biological properties of oligodendrocyte progenitor cells. Stem Cells Dev. 21, 2905–2914 (2012).
Lu, Y.-B. et al. Reactive glial cells: increased stiffness correlates with increased intermediate filament expression. FASEB J. 25, 624–631 (2011).
Vergara, D. et al. Biomechanical and proteomic analysis of INF- β-treated astrocytes. Nanotechnology 20, 455106 (2009).
Miller, W. J. et al. Mechanically induced reactive gliosis causes ATP-mediated alterations in astrocyte stiffness. J. Neurotrauma 26, 789–797 (2009).
Novak, U. & Kaye, A. H. Extracellular matrix and the brain: components and function. J. Clin. Neurosci. 7, 280–290 (2000).
Galtrey, C. M., Kwok, J. C. F., Carulli, D., Rhodes, K. E. & Fawcett, J. W. Distribution and synthesis of extracellular matrix proteoglycans, hyaluronan, link proteins and tenascin-R in the rat spinal cord. Eur. J. Neurosci. 27, 1373–1390 (2008).
Barros, C. S., Franco, S. J. & Müller, U. Extracellular matrix: functions in the nervous system. Cold Spring Harb. Perspect. Biol. 3, a005108 (2011).
Gaudet, A. D. & Popovich, P. G. Extracellular matrix regulation of inflammation in the healthy and injured spinal cord. Exp. Neurol. 258, 24–34 (2014).
Swift, J. et al. Nuclear lamin-A scales with tissue stiffness and enhances matrix-directed differentiation. Science 341, 1240104 (2013).
Syková, E. & Nicholson, C. Diffusion in brain extracellular space. Physiol. Rev. 88, 1277–1340 (2008).
Hemphill, M. A., Dauth, S., Yu, C. J., Dabiri, B. E. & Parker, K. K. Traumatic brain injury and the neuronal microenvironment: a potential role for neuropathological mechanotransduction. Neuron 85, 1177–1192 (2015).
Pogoda, K. et al. Compression stiffening of brain and its effect on mechanosensing by glioma cells. New J. Phys. 16, 075002 (2014).
Javid, S., Rezaei, A. & Karami, G. A micromechanical procedure for viscoelastic characterization of the axons and ECM of the brainstem. J. Mechan. Behav. Biomed. Mater. 30, 290–299 (2014).
Cheng, S., Clarke, E. C. & Bilston, L. E. Rheological properties of the tissues of the central nervous system: a review. Med. Eng. Phys. 30, 1318–1337 (2008).
Goriely, A. et al. Mechanics of the brain: perspectives, challenges, and opportunities. Biomech. Model. Mechanobiol. 14, 931–965 (2015).
Fallenstein, G. T., Hulce, V. D. & Melvin, J. W. Dynamic mechanical properties of human brain tissue. J. Biomech. 2, 217–226 (1969).
Ommaya, A. K. Mechanical properties of tissues of the nervous system. J. Biomech. 1, 137–138 (1968).
Chatelin, S., Constantinesco, A. & Willinger, R. Fifty years of brain tissue mechanical testing: from in vitro to in vivo investigations. Biorheology 47, 255–276 (2010).
Koser, D. E., Moeendarbary, E., Hanne, J., Kuerten, S. & Franze, K. CNS cell distribution and axon orientation determine local spinal cord mechanical properties. Biophys. J. 108, 2137–2147 (2015).
Franze, K. et al. Spatial mapping of the mechanical properties of the living retina using scanning force microscopy. Soft Matter 7, 3147–3154 (2011).
Budday, S. et al. Mechanical properties of gray and white matter brain tissue by indentation. J. Mechan. Behav. Biomed. Mater. 46, 318–330 (2015).
Elkin, B. S., Azeloglu, E. U., Costa, K. D. & Morrison, B. Mechanical heterogeneity of the rat hippocampus measured by atomic force microscope indentation. J. Neurotrauma 24, 812–822 (2007).
Elkin, B. S., Ilankovan, A. I. & Morrison, B. A detailed viscoelastic characterization of the p17 and adult rat brain. J. Neurotrauma 28, 2235–2244 (2011).
MacManus, D. B., Pierrat, B., Murphy, J. G. & Gilchrist, M. D. Dynamic mechanical properties of murine brain tissue using micro-indentation. J. Biomech. 48, 3213–3218 (2015).
Prange, M. T. & Margulies, S. S. Regional, directional, and age-dependent properties of the brain undergoing large deformation. J. Biomech. Eng. 124, 244–252 (2002).
Christ, A. F. et al. Mechanical difference between white and gray matter in the rat cerebellum measured by scanning force microscopy. J. Biomech. 43, 2986–2992 (2010).
