Abstract
A number of large, rare copy number variants (CNVs) are deleterious for neurodevelopmental disorders, but large, rare, protective CNVs have not been reported for such phenotypes. Here we show in a CNV analysis of 47 005 individuals, the largest CNV analysis of schizophrenia to date, that large duplications (1.5–3.0 Mb) at 22q11.2—the reciprocal of the well-known, risk-inducing deletion of this locus—are substantially less common in schizophrenia cases than in the general population (0.014% vs 0.085%, OR=0.17, P=0.00086). 22q11.2 duplications represent the first putative protective mutation for schizophrenia.
Similar content being viewed by others
Main
Large, rare copy number variants (CNVs) at several genomic loci increase risk for schizophrenia and other neurodevelopmental disorders including intellectual disability (ID), autism spectrum disorders and attention-deficit hyperactivity disorder.1, 2 The first CNV to be unequivocally implicated in schizophrenia was the 22q11.2 deletion, which also causes most cases of DiGeorge and Velocardiofacial Syndromes (OMIM #188400 and #192430).3 The 22q11.2 deletion is estimated to occur in about 1 in 4000 live births4, and is one of the most common CNVs associated with ID.1 It is also the strongest known specific risk factor for developing schizophrenia in adulthood.5, 6 22q11.2del is also associated with psychiatric problems in childhood such as attention-deficit hyperactivity disorder, autism, depression and anxiety as well as a range of physical phenotypes.7
CNVs arise at this locus from non-allelic homologous recombination between low copy repeats8 and, as a result, the deletions occur in a set of low copy repeat-dependent sizes, the majority being about 3 Mb, and most of the remainder (<10%) being nested deletions of 1.5 Mb within that region.9 Even among those with the same sized CNV, the phenotype of 22q11.2del carriers is highly heterogeneous with respect to physical, psychiatric and cognitive sequelae,9 suggesting the involvement of other genetic, environmental or stochastic factors.
Given the non-allelic homologous recombination mechanism by which deletions are produced, it is not surprising that reciprocal 22q11.2 duplications also arise in human populations,10 although to date fewer such events have been ascertained. There are several reports that the phenotypic spectrum of 22q11.2dup is wide, including apparently unaffected transmitting parents.11, 12 As well as having a variety of physical manifestations, 22q11.2dup is reported to associate with ID and developmental delay in children1, 12, 13 and a wide range of psychiatric and behavioral abnormalities have been reported including attention-deficit hyperactivity disorder and autism, as well as other social and behavioral problems.1, 12, 14 The prevalence of the duplication in adults with psychiatric disorders has not been widely studied.
In the present study, we have established the rate of 22q11.2dup in a discovery sample of 6882 schizophrenia cases and 11 255 controls. The schizophrenia cases were genotyped on Illumina HumanOmniExpress-12v1 or HumanOmniExpressExome-8v1 arrays, and have been described elsewhere15 (Supplementary Material). The controls were obtained from four non-psychiatric data sets available through repositories, also genotyped on Illumina arrays (Supplementary Material). CNVs were detected using PennCNV.16 The probe set used for CNV calling was restricted to those common to all arrays used (520 766 probes). Full details of CNV calling and quality control are provided in the Supplementary Material. It should be noted that CNVs larger than 1 Mb are readily called with essentially perfect sensitivity by PennCNV on almost any SNP genotyping array. Significance of association was evaluated using a Fisher’s exact test or a Cochran–Mantel–Haenszel test stratified by ethnicity and study. We also used RNAseq to determine the mRNA abundance in lymphoblastoid cell lines for 31 genes across the 22q11.2 CNV region (chr22:18 893 541–21 901 736, hg19) and genes 3 Mb either side, in 16 carriers of 22q11.21del, 6 carriers of 22q11.2dup and 821 individuals without a CNV at this locus.
In our discovery sample, we found no 22q11.2 duplications in schizophrenia cases (0%) but 10 (0.089%) in controls (Figure 1a, Table 1, Fisher’s exact P=0.017). By way of contrast, we found reciprocal 22q11.2 deletions (a known strong risk factor) in 20 schizophrenia cases (0.29%) but in zero controls. No other CNV at any locus in the genome was found to be a putative protective factor at a nominal level of significance in this sample (data not shown).
