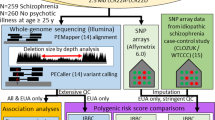
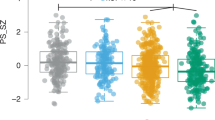
Abstract
Rare copy number variants contribute significantly to the risk for schizophrenia, with the 22q11.2 locus consistently implicated. Individuals with the 22q11.2 deletion syndrome (22q11DS) have an estimated 25-fold increased risk for schizophrenia spectrum disorders, compared to individuals in the general population. The International 22q11DS Brain Behavior Consortium is examining this highly informative neurogenetic syndrome phenotypically and genomically. Here we detail the procedures of the effort to characterize the neuropsychiatric and neurobehavioral phenotypes associated with 22q11DS, focusing on schizophrenia and subthreshold expression of psychosis. The genomic approach includes a combination of whole-genome sequencing and genome-wide microarray technologies, allowing the investigation of all possible DNA variation and gene pathways influencing the schizophrenia-relevant phenotypic expression. A phenotypically rich data set provides a psychiatrically well-characterized sample of unprecedented size (n=1616) that informs the neurobehavioral developmental course of 22q11DS. This combined set of phenotypic and genomic data will enable hypothesis testing to elucidate the mechanisms underlying the pathogenesis of schizophrenia spectrum disorders.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
CNV and Schizophrenia Working Groups of the Psychiatric Genomics Consortium. Contribution of copy number variants to schizophrenia from a genome-wide study of 41,321 subjects. Nat Genet 2017; 49: 27–35.
Walsh T, McClellan JM, McCarthy SE, Addington AM, Pierce SB, Cooper GM et al. Rare structural variants disrupt multiple genes in neurodevelopmental pathways in schizophrenia. Science 2008; 320: 539–543.
International Schizophrenia Consortium. Rare chromosomal deletions and duplications increase risk of schizophrenia. Nature 2008; 455: 237–241.
Bassett AS, Scherer SW, Brzustowicz LM . Copy number variations in schizophrenia: critical review and new perspectives on concepts of genetics and disease. Am J Psychiatry 2010; 167: 899–914.
Shprintzen RJ, Goldberg R, Golding-Kushner KJ, Marion RW . Late-onset psychosis in the velo-cardio-facial syndrome. Am J Med Genet 1992; 42: 141–142.
Chow EW, Bassett AS, Weksberg R . Velo-cardio-facial syndrome and psychotic disorders: implications for psychiatric genetics. Am J Med Genet A 1994; 54: 107–112.
Karayiorgou M, Morris MA, Morrow B, Shprintzen RJ, Goldberg R, Borrow J et al. Schizophrenia susceptibility associated with interstitial deletions of chromosome 22q11. Proc Natl Acad Sci USA 1995; 92: 7612–7616.
Gothelf D, Frisch A, Munitz H, Rockah R, Aviram A, Mozes T et al. Velocardiofacial manifestations and microdeletions in schizophrenic inpatients. Am J Med Genet A 1997; 72: 455–461.
Bassett AS, Hodgkinson K, Chow EW, Correia S, Scutt LE, Weksberg R . 22q11 deletion syndrome in adults with schizophrenia. Am J Med Genet 1998; 81: 328–337.
Bassett AS, Chow EW . 22q11 deletion syndrome: a genetic subtype of schizophrenia. Biol Psychiatry 1999; 46: 882–891.
Murphy KC, Jones LA, Owen MJ . High rates of schizophrenia in adults with velo-cardio-facial syndrome. Arch Gen Psychiatry 1999; 56: 940–945.
Costain G, Lionel AC, Merico D, Forsythe P, Russell K, Lowther C et al. Pathogenic rare copy number variants in community-based schizophrenia suggest a potential role for clinical microarrays. Hum Mol Genet 2013; 22: 4485–4501.
McDonald-McGinn DM, Sullivan KE, Marino B, Philip N, Swillen A, Vorstman JAS et al. 22q11.2 deletion syndrome. Nat Rev Dis Prim 2015; 1: 15071.
Insel TR . Rethinking schizophrenia. Nature 2010; 468: 187–193.
