Abstract
In men, data regarding breast cancer carcinogenesis are limited. The aim of our study was to describe the presence of precursor lesions adjacent to invasive male breast cancer, in order to increase our understanding of carcinogenesis in these patients. Central pathology review was performed for 1328 male breast cancer patients, registered in the retrospective joint analysis of the International Male Breast Cancer Program, which included the presence and type of breast cancer precursor lesions. In a subset, invasive breast cancer was compared with the adjacent precursor lesion by immunohistochemistry (n=83) or targeted next generation sequencing (n=7). Additionally, we correlated the presence of ductal carcinoma in situ with outcome. A substantial proportion (46.2%) of patients with invasive breast cancer also had an adjacent precursor lesion, mainly ductal carcinoma in situ (97.9%). The presence of lobular carcinoma in situ and columnar cell-like lesions were very low (<1%). In the subset of invasive breast cancer cases with adjacent ductal carcