Abstract
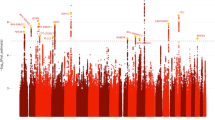
We report a genome-wide association study (GWAS) for cocaine dependence (CD) in three sets of African- and European-American subjects (AAs and EAs, respectively) to identify pathways, genes and alleles important in CD risk. The discovery GWAS data set (n=5697 subjects) was genotyped using the Illumina OmniQuad microarray (8 90 000 analyzed single-nucleotide polymorphisms (SNPs)). Additional genotypes were imputed based on the 1000 Genomes reference panel. Top-ranked findings were evaluated by incorporating information from publicly available GWAS data from 4063 subjects. Then, the most significant GWAS SNPs were genotyped in 2549 independent subjects. We observed one genome-wide-significant (GWS) result: rs2629540 at the FAM53B (‘family with sequence similarity 53, member B’) locus. This was supported in both AAs and EAs; P-value (meta-analysis of all samples)=4.28 × 10−8. The gene maps to the same chromosomal region as the maximum peak we observed in a previous linkage study. NCOR2 (nuclear receptor corepressor 2) SNP rs150954431 was associated with P=1.19 × 10−9 in the EA discovery sample. SNP rs2456778, which maps to CDK1 (‘cyclin-dependent kinase 1’), was associated with cocaine-induced paranoia in AAs in the discovery sample only (P=4.68 × 10−8). This is the first study to identify risk variants for CD using GWAS. Our results implicate novel risk loci and provide insights into potential therapeutic and prevention strategies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Compton WM, Thomas YF, Stinson FS, Grant BF . Prevalence, correlates, disability, and comorbidity of DSM-IV drug abuse and dependence in the United States: results from the National Epidemiologic Survey on Alcohol and Related Conditions. Arch Gen Psychiatry 2007; 64: 566–576.
Lobo MK, Covington HE III, Chaudhury D, Friedman AK, Sun HS, Damez-Werno D et al. Cell type specific loss of BDNF signaling mimics optogenetic control of cocaine reward. Science 2010; 330: 385–390.
Kendler KS, Prescott CA . Cocaine use, abuse and dependence in a population-based sample of female twins. Br J Psychiatry 1998; 173: 345–350.
Kendler KS, Karkowski LM, Neale MC, Prescott CA . Illicit psychoactive substance use, heavy use, abuse, and dependence in a US population-based sample of male twins. Arch Gen Psychiatry 2000; 57: 261–269.
Gelernter J, Panhuysen C, Weiss R, Brady K, Hesselbrock V, Rounsaville B et al. Genomewide linkage scan for cocaine dependence and related traits: linkages for a cocaine-related trait and cocaine-induced paranoia. Am J Med Genet Neuropsych Genet 2005; 136: 45–52.
Yang BZ, Han S, Kranzler HR, Farrer LA, Elston RC, Gelernter J . Autosomal linkage scan for loci predisposing to comorbid dependence on multiple substances. Am J Med Genet B Neuropsychiatr Genet 2012; 159B: 361–369.
Gelernter J, Kranzler HR, Sherva R, Koesterer R, Sun J, Bi J Genomewide association study of opioid dependence and related traits: multiple associations mapped to calcium and potassium pathways, in review.
Agrawal A, Lynskey MT, Hinrichs A, Grucza R, Saccone SF, Krueger R et al. A genome-wide association study of DSM-IV cannabis dependence. Addict Biol 2011; 16: 514–518.
Pierucci-Lagha A, Gelernter J, Feinn R, Cubells JF, Pearson D, Pollastri A et al. Diagnostic reliability of the Semi-Structured Assessment for Drug Dependence and Alcoholism (SSADDA). Drug Alcohol Depend 2005; 80: 303–312.
American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders 4th edn. American Psychiatric Press: Washington, DC, 1994.
Cubells JF, Feinn R, Pearson D, Burda J, Tang Y, Farrer LA et al. Rating the severity and character of transient cocaine-induced delusions and hallucinations with a new instrument, the Scale for Assessment of Positive Symptoms for Cocaine-Induced Psychosis (SAPS-CIP). Drug Alcohol Depend 2005; 80: 23–33.
Holland PM, Abramson RD, Watson R, Gelfand DH . Detection of specific polymerase chain reaction product by utilizing the 5′→3′ exonuclease activity of Thermus aquaticus DNA polymerase. Proc Natl Acad Sci USA 1991; 88: 7276–7280.
Price AL, Patterson NJ, Plenge RM, Weinblatt ME, Shadick NA, Reich D . Principal components analysis corrects for stratification in genome-wide association studies. Nat Genet 2006; 38: 904–909.
Patterson N, Price AL, Reich D . Population structure and eigenanalysis. PLoS Genet. 2006; 2: e190.
Hartigan JA, Wong MAA . K-means clustering algorithm. Appl Statist 1979; 28: 100–108.
Edenberg HJ . The collaborative study on the genetics of alcoholism: an update. Alcohol Res Health 2002; 26: 214–218.
Bierut LJ, Strickland JR, Thompson JR, Afful SE, Cottler LB . Drug use and dependence in cocaine dependent subjects, community-based individuals, and their siblings. Drug Alcohol Depend 2008; 95: 14–22.
Bierut LJ . Genetic variation that contributes to nicotine dependence. Pharmacogenomics 2007; 8: 881–883.
Howie BN, Donnelly P, Marchini JA . Flexible and accurate genotype imputation method for the next generation of genome-wide association studies. PLoS Genet 2009; 5: e1000529.
1000 Genomes Project Consortium. A map of human genome variation from population-scale sequencing. Nature 2010; 467: 1061–1073.
Brady KT, Lydiard RB, Malcolm R, Ballenger JC . Cocaine-induced psychosis. J Clin Psychiatry 1991; 52: 509–512.
Satel SL, Southwick SM, Gawin FH . Clinical features of cocaine-induced paranoia. Am J Psychiatry 1991; 148: 495–498.
Cubells JF, Feinn R, Pearson D, Burda J, Tang Y, Farrer LA et al. Rating the severity and character of transient cocaine-induced delusions and hallucinations with a new instrument, the Scale for Assessment of Positive Symptoms for Cocaine-Induced Psychosis (SAPS-CIP). Drug Alcohol Depend 2005; 80: 23–33.
Farrer LA, Kranzler HR, Yu Y, Weiss RD, Brady KT, Cubells JF et al. Association of variants in MANEA with cocaine-related behaviors. Arch Gen Psychiat 2009; 3: 267–274.
Willer CJ, Li Y, Abecasis GR . METAL: fast and efficient meta-analysis of genomewide association scans. Bioinformatics 2010; 26: 2190–2191.
Li J, Ji L . Adjusting multiple testing in multilocus analyses using the eigenvalues of a correlation matrix. Heredity (Edinb) 2005; 95: 221–227.
Thermes V, Candal E, Alunni A, Serin G, Bourrat F, Joly JS . Medaka simplet (FAM53B) belongs to a family of novel vertebrate genes controlling cell proliferation. Development 2006; 133: 1881–1890.
Hinrichs AL, Wang JC, Bufe B, Kwon JM, Budde J, Allen R et al. Functional variant in a bitter-taste receptor (hTAS2R16) influences risk of alcohol dependence. Am J Hum Genet 2006; 78: 103–111.
Multani PK, Clarke TK, Narasimhan S, Ambrose-Lanci L, Kampman KM, Pettinati HM et al. Neuronal calcium sensor-1 and cocaine addiction: a genetic association study in African-Americans and European Americans. Neurosci Lett 2012; 531: 46–51.
Wang JC, Foroud T, Hinrichs AL, Le NX, Bertelsen S, Budde JP et al. A genome-wide association study of alcohol-dependence symptom counts in extended pedigrees identifies C15orf53. Mol Psychiatry, advance online publication, 23 October 2012 doi:10.1038/mp.2012.143(e-pub ahead of print).
Hart AB, Engelhardt BE, Wardle MC, Sokoloff G, Stephens M et al. Genome-wide association study of d-amphetamine response in healthy volunteers identifies putative associations, including cadherin 13 (CDH13). PLoS One 2012; 7: e42646.