Merico D, Zarrei M, Costain G, Ogura L, Alipanahi B, Gazzellone MJ et al. Whole-genome sequencing suggests schizophrenia risk mechanisms in humans with 22q11.2 deletion syndrome. G3 2015; 5: 2453–2461.
Meechan DW, Maynard TM, Tucker ES, Fernandez A, Karpinski BA, Rothblat LA et al. Modeling a model: Mouse genetics, 22q11.2 Deletion Syndrome, and disorders of cortical circuit development. Prog Neurobiol 2015; 130: 1–28.
Puech A, Saint-Jore B, Funke B, Gilbert DJ, Sirotkin H, Copeland NG et al. Comparative mapping of the human 22q11 chromosomal region and the orthologous region in mice reveals complex changes in gene organization. Proc Natl Acad Sci USA 1997; 94: 14608–14613.
Guna A, Butcher NJ, Bassett AS . Comparative mapping of the 22q11.2 deletion region and the potential of simple model organisms. J Neurodev Disord 2015; 7: 18.
Shaikh TH, Kurahashi H, Saitta SC, O'Hare AM, Hu P, Roe BA et al. Chromosome 22-specific low copy repeats and the 22q11.2 deletion syndrome: genomic organization and deletion endpoint analysis. Hum Mol Genet 2000; 9: 489–501.
Edelmann L, Pandita RK, Spiteri E, Funke B, Goldberg R, Palanisamy N et al. A common molecular basis for rearrangement disorders on chromosome 22q11. Hum Mol Genet 1999; 8: 1157–1167.
Edelmann L, Pandita RK, Morrow BE . Low-copy repeats mediate the common 3- Mb deletion in patients with velo-cardio-facial syndrome. Am J Hum Genet 1999; 64: 1076–1086.
Schneider M, Debbané M, Bassett AS, Chow EWC, Fung WLA, Van den Bree MBM et al. Psychiatric disorders from childhood to adulthood in 22q11.2 deletion syndrome: results from the International Consortium on Brain and Behavior in 22q11.2 deletion syndrome. Am J Psychiat 2014; 171: 627–639.
Green T, Gothelf D, Glaser B, Debbane M, Frisch A, Kotler M et al. Psychiatric disorders and intellectual functioning throughout development in velocardiofacial (22q11.2 deletion) syndrome. J Am Acad Child Adolesc Psychiatry 2009; 48: 1060–1068.
Gur RE, Yi JJ, McDonald-McGinn DM, Tang SX, Calkins ME, Whinna D et al. Neurocognitive development in 22q11.2 deletion syndrome: comparison with youth having developmental delay and medical comorbidities. Mol Psychiatry 2014; 19: 1205–1211.
Tang SX, Yi JJ, Calkins ME, Whinna DA, Kohler CG, Souders MC et al. Psychiatric disorders in 22q11.2 deletion syndrome are prevalent but undertreated. Psychol Med 2014; 44: 1267–1277.
Niarchou M, Martin J, Thapar A, Owen MJ, van den Bree MB . The clinical presentation of attention deficit-hyperactivity disorder (ADHD) in children with 22q11.2 deletion syndrome. Am J Med Genet B Neuropsychiatr Genet 2015; 168: 730–738.
Morgan VA, Leonard H, Bourke J, Jablensky A . Intellectual disability co-occurring with schizophrenia and other psychiatric illness: population-based study. Br J Psychiatry 2008; 193: 364–372.
Karayiorgou M, Simon TJ, Gogos JA . 22q11.2 microdeletions: linking DNA structural variation to brain dysfunction and schizophrenia. Nat Rev Neurosci 2010; 11: 402–416.
Jonas RK, Montojo CA, Bearden CE . The 22q11.2 deletion syndrome as a window into complex neuropsychiatric disorders over the lifespan. Biol Psychiatry 2014; 75: 351–360.
Hiroi N, Takahashi T, Hishimoto A, Izumi T, Boku S, Hiramoto T . Copy number variation at 22q11.2: from rare variants to common mechanisms of developmental neuropsychiatric disorders. Mol Psychiatry 2013; 18: 1153–1165.
Tamura M, Mukai J, Gordon JA, Gogos JA . Developmental inhibition of Gsk3 rescues behavioral and neurophysiological deficits in a mouse model of schizophrenia predisposition. Neuron 2016; 89: 1100–1109.
Hamm JP, Peterka DS, Gogos JA, Yuste R . Altered cortical ensembles in mouse models of schizophrenia. Neuron 2017; 94: 153–167.
Bassett AS, Chow EW, Husted J, Weksberg R, Caluseriu O, Webb GD et al. Clinical features of 78 adults with 22q11 deletion syndrome. Am J Med Genet A 2005; 138: 307–313.
Murphy KC . Schizophrenia and velo-cardio-facial syndrome. Lancet 2002; 359: 426–430.
Bassett AS, Chow EW, AbdelMalik P, Gheorghiu M, Husted J, Weksberg R . The schizophrenia phenotype in 22q11 deletion syndrome. Am J Psychiat 2003; 160: 1580–1586.
Butcher N, Fung W, Fitzpatrick L, Guna A, Andrade D, Lang A et al. Response to clozapine in a clinically identifiable subtype of schizophrenia. Br J Psychiat 2015; 206: 484–491.
Dori N, Green T, Weizman A, Gothelf D . The effectiveness and safety of antipsychotic and antidepressant medications in individuals with 22q11.2 deletion syndrome. J Child Adolesc Psychopharmacol 2017; 27: 83–90.
Cannon TD, Cadenhead K, Cornblatt B, Woods SW, Addington J, Walker E et al. Prediction of psychosis in youth at high clinical risk: a multisite longitudinal study in North America. Arch Gen Psychiatry 2008; 65: 28–37.
Fusar-Poli P, Rutigliano G, Stahl D, Schmidt A, Ramella-Cravaro V, Hitesh S et al. Deconstructing pretest risk enrichment to optimize prediction of psychosis in individuals at clinical high risk. JAMA Psychiatry 2016; 73: 1260–1267.
Kelleher I, Connor D, Clarke MC, Devlin N, Harley M, Cannon M . Prevalence of psychotic symptoms in childhood and adolescence: a systematic review and meta-analysis of population-based studies. Psychol Med 2012; 42: 1857–1863.
Calkins ME, Moore TM, Merikangas KR, Burstein M, Satterthwaite TD, Bilker WB et al. The psychosis spectrum in a young U.S. community sample: findings from the Philadelphia Neurodevelopmental Cohort. World Psychiatry 2014; 13: 296–305.
Seidman LJ, Shapiro DI, Stone WS, Woodberry KA, Ronzio A, Cornblatt BA et al. Association of neurocognition with transition to psychosis: baseline functioning in the second phase of the North American prodrome longitudinal study. JAMA Psychiatry 2016; 73: 1239–1248.
Gur RC, Calkins ME, Satterthwaite TD, Ruparel K, Bilker WB, Moore TM et al. Neurocognitive growth charting in psychosis spectrum youths. JAMA Psychiatry 2014; 71: 366–374.
Satterthwaite TD, Vandekar SN, Wolf DH, Bassett DS, Ruparel K, Shehzad Z et al. Connectome-wide network analysis of youth with psychosis-spectrum symptoms. Mol Psychiatry 2015; 20: 1508–1515.
Hooper SR, Curtiss K, Schoch K, Keshavan MS, Allen A, Shashi V . A longitudinal examination of the psychoeducational, neurocognitive, and psychiatric functioning in children with 22q11.2 deletion syndrome. Res Dev Disabil 2013; 34: 1758–1769.
Duijff SN, Klaassen PW, Swanenburg de Veye HF, Beemer FA, Sinnema G, Vorstman JA . Cognitive and behavioral trajectories in 22q11DS from childhood into adolescence: a prospective 6-year follow-up study. Res Dev Disabil 2013; 34: 2937–2945.
Antshel KM, Fremont W, Ramanathan S, Kates WR . Predicting cognition and psychosis in young adults with 22q11.2 deletion syndrome. Schizophr Bull 2016; pii: sbw135 [e-pub ahead of print 25 October 2016].
Tang SX, Yi JJ, Moore TM, Calkins ME, Kohler CG, Whinna DA et al. Subthreshold psychotic symptoms in 22q11.2 deletion syndrome. J Am Acad Child Adolesc Psychiatry 2014; 53: 991–1000, e1002.
Armando M, Girardi P, Vicari S, Menghini D, Digilio MC, Pontillo M et al. Adolescents at ultra-high risk for psychosis with and without 22q11 deletion syndrome: a comparison of prodromal psychotic symptoms and general functioning. Schizophr Res 2012; 139: 151–156.
Gothelf D, Schneider M, Green T, Debbane M, Frisch A, Glaser B et al. Risk factors and the evolution of psychosis in 22q11.2 deletion syndrome: a longitudinal 2-site study. J Am Acad Child Adolesc Psychiatry 2013; 52: 1192–1203.
Kahn RS, Keefe RS . Schizophrenia is a cognitive illness: time for a change in focus. JAMA Psychiatry 2013; 70: 1107–1112.
Meier MH, Caspi A, Reichenberg A, Keefe RS, Fisher HL, Harrington H et al. Neuropsychological decline in schizophrenia from the premorbid to the postonset period: evidence from a population-representative longitudinal study. Am J Psychiatry 2014; 171: 91–101.
Cannon M, Caspi A, Moffitt TE, Harrington H, Taylor A, Murray RM et al. Evidence for early-childhood, pan-developmental impairment specific to schizophreniform disorder: results from a longitudinal birth cohort. Arch Gen Psychiatry 2002; 59: 449–456.
Vorstman JA, Breetvelt E, Duijff SN, Jalbrzikowsk M, Vogels A, Swillen A et al. Cognitive decline preceding the onset of psychosis in patients with 22q11.2 deletion syndrome. JAMA Psychiatry 2015; 72: 377–385.
Zai G, Robbins TW, Sahakian BJ, Kennedy JL . A review of molecular genetic studies of neurocognitive deficits in schizophrenia. Neurosci Biobehav Rev 2017; 72: 50–67.
Fusar-Poli P, Borgwardt S, Bechdolf A, Addington J, Riecher-Rossler A, Schultze-Lutter F et al. The psychosis high-risk state: a comprehensive state-of-the-art review. JAMA Psychiatry 2012; 70: 1–14.
Carrion RE, McLaughlin D, Auther AM, Olsen R, Correll CU, Cornblatt BA . The impact of psychosis on the course of cognition: a prospective, nested case-control study in individuals at clinical high-risk for psychosis. Psychol Med 2015; 45: 3341–3354.
Gur RC, Braff DL, Calkins ME, Dobie DJ, Freedman R, Green MF et al. Neurocognitive performance in family-based and case-control studies of schizophrenia. Schizophr Res 2015; 163: 17–23.
Gur RE, Nimgaonkar VL, Almasy L, Calkins ME, Ragland JD, Pogue-Geile MF et al. Neurocognitive endophenotypes in a multiplex multigenerational family study of schizophrenia. Am J Psychiatry 2007; 164: 813–819.
Duijff SN, Klaassen PW, de Veye HF, Beemer FA, Sinnema G, Vorstman JA . Cognitive development in children with 22q11.2 deletion syndrome. Br J Psychiatry 2012; 200: 462–468.
De Smedt B, Devriendt K, Fryns JP, Vogels A, Gewillig M, Swillen A . Intellectual abilities in a large sample of children with velo-cardio-facial syndrome: an update. J Intellect Disabil Res 2007; 51: 666–670.
Chow EW, Watson M, Young DA, Bassett AS . Neurocognitive profile in 22q11 deletion syndrome and schizophrenia. Schizophr Res 2006; 87: 270–278.
Miller TJ, McGlashan TH, Rosen JL, Cadenhead K, Cannon T, Ventura J et al. Prodromal assessment with the structured interview for prodromal syndromes and the scale of prodromal symptoms: predictive validity, interrater reliability, and training to reliability. Schizophr Bull 2003; 29: 703–715.
First MB, Spitzer RL, Gibbon M et al. Structured Clinical Interview for DSM‐IV‐TR Axis I Disorders, Research Version, Patient Edition. Biometrics Research, New York State Psychiatric Institute: New York, 2002.
Kaufman J, Birmaher B, Brent D, Rao U, Flynn C, Moreci P et al. Schedule for Affective Disorders and Schizophrenia for School-Age Children—Present and Lifetime Version (K-SADS-PL): initial reliability and validity data. J Am Acad Child Adolesc Psychiatry 1997; 36: 980–988.
Miller TJ, McGlashan TH, Rosen JL, Somjee L, Markovich PJ, Stein K et al. Prospective diagnosis of the initial prodrome for schizophrenia based on the Structured Interview for Prodromal Syndromes: preliminary evidence of interrater reliability and predictive validity. Am J Psychiatry 2002; 159: 863–865.
McGorry PD, Singh B Schizophrenia: risk and possibility. In: Burrows G (ed). Handbook of Studies on Preventive Psychiatry. Elsevier: Amsterdam, 1995, pp 491–514.
Meienberg J, Bruggmann R, Oexle K, Matyas G . Clinical sequencing: is WGS the better WES? Hum Genet 2016; 135: 359–362.
Johnston HR, Chopra P, Wingo TS, Patel V International Consortium on Brain and Behavior in 22q11.2 Deletion Syndrome Epstein MP et al. PEMapper and PECaller provide a simplified approach to whole-genome sequencing. Proc Natl Acad Sci USA 2017; 114: E1923–E1932.
Wang K, Li M, Hakonarson H . ANNOVAR: functional annotation of genetic variants from high-throughput sequencing data. Nucleic Acids Res 2010; 38: e164.
Genomes Project C, Auton A, Brooks LD, Durbin RM, Garrison EP, Kang HM et al. A global reference for human genetic variation. Nature 2015; 526: 68–74.
Tennessen JA, Bigham AW, O'Connor TD, Fu W, Kenny EE, Gravel S et al. Evolution and functional impact of rare coding variation from deep sequencing of human exomes. Science 2012; 337: 64–69.
Pollard KS, Hubisz MJ, Rosenbloom KR, Siepel A . Detection of nonneutral substitution rates on mammalian phylogenies. Genome Res 2010; 20: 110–121.
Yuen RK, Thiruvahindrapuram B, Merico D, Walker S, Tammimies K, Hoang N et al. Whole-genome sequencing of quartet families with autism spectrum disorder. Nat Med 2015; 21: 185–191.
Karczewski KJ, Weisburd B, Thomas B, Solomonson M, Ruderfer DM, Kavanagh D et al. The ExAC browser: displaying reference data information from over 60 000 exomes. Nucleic Acids Res 2017; 45: D840–D845.
Kircher M, Witten DM, Jain P, O'Roak BJ, Cooper GM, Shendure J . A general framework for estimating the relative pathogenicity of human genetic variants. Nat Genet 2014; 46: 310–315.
Adzhubei IA, Schmidt S, Peshkin L, Ramensky VE, Gerasimova A, Bork P et al. A method and server for predicting damaging missense mutations. Nat Methods 2010; 7: 248–249.
Gene Ontology C, Blake JA, Dolan M, Drabkin H, Hill DP, Li N et al. Gene ontology annotations and resources. Nucleic Acids Res 2013; 41: D530–D535.
Zhu M, Need AC, Han Y, Ge D, Maia JM, Zhu Q et al. Using ERDS to infer copy-number variants in high-coverage genomes. Am J Hum Genet 2012; 91: 408–421.
Abyzov A, Urban AE, Snyder M, Gerstein M . CNVnator: an approach to discover, genotype, and characterize typical and atypical CNVs from family and population genome sequencing. Genome Res 2011; 21: 974–984.
Ye K, Schulz MH, Long Q, Apweiler R, Ning Z . Pindel: a pattern growth approach to detect break points of large deletions and medium sized insertions from paired-end short reads. Bioinformatics 2009; 25: 2865–2871.
Wang J, Mullighan CG, Easton J, Roberts S, Heatley SL, Ma J et al. CREST maps somatic structural variation in cancer genomes with base-pair resolution. Nat Methods 2011; 8: 652–654.
Mlynarski EE, Sheridan MB, Xie M, Guo T, Racedo SE, McDonald-McGinn DM et al. Copy-number variation of the glucose transporter gene SLC2A3 and congenital heart defects in the 22q11.2 deletion syndrome. Am J Hum Genet 2015; 96: 753–764.
Mlynarski EE, Xie M, Taylor D, Sheridan MB, Guo T, Racedo SE et al. Rare copy number variants and congenital heart defects in the 22q11.2 deletion syndrome. Hum Genet 2016; 135: 273–285.