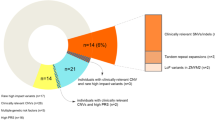
(a) Location of 22q11.2 duplications in discovery cases and replication cases and controls that cover the 1.5 Mb nested region. (b) Log2 RPKM mRNA abundance for genes in the 22q11 deletion and duplication carriers normalized to diploid subjects. Each gene (their positions depicted in alternating white or gray bars) has three measurements: deletion carriers, diploid subjects and duplication carriers. Genes are shown in map order, though their relative position is not drawn to scale. CNV, copy number variant.
Expression analysis of 22q11.2 deletion and duplication carriers indicated that the great majority of genes within the CNV region showed the expected increase or decrease in gene dosage, and that expression of genes flanking the CNV were not significantly affected by copy number change (Figure 1b). There have been relatively few systematic studies of gene expression in human 22q11.2 deletions and none of duplication carriers. Our data are highly congruent with a transcriptome-wide microarray study of RNA from untransformed peripheral blood mononuclear cells in showing significantly reduced expression of genes in the deleted region17 (further details in Supplementary Material).
In order to critically evaluate the reduced frequency of 22q11.2dup in cases in additional cohorts, we obtained data from the largest available CNV data sets known to us, in total comprising 14 256 additional cases and 14 612 additional controls. In this independent cohort, 22q11.2dups were also significantly rarer in cases than controls (0.021% vs 0.082%, Fisher’s exact test P=0.020 and Table 1). A combined analysis of discovery and replication data found 22q11.2dup in 0.014% of cases and 0.085% of controls (Fisher’s exact: P=0.00086, Cochran–Mantel–Haenszel: P=0.0019, OR=0.17, 95% confidence interval=0.05–0.56). The age of onset for the schizophrenia cases carrying the 22q11.2dup was 34, 28 and 43 years. Two of these cases have a history of seizures and none of them had any additional known pathogenic CNVs. Of the controls with a 22q11.2dup for which we have psychiatric data (n=6 from MGS sample), none had histories compatible with schizophrenia, schizoaffective disorder or major affective disorder. All graduated from high school and several had higher education degrees (confirming the notion that the duplication has incomplete penetrance for developmental delay).
Our study therefore identifies as the first putative protective mutation for schizophrenia duplications of the genomic segment that, when deleted, is the most potent genetic risk factor for the disorder. We can discount on several grounds the alternative explanation that those with 22q11.2dup have such a severe neurodevelopmental phenotype as to preclude a diagnosis of schizophrenia. First, as noted above, unaffected carriers are observed frequently (0.1% of controls across a large number of studies of psychiatric and non-psychiatric phenotypes). Second, the highly variable phenotype observed in 22q11.2dup carriers is often milder than that seen with many other schizophrenia-associated CNVs, as evidenced by its inheritance from an apparently unaffected parent in 69–74% of cases.11, 12, 18, 19 Most relevant to the present study, 22q11.2 deletions have consistently been reported to be associated with severe cognitive phenotypes such as autism and ID,20 but are seen at an appreciable rate in schizophrenia cohorts, including in the current samples (see above). If the duplication resulted in phenotypes inconsistent with inclusion in studies, we would expect greater depletion in controls, which are typically screened for health more intensively than cases, consistent with the finding of no 22q11 deletions among our controls. Finally, in the genome-wide analysis of our discovery sample, we also obtained evidence to support the association of deletions at 1q21.1, NRXN1, 3q29, 15q11.2, 15q13.3, 17q12 and 22q11.2 and duplications at 1q21.1, Williams–Beuren syndrome region, Prader–Willi/Angleman syndrome region, 16p13.11 and 16p11.2.21 These CNVs have all been associated with a similar range of neurodevelopmental phenotypes and our findings therefore argue strongly against the conclusion that our methods of ascertainment precluded the inclusion of cases carrying 22q11.2dup.
In all, 3 out of the 21 138 cases tested carried the 3 Mb 22q11.2 duplication, indicating that its putative protective effect is incomplete. However, our data suggest that its strength of effect may be strong, with an odds ratio less than 0.20 and an upper confidence limit of 0.56. The three case carriers had no additional pathogenic CNVs and we must assume that the development of schizophrenia resulted from other genetic or environmental factors. It was not possible to identify any particular characteristics of the cases carrying duplications but given their rarity power to do so was extremely limited.
To our knowledge, 22q11.2dup is the first putative protective mutation for schizophrenia that has been described in the literature. Our study suggests the existence of one or more dosage-sensitive gene in the duplication with the capacity to reduce risk of schizophrenia with implications here for further studies aimed at identifying targets for treating the disorder. This finding is of additional interest as this mutation does not appear to be similarly protective against the other neurodevelopmental phenotypes with which schizophrenia-associated CNVs are frequently associated.1 The present study in contrast provides a clear, opposite-direction dissociation between schizophrenia risk and both ID and autism spectrum disorder, both of which are more common in 22q11.2dup than in controls (Table 1). Neither the gene(s) nor the brain mechanisms by which 22q11.2del confers increased risk of neuropsychiatric and neurodevelopmental outcomes are known.22 However, that deletions are congruent in increasing risk of the disorders, whereas duplications act incongruently, simultaneously protecting against schizophrenia but predisposing to other neurodevelopmental disorders, suggests that at least some of the brain mechanisms are selective for schizophrenia. Moreover, that schizophrenia risk can potentially be reduced by a lesion that increases neurodevelopmental adversity (indexed by autism spectrum disorder and ID risk), suggests that the dosage-sensitive gene or genes might not just point the way to treatment, it may also hold clues to enhancing resilience among those who would generally be thought to be of elevated risk of the disorder.
Although large CNVs associate to risk of many disorders, it is generally unknown whether such effects arise from alterations of gene dosage or from other mechanisms. The lack (to date) of point mutations that phenocopy the neurodevelopmental and psychiatric effects of large deletions has invited alternative hypotheses, such as large-scale disruptions of chromatin or chromosomal pairing. Our finding that the reciprocal deletion and duplication of the same locus have potent risk and potential protective effects, respectively, for schizophrenia supports the hypothesis that one or more genes at 22q11.2 are dosage sensitive.22 The identification of the dosage-sensitive gene(s) at 22q11.2 and the implication of risk and protective mechanisms is therefore an important direction for research—particularly as pharmacological intervention might offer protection from schizophrenia. Our study was limited in its scope to implicate specific genes and possible mechanisms because CNVs at this locus affect multiple genes and the three duplications observed in cases of schizophrenia all involved the whole 3 Mb region. Further genetic studies on larger samples might inform this issue but it seems likely that a detailed understanding will only be provided by mechanistic studies in deletion and duplication carriers and in animal and cellular models.
References
Malhotra D, Sebat J . CNVs: Harbingers of a rare variant revolution in psychiatric genetics. Cell 2012; 148: 1223–1241.
Owen MJ, Craddock N, O'Donovan MC . Suggestion of roles for both common and rare risk variants in genome-wide studies of schizophrenia. Arch Gen Psychiatry 2010; 67: 667–673.
Pulver AE, Nestadt G, Goldberg R, Shprintzen RJ, Lamacz M, Wolyniec PS et al. Psychotic illness in patients diagnosed with velo-cardio-facial syndrome and their relatives. J Nerv Ment Dis 1994; 182: 476–477.
Devriendt K, Fryns JP, Mortier G, Van Thienen MN, Keymolen K . The annual incidence of DiGeorge/velocardiofacial syndrome. J Med Genet 1998; 35: 789–790.
Bassett AS, Chow EWC, Husted J, Weksberg R, Caluseriu O, Webb GD et al. Clinical features of 78 adults with 22q11 deletion syndrome. Am J Med Genet A 2005; 138A: 307–313.
Murphy KC, Jones LA, Owen MJ . High rates of schizophrenia in adults with velo-cardio-facial syndrome. Arch Gen Psychiatry 1999; 56: 940–945.
Shprintzen RJ . Velo-cardio-facial syndrome: a distinctive behavioral phenotype. Ment Retard Dev Disabil Res Rev 2000; 6: 142–147.
Bassett AS, Marshall CR, Lionel AC, Chow EWC, Scherer SW . Copy number variations and risk for schizophrenia in 22q11.2 deletion syndrome. Hum Mol Genet 2008; 17: 4045–4053.
Shprintzen RJ . Velo-cardio-facial syndrome: 30 years of study. Dev Disabil Res Rev 2008; 14: 3–10.
Portnoï MF . Microduplication 22q11.2: a new chromosomal syndrome. Eur J Med Genet 2009; 52: 88–93.
Ou Z, Berg JS, Yonath H, Enciso VB, Miller DT, Picker J et al. Microduplications of 22q11.2 are frequently inherited and are associated with variable phenotypes. Genet Med 2008; 10: 267–277.
Van Campenhout S, Devriendt K, Breckpot J, Frijns JP, Peeters H, Van Buggenhout G et al. Microduplication 22q11. 2: a description of the clinical, developmental and behavioral characteristics during childhood. Genet Couns 2012; 23: 135–148.
Kaminsky EB, Kaul V, Paschall J, Church DM, Bunke B, Kunig D et al. An evidence-based approach to establish the functional and clinical significance of copy number variants in intellectual and developmental disabilities. Genet Med 2011; 13: 777–784.
Yobb TM, Somerville MJ, Willatt L, Firth HV, Harrison K, MacKenzie J et al. Microduplication and Triplication of 22q11.2: a highly variable syndrome. Am J Hum Genet 2005; 76: 865–876.
Hamshere ML, Walters JTR, Smith R, Richards AL, Green E, Grozeva D et al. Genome-wide significant associations in schizophrenia to ITIH3/4, CACNA1C and SDCCAG8, and extensive replication of associations reported by the Schizophrenia PGC. Mol Psychiatry 2012; 18: 708–712.
Wang K, Li M, Hadley D, Liu R, Glessner J, Grant SFA et al. PennCNV: an integrated hidden Markov model designed for high-resolution copy number variation detection in whole-genome SNP genotyping data. Genome Res 2007; 17: 1665–1674.
van Beveren NJM, Krab LC, Swagemakers S, Buitendijk G, Boot E, van der Spek P et al. Functional gene-expression analysis shows involvement of schizophrenia-relevant pathways in patients with 22q11 deletion syndrome. PLoS ONE 2012; 7: e33473.
Rosenfeld JA, Coe BP, Eichler EE, Cuckle H, Shaffer LG . Estimates of penetrance for recurrent pathogenic copy-number variations. Genet Med 2012; 15: 478–481.
Wentzel C, Fernström M, Öhrner Y, Annerén G, Thuresson A-C . Clinical variability of the 22q11.2 duplication syndrome. Eur J Med Genet 2008; 51: 501–510.
Hiroi N, Takahashi T, Hishimoto A, Izumi T, Boku S, Hiramoto T . Copy number variation at 22q11.2: from rare variants to common mechanisms of developmental neuropsychiatric disorders. Mol Psychiatry 2013; 18: 1153–1165.
Rees E, Walters JTR, Georgieva L, Isles AR, Chambert KD, Richards AL et al. Analysis of copy number variations at 15 schizophrenia-associated loci. Br J Psychiatry 2013 (in press).
Karayiorgou M, Simon TJ, Gogos JA . 22q11.2 microdeletions: linking DNA structural variation to brain dysfunction and schizophrenia. Nat Rev Neurosci 2010; 11: 402–416.