Ichihara, K., Taguchi, T. & Shimada, Y. Gray matter of the bovine cervical spinal cord is mechanically more rigid and fragile than the white matter. J. Neurotrauma 18, 361–367 (2001).
Shreiber, D. I., Hao, H. & Elias, R. A. Probing the influence of myelin and glia on the tensile properties of the spinal cord. Biomech. Model. Mechanobiol. 8, 311–321 (2009).
Schregel, K. K. et al. Demyelination reduces brain parenchymal stiffness quantified in vivo by magnetic resonance elastography. Proc. Natl Acad. Sci. USA 109, 6650–6655 (2012).
Elkin, B. S., Ilankovan, A. & Morrison, B. Age-dependent regional mechanical properties of the rat hippocampus and cortex. J. Biomech. Eng. 132, 011010 (2010).
Sack, I. et al. The impact of aging and gender on brain viscoelasticity. Neuroimage 46, 652–657 (2009).
de Rooij, R. & Kuhl, E. Constitutive modeling of brain tissue: current perspectives. Appl. Mech. Rev. 68, 010801–010823 (2016).
McKee, C. T., Last, J. A. & Russell, P. Indentation versus tensile measurements of Young's modulus for soft biological tissues. Tissue Eng. Part B Rev. 17, 155–164 (2011).
Sridharan, A., Rajan, S. D. & Muthuswamy, J. Long-term changes in the material properties of brain tissue at the implant–tissue interface. J. Neural Eng. 10, 066001 (2013).
Saxena, T., Gilbert, J., Stelzner, D. & Hasenwinkel, J. Mechanical characterization of the injured spinal cord after lateral spinal hemisection injury in the rat. J. Neurotrauma 29, 1747–1757 (2012).
Estes, M. S. & McElhaney, J. H. Response of brain tissue of compressive loading. Mech. Eng. 92, 58–61 (1970).
Goldstein, S. R. & Salcman, M. Mechanical factors in the design of chronic recording intracortical microelectrodes. IEEE Trans. Biomed. Eng. 20, 260–269 (1973).
Chew, D. J. et al. A microchannel neuroprosthesis for bladder control after spinal cord injury in rat. Sci. Transl. Med. 5, 210ra155 (2013).
Sharp, A. A., Ortega, A. M., Restrepo, D., Curran-Everett, D. & Gall, K. In vivo penetration mechanics and mechanical properties of mouse brain tissue at micrometer scales. IEEE Trans.Biomed. Eng. 56, 45–53 (2009).
Hyunjung, L., Ravi, V. B., Wei, S. & Marc, E. L. Biomechanical analysis of silicon microelectrode-induced strain in the brain. J. Neural Engineer. 2, 81–89 (2005).
Suo, Z., Ma, W. Y., Gleskova, H. & Wagner, S. Mechanics of rollable and foldable film-on-foil electronics. Appl. Phys. Lett. 74, 1177–1179 (1999).
Kim, D.-H. et al. Dissolvable films of silk fibroin for ultrathin conformable biointegrated electronics. Nat. Mater. 9, 511–517 (2010).
Matsuo, T. et al. Intrasulcal electrocorticography in macaque monkeys with minimally invasive neurosurgical protocols. Front. Syst. Neurosci. 5, 34 (2011).
Guo, C. F. Sun, T. Liu, Q. Suo, Z. & Ren, Z. Highly stretchable and transparent nanomesh electrodes made by grain boundary lithography. Nat. Commun. 5, 3121 (2014).
Fan, J. A. et al. Fractal design concepts for stretchable electronics. Nat. Commun. 5, 3266 (2014).
Xu, L. et al. 3D multifunctional integumentary membranes for spatiotemporal cardiac measurements and stimulation across the entire epicardium. Nat. Commun. 5, 3329 (2014).
Minev, I. R., Wenger, N., Courtine, G. & Lacour, S. P. Research update: platinum-elastomer mesocomposite as neural electrode coating. APL Mater. 3, 014701 (2015).
Kang, S.-K. et al. Bioresorbable silicon electronic sensors for the brain. Nature 530, 71–76 (2016).
Yu, K. J., Kuzum, D., Hwang, S. W., Kim, B. H. & Juul, H. Bioresorbable silicon electronics for transient spatiotemporal mapping of electrical activity from the cerebral cortex. Nat. Mater. http://dx.doi.org/10.1038/nmat4624 (2016).
Edell, D. J., Toi, V. V., McNeil, V. M. & Clark, L. D. Factors influencing the biocompatibility of insertable silicon microshafts in cerebral cortex. IEEE Trans. Biomed. Eng. 39, 635–643 (1992).