Nishizawa D, Fukuda K, Kasai S, Hasegawa J, Aoki Y, Nishi A et al. Genome-wide association study identifies a potent locus associated with human opioid sensitivity. Mol Psychiatry, advance online publication, 27 November 2012 doi:10.1038/mp.2012.164(e-pub ahead of print).
Acknowledgements
We appreciate the work in recruitment and assessment provided at McLean Hospital by Roger Weiss at the Medical University of South Carolina by Kathleen Brady and Raymond Anton and at the University of Pennsylvania by David Oslin. Genotyping services for a part of our GWAS study were provided by the Center for Inherited Disease Research (CIDR) and Yale University (Center for Genome Analysis). CIDR is fully funded through a federal contract from the National Institutes of Health to The Johns Hopkins University (contract number N01-HG-65403). We are grateful to Ann Marie Lacobelle, Michelle Cucinelli, Christa Robinson and Greg Dalton-Kay for their excellent technical assistance, to the SSADDA interviewers, led by Yari Nuñez and Michelle Slivinsky, who devoted substantial time and effort to phenotype the study sample and to John Farrell for database management assistance. This study was supported by National Institutes of Health Grants RC2 DA028909, R01 DA12690, R01 DA12849, R01 DA18432, R01 AA11330, R01 AA017535 and the VA Connecticut and Philadelphia VA MIRECCs. The publicly available data sets used for the analyses described in this manuscript were obtained from dbGaP at http://www.ncbi.nlm.nih.gov/projects/gap/cgi-bin/study.cgi?study_id=phs000092.v1.p1 through dbGaP accession number phs000092.v1.p. Funding support for the Study of Addiction: Genetics and Environment (SAGE) was provided through the NIH Genes, Environment and Health Initiative (GEI; U01 HG004422). SAGE is one of the genome-wide association studies funded as part of the Gene Environment Association Studies (GENEVA) under GEI. Assistance with phenotype harmonization and genotype cleaning, as well as with general study coordination, was provided by the GENEVA Coordinating Center (U01 HG004446). Assistance with data cleaning was provided by the National Center for Biotechnology Information. Support for collection of data sets and samples was provided by the Collaborative Study on the Genetics of Alcoholism (COGA; U10 AA008401), the Collaborative Genetic Study of Nicotine Dependence (COGEND; P01 CA089392) and the Family Study of Cocaine Dependence (FSCD; R01 DA013423). Funding support for genotyping, which was performed at the Johns Hopkins University Center for Inherited Disease Research, was provided by the NIH GEI (U01HG004438), the National Institute on Alcohol Abuse and Alcoholism, the National Institute on Drug Abuse and the NIH contract ‘High throughput genotyping for studying the genetic contributions to human disease’ (HHSN268200782096C).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Although unrelated to the current study, Dr Kranzler has been a consultant or advisory board member for Alkermes, Lilly, Lundbeck, Pfizer and Roche. He is also a member of the American Society of Clinical Psychopharmacology’s Alcohol Clinical Trials Initiative, which is supported by Lilly, Lundbeck, Abbott and Pfizer.
Additional information
Supplementary Information accompanies the paper on the Molecular Psychiatry website
Rights and permissions
About this article
Cite this article
Gelernter, J., Sherva, R., Koesterer, R. et al. Genome-wide association study of cocaine dependence and related traits: FAM53B identified as a risk gene. Mol Psychiatry 19, 717–723 (2014). https://doi.org/10.1038/mp.2013.99
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/mp.2013.99
Keywords
This article is cited by
-
Voltage-gated potassium channels control extended access cocaine seeking: a role for nucleus accumbens astrocytes
Neuropsychopharmacology (2024)
-
METH exposure alters sperm DNA methylation in F0 mice and mPFC transcriptome in male F1 mice
Psychopharmacology (2024)
-
Genotype-by-diagnosis interaction influences self-control in human cocaine addiction
Translational Psychiatry (2023)
-
Brain scans from 21,297 individuals reveal the genetic architecture of hippocampal subfield volumes
Molecular Psychiatry (2020)
-
Genomewide Study of Epigenetic Biomarkers of Opioid Dependence in European- American Women
Scientific Reports (2019)