Acknowledgements
The 6882 schizophrenia cases from the discovery sample were genotyped at the Broad Institute and funded by a philanthropic gift to the Stanley Center for Psychiatric Research. We thank the participants and clinicians who took part in the Cardiff COGS study. This work was supported by a clinical research fellowship to JW from the MRC/Welsh Assembly Government and the Margaret Temple Award from the British Medical Association. We acknowledge Andrew Iles, David Parslow, Carissa Philipart and Sophie Canton for their work in recruitment, interviewing and rating. For the CLOZUK sample, we thank Novartis for their guidance and co-operation. We also thank staff at The Doctor's Laboratory, in particular Lisa Levett and Andrew Levett, for help and advice regarding sample acquisition. We acknowledge Kiran Mantripragada, Lesley Bates, Catherine Bresner and Lucinda Hopkins for laboratory sample management. The work at Cardiff University was funded by the Medical Research Council (MRC) Centre (G0800509) and Program Grants (G0801418), the European Community’s Seventh Framework Programme (HEALTH-F2–2010–241909 (Project EU-GEI) and a PhD to ER. Funding support for the Swedish study was provided by NIMH R01 MH077139 (P Sullivan), NIMH R01 MH095034 (P Sklar), the Stanley Center for Psychiatric Research, the Karolinska Institutet, Karolinska University Hospital, the Swedish Research Council, an ALF grant from Swedish County Council, the Söderström Königska Foundation and the Netherlands Scientific Organization (NWO 645–000–003). For Irish/WTCCC2 study, funding was provided by the Wellcome Trust Case Control Consortium 2 project (085475/B/08/Z and 085475/Z/08/Z), the Wellcome Trust (072894/Z/03/Z, 090532/Z/09/Z and 075491/Z/04/B), NIMH grants (MH 41953 and MH083094) and Science Foundation Ireland (08/IN.1/B1916). The LCL expression data was generated with grant support from RC2MH090030 and R01MH094091. The authors acknowledge the contribution of data from outside sources: (1) Genetic Architecture of Smoking and Smoking Cessation accessed through dbGAP: Study Accession: phs000404.v1.p1. Funding support for genotyping, which was performed at the Center for Inherited Disease Research (CIDR), was provided by 1 × 01 HG005274–01. CIDR is fully funded through a federal contract from the National Institutes of Health to The Johns Hopkins University, contract number HHSN268200782096C. Assistance with genotype cleaning, as well as with general study coordination, was provided by the Gene Environment Association Studies (GENEVA) Coordinating Center (U01 HG004446). Funding support for collection of data sets and samples was provided by the Collaborative Genetic Study of Nicotine Dependence (COGEND; P01 CA089392) and the University of Wisconsin Transdisciplinary Tobacco Use Research Center (P50 DA019706, P50 CA084724). (2) High Density SNP Association Analysis of Melanoma: Case-Control and Outcomes Investigation, dbGaP Study Accession: phs000187.v1.p1: Research support to collect data and develop an application to support this project was provided by 3P50CA093459, 5P50CA097007, 5R01ES011740 and 5R01CA133996. (3) Genetic Epidemiology of Refractive Error in the KORA (Kooperative Gesundheitsforschung in der Region Augsburg) Study, dbGaP Study Accession: phs000303.v1.p1. Principal Investigators: Dwight Stambolian, University of Pennsylvania, Philadelphia, PA, USA; H Erich Wichmann, Institut für Humangenetik, Helmholtz-Zentrum München, Germany, National Eye Institute, National Institutes of Health, Bethesda, MD, USA. Funded by R01 EY020483, National Institutes of Health, Bethesda, MD, USA. (4) WTCCC2 project samples from National Blood Doners (NBS) Cohort, EGAD00000000024. WTCCC2 project samples from 1958 British Birth Cohort, EGAD00000000022.
Author information
Authors and Affiliations
Consortia
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Molecular Psychiatry website
PowerPoint slides
Rights and permissions
This work is licensed under a Creative Commons Attribution 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by/3.0/
About this article
Cite this article
Rees, E., Kirov, G., Sanders, A. et al. Evidence that duplications of 22q11.2 protect against schizophrenia. Mol Psychiatry 19, 37–40 (2014). https://doi.org/10.1038/mp.2013.156
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/mp.2013.156
Keywords
This article is cited by
-
Striatal dopaminergic alterations in individuals with copy number variants at the 22q11.2 genetic locus and their implications for psychosis risk: a [18F]-DOPA PET study
Molecular Psychiatry (2023)
-
Exploring the association between birthweight and breast cancer using summary statistics from a perspective of genetic correlation, mediation, and causality
Journal of Translational Medicine (2022)
-
Copy number variation at the 22q11.2 locus influences prevalence, severity, and psychiatric impact of sleep disturbance
Journal of Neurodevelopmental Disorders (2022)
-
Longitudinal trajectories of cortical development in 22q11.2 copy number variants and typically developing controls
Molecular Psychiatry (2022)
-
Integration of genetic, transcriptomic, and clinical data provides insight into 16p11.2 and 22q11.2 CNV genes
Genome Medicine (2021)