Schizophrenia Working Group of the PGC. Biological insights from 108 schizophrenia-associated genetic loci. Nature 2014; 511: 421–427.
Bassett AS, Marshall CR, Lionel AC, Chow EW, Scherer SW . Copy number variations and risk for schizophrenia in 22q11.2 deletion syndrome. Hum Mol Genet 2008; 17: 4045–4053.
Bassett AS, Lowther C, Merico D, Costain G, Chow EWC, Van Amelsvoort T et al. Rare genome-wide copy number variation affects expression of schizophrenia in 22q11.2 deletion syndrome. Am J Psychiatry; e-pub ahead of print 28 July 2017.
Hestand MS, Nowakowska BA, Vergaelen E, Van Houdt J, Dehaspe L, Suhl JA et al. A catalog of hemizygous variation in 127 22q11 deletion patients. Hum Genome Var 2016; 3: 15065.
Guo X, Delio M, Haque N, Castellanos R, Hestand MS, Vermeesch JR et al. Variant discovery and breakpoint region prediction for studying the human 22q11.2 deletion using BAC clone and whole genome sequencing analysis. Hum Mol Genet 2016; 25: 3754–3767.
Bailey JA, Yavor AM, Viggiano L, Misceo D, Horvath JE, Archidiacono N et al. Human-specific duplication and mosaic transcripts: the recent paralogous structure of chromosome 22. Am J Hum Genet 2002; 70: 83–100.
Babcock M, Pavlicek A, Spiteri E, Kashork CD, Ioshikhes I, Shaffer LG et al. Shuffling of genes within low-copy repeats on 22q11 (LCR22) by Alu-mediated recombination events during evolution. Genome Res 2003; 13: 2519–2532.
Lee S, Emond MJ, Bamshad MJ, Barnes KC, Rieder MJ, Nickerson DA et al. Optimal unified approach for rare-variant association testing with application to small-sample case-control whole-exome sequencing studies. Am J Hum Genet 2012; 91: 224–237.
Lin JR, Cai Y, Zhang Q, Zhang W, Nogales-Cadenas R, Zhang ZD . Integrated post-GWAS analysis sheds new light on the disease mechanisms of schizophrenia. Genetics 2016; 204: 1587–1600.
Dickson H, Laurens KR, Cullen AE, Hodgins S . Meta-analyses of cognitive and motor function in youth aged 16 years and younger who subsequently develop schizophrenia. Psychol Med 2012; 42: 743–755.
Weisman O, Guri Y, Gur RE, McDonald-McGinn DM, Calkins MET S . Subthreshold psychosis in 22q11.2 deletion syndrom: multi-site naturalistic study. Schizophr Bull, doi: 10.1093/schbul/sbx005 [e-pub ahead of print 13 February 2017].
Lionel AC, Crosbie J, Barbosa N, Goodale T, Thiruvahindrapuram B, Rickaby J et al. Rare copy number variation discovery and cross-disorder comparisons identify risk genes for ADHD. Sci Transl Med 2011; 3: 95ra75.
Scherer SW, Lee C, Birney E, Altshuler DM, Eichler EE, Carter NP et al. Challenges and standards in integrating surveys of structural variation. Nat Genet 2007; 39: S7–S15.
Korn JM, Kuruvilla FG, McCarroll SA, Wysoker A, Nemesh J, Cawley S et al. Integrated genotype calling and association analysis of SNPs, common copy number polymorphisms and rare CNVs. Nat Genet 2008; 40: 1253–1260.
Pinto D, Pagnamenta AT, Klei L, Anney R, Merico D, Regan R et al. Functional impact of global rare copy number variation in autism spectrum disorders. Nature 2010; 466: 368–372.
Silversides CK, Lionel AC, Costain G, Merico D, Migita O, Liu B et al. Rare copy number variations in adults with tetralogy of Fallot implicate novel risk gene pathways. PLoS Genet 2012; 8: e1002843.
Marshall CR, Noor A, Vincent JB, Lionel AC, Feuk L, Skaug J et al. Structural variation of chromosomes in autism spectrum disorder. Am J Hum Genet 2008; 82: 477–488.