Bjornsson, C. S. et al. Effects of insertion conditions on tissue strain and vascular damage during neuroprosthetic device insertion. J. Neural Engineer. 3, 196–207 (2006).
Dryg, I. D. et al. Magnetically inserted neural electrodes: tissue response and functional lifetime. IEEE Trans. Neural Syst. Rehab. 23, 562–571 (2015).
Kim, T. et al. Injectable, cellular-scale optoelectronics with applications for wireless optogenetics. Science 12, 211–216 (2013).
Lee, K. et al. Polyimide based neural implants with stiffness improvement. Sens. Actuators B 102, 67–72 (2004).
Ware, T. et al. Three-dimensional flexible electronics enabled by shape memory polymer substrates for responsive neural interfaces. Macromol. Mater. Eng. 297, 1193–1202 (2012).
Capadona, J. et al. A versatile approach for the processing of polymer nanocomposites with self-assembled nanofibre templates. Nat. Nanotechnol. 2, 765–769 (2007).
Sridharan, A., Nguyen, J. K., Capadona, J. R. & Muthuswamy, J. Compliant intracortical implants reduce strains and strain rates in brain tissue in vivo. J. Neural Eng. 12, 036002 (2015).
Nguyen, J. K. et al. Mechanically-compliant intracortical implants reduce the neuroinflammatory response. J. Neural Eng. 11, 056014 (2014).
Karnaushenko, D. et al. Biomimetic microelectronics for regenerative neuronal cuff implants. Adv. Mater. 27, 6797–6805 (2015).
Takashi, D. et al. Ultrasmall implantable composite microelectrodes with bioactive surfaces for chronic neural interfaces. Nat. Mater. 11, 1065–1073 (2012).
Canales, A. et al. Multifunctional fibers for simultaneous optical, electrical and chemical interrogation of neural circuits in vivo. Nat. Biotechnol. 33, 277–284 (2015).
Lu, C. et al. Polymer fiber probes enable optical control of spinal cord and muscle function in vivo. Adv. Funct. Mater. 24, 6594–6600 (2014).
Liu, J. et al. Syringe-injectable electronics. Nat. Nanotechnol. 10, 629636 (2015).
Xie, C. et al. Three-dimensional macroporous nanoelectronic networks as minimally invasive brain probes. Nat. Mater. 14, 1286–1292 (2015).
Thomas, J. et al. Minimally invasive endovascular stent-electrode array for high-fidelity, chronic recordings of cortical neural activity. Nat. Biotechnol. 34, 320–327 (2016).
Discher, D. E., Janmey, P. & Wang, Y. L. Cells feel and respond to the stiffness of their substrate. Science 310, 1139–1143 (2005).
Georges, P. C., Miller, W. J., Meaney, D. F., Sawyer, E. S. & Janmey, P. A. Matrices with compliance comparable to that of brain tissue select neuronal over glial growth in mixed cortical cultures. Biophys. J. 90, 3012–3018 (2006).
Moshayedi, P. et al. The relationship between glial cell mechanosensitivity and foreign body reactions in the central nervous system. Biomaterials 35, 3919–3925 (2014).
Bollmann, L. et al. Microglia mechanics: immune activation alters traction forces and durotaxis. Front. Cell. Neurosci. 9, 363 (2015).
Franze, K. & Guck, J. The biophysics of neuronal growth. Rep. Progress Phys. 73, 094601 (2010).
Janmey, P. A. & Miller, R. T. Mechanisms of mechanical signaling in development and disease. J. Cell Sci. 124, 9–18 (2011).
McWhorter, F. Y., Davis, C. T. & Liu, W. F. Physical and mechanical regulation of macrophage phenotype and function. Cell. Mol. Life Sci. 72, 1303–1316 (2015).
Kozai, T. D. Y., Jaquins-Gerstl, A. S., Vazquez, A. L., Michael, A. C. & Cui, X. T. Dexamethasone retrodialysis attenuates microglial response to implanted probes in vivo. Biomaterials 87, 157–169 (2016).
Chikar, J. A. et al. The use of a dual PEDOT and RGD-functionalized alginate hydrogel coating to provide sustained drug delivery and improved cochlear implant function. Biomaterials 33, 1982–1990 (2012).
Aregueta-Robles, U. A. Woolley, A. J. Poole-Warren, L. A. Lovell, N. H. & Green, R. A. Organic electrode coatings for next-generation neural interfaces. Front. Neuroeng. 7, 15 (2014).
Khodagholy, D. et al. In vivo recordings of brain activity using organic transistors. Nat. Commun. 4, 1575 (2013).
Rivnay, J., Owens, R. s. n. M. & Malliaras, G. G. The rise of organic bioelectronics. Chem. Mater. 26, 679–685 (2014).