Acknowledgements
The IBBC is supported by the National Institute of Mental Health grants USA U01MH101719, U01MH0101720, CAN+: U01MH0101723, EUA: U01MH101722, EUB: U01MH101724. Additional support includes: NIH R01MH087626 (REG), R01MH087636 (BSE, DMM-M), R01MH085953 (CEB), Simons Foundation Explorer Award (CEB), RO1MH064824 (WRK), P01HD070454 (BEM, BSE, DMM-M), R01MH107018 (TJS), NIH U54 HD079125 MIND Institute Intellectual and Developmental Disabilities Research Center (TJS), the Emory Integrated Genomics Core (EIGC), which is subsidized by the Emory University School of Medicine and is one of the Emory Integrated Core Facilities, National Center for Advancing Translational Sciences of the National Institutes of Health under Award Number UL1TR000454, RW Woodruff Fund (JFC, OYO), the Canadian Institutes of Health Research (ASB, EWCC, MOP-74631, MOP-97800; ASB, MOP-111238), the Canada Research Chair in Schizophrenia Genetics and Genomic Disorders (ASB), Brain Canada Mental Health Training Award (NJB), Baily Thomas Charitable Trust (2315/1) (MvdB), The Waterloo Foundation (WF918-1234) (MvdB), Wellcome Trust ISSF grant (MvdB), A Medical Research Council (UK) Centre grant G0800509 (MO), CIBERSAM (CA), the Binational Science Foundation, grant number 2011378 (DG), FONDECYT-Chile #1130392 and 1171014 (GMR), Wellcome Trust 110222/Z/15/Z (MN), The FWO grant G.0E11.17 N (JRV), the Brain and Behavior Research Foundation 22597 (AW), the Swiss National Science Foundation (324730_144260) (SE), the EU IMI initiative EU AIMS (DM), the Brain and Behavior Research Foundation (JV), the Brain and Behavior Research Foundation 21278 (MA), the NHMRC 1072593 (AL). We thank the research teams across the international sites and the individuals and family members who have enabled the establishment of the Consortium and have contributed to advancing the understanding and treatment of 22q11DS.
Author information
Authors and Affiliations
Consortia
Corresponding authors
Ethics declarations
Competing interests
DMM-M has given lectures on 22q11DS for Natera; CA has been a consultant to or has received honoraria or grants from Abbot, AMGEN, AstraZeneca, CIBERSAM, Dainippon Sumitomo Pharma, Fundación Alicia Koplowitz, Forum, Instituto de Salud Carlos III, Gedeon Richter, Janssen Cilag, Lundbeck, Ministerio de Ciencia e Innovación, Ministerio de Sanidad, Ministerio de Economía y Competitividad, Mutua Madrileña, Otsuka, Pfizer, Roche, Servier, Shire, Schering Plough, Sunovio and Takeda. DF has been a consultant and/or advisor to or has received honoraria from AstraZeneca, Bristol-Myers Squibb, Eisai, Janssen, Lundbeck, Otsuka and Ministerio de Ciencia e Innovación, Ministerio de Sanidad, Ministerio de Economía. The other authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Molecular Psychiatry website
Supplementary information
PowerPoint slides
Rights and permissions
About this article
Cite this article
Gur, R., Bassett, A., McDonald-McGinn, D. et al. A neurogenetic model for the study of schizophrenia spectrum disorders: the International 22q11.2 Deletion Syndrome Brain Behavior Consortium. Mol Psychiatry 22, 1664–1672 (2017). https://doi.org/10.1038/mp.2017.161
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/mp.2017.161
This article is cited by
-
22q11.2 Deletion-Associated Blood-Brain Barrier Permeability Potentiates Systemic Capillary Leak Syndrome Neurologic Features
Journal of Clinical Immunology (2024)
-
A genetics-first approach to understanding autism and schizophrenia spectrum disorders: the 22q11.2 deletion syndrome
Molecular Psychiatry (2023)
-
Rare coding variants as risk modifiers of the 22q11.2 deletion implicate postnatal cortical development in syndromic schizophrenia
Molecular Psychiatry (2023)
-
Stimulant treatment effectiveness, safety and risk for psychosis in individuals with 22q11.2 deletion syndrome
European Child & Adolescent Psychiatry (2022)
-
A normative chart for cognitive development in a genetically selected population
Neuropsychopharmacology (2022)