Williamson, A. et al. Controlling epileptiform activity with organic electronic ion pumps. Adv. Mater. 27, 3138–3144 (2015).
Jonsson, A. et al. Therapy using implanted organic bioelectronics. Sci. Adv. 1, e1500039 (2015).
Rylie, A. Green, Baek, Sungchul Poole-Warren, L. A. & Martens, P. J. Conducting polymer-hydrogels for medical electrode applications. Sci. Tech. Adv. Mater. 11, 014107 (2010).
Montgomery, K. L., Iyer, S. M., Christensen, A. J., Deisseroth, K. & Delp, S. L. Beyond the brain: optogenetic control in the spinal cord and peripheral nervous system. Sci. Transl. Med. 8, 337rv5 (2016).
Grosenick, L., Marshel, J. H. & Deisseroth, K. Closed-loop and activity-guided optogenetic control. Neuron 86, 106–139 (2015).
Jeong, J.-W. et al. Wireless optofluidic systems for programmable in vivo pharmacology and optogenetics. Cell 162, 1–13 (2015).
Courtine, G. et al. Transformation of nonfunctional spinal circuits into functional states after the loss of brain input. Nat. Neurosci. 12, 1333–1342 (2009).
Wenger, N. et al. Spatiotemporal neuromodulation therapies engaging muscle synergies improve motor control after spinal cord injury. Nat. Med. 22, 138–145 (2016).
McIntyre, C. C., Chaturvedi, A., Shamir, R. R. & Lempka, S. F. Engineering the next generation of clinical deep brain stimulation technology. Brain Stimul. 8, 21–26 (2015).
Courtine, G. & Bloch, J. Defining ecological strategies in neuroprosthetics. Neuron 86, 29–33 (2015).
Borton, D., Micera, S., Millan, J. d. R. & Courtine, G. Personalized neuroprosthetics. Sci. Transl. Med. 5, 210rv212 (2013).
Branner, A. & Normann, R. A. A multielectrode array for intrafascicular recording and stimulation in sciatic nerve of cats. Brain Res. Bull. 51, 293–306 (2000).
Rubehn, B., Bosman, C., Oostenveld, R., Fries, P. & Stiegltiz, T. S. A. MEMS-based flexible multichannel ECoG-electrode array. J. Neural Eng. 6, 036003 (2009).
Stieglitz, T., Beutel, H., Schuettler, M. & Meyer, J. U. Micromachined, polyimide-based devices for flexible neural interfaces. Biomed. Microdevices 2, 283–294 (2000).
Towne, C., Montgomery, K. L., Iyer, S. M., Deisseroth, K. & Delp, S. L. Optogenetic control of targeted peripheral axons in freely moving animals. PLoS One 8, e72691 (2013).
Boretius, T. et al. A transverse intrafascicular multichannel electrode (TIME) to interface with the peripheral nerve. Biosens. Bioelectron. 26, 62–69 (2010).
Raspopovic, S. et al. Restoring natural sensory feedback in real-time bidirectional hand prostheses. Sci. Transl. Med. 6, 222ra219 (2014).
Musick, K. M. et al. Chronic multichannel neural recordings from soft regenerative microchannel electrodes during gait. Sci. Rep. 5, 14363 (2015).
Acknowledgements
Financial support was provided by the Bertarelli Foundation (SPL), Starting Grants from the European Research Council (ERC 259419 ESKIN (SPL), ERC 261247, Walk Again (GC)), the European Community's Seventh Framework Program (CP-IP 258654, NeuWALK (GC)) and the Alexander-von-Humboldt Foundation (Alexander-von-Humboldt Professorship (JG)). The authors thank A. Goriely, K. Franze, P. Janmey, K. Van Vliet, J. Fawcett, R. Franklin, M. Reimer and J. Bloch for useful discussions.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Rights and permissions
About this article
Cite this article
Lacour, S., Courtine, G. & Guck, J. Materials and technologies for soft implantable neuroprostheses. Nat Rev Mater 1, 16063 (2016). https://doi.org/10.1038/natrevmats.2016.63
Published:
DOI: https://doi.org/10.1038/natrevmats.2016.63
This article is cited by
-
Shaping high-performance wearable robots for human motor and sensory reconstruction and enhancement
Nature Communications (2024)
-
3D spatiotemporally scalable in vivo neural probes based on fluorinated elastomers
Nature Nanotechnology (2024)
-
Semi-implantable device based on multiplexed microfilament electrode cluster for continuous monitoring of physiological ions
Bio-Design and Manufacturing (2024)
-
Innovative solutions for disease management
Bioelectronic Medicine (2023)
-
Bridging polymer chemistry and cryobiology
Polymer Journal (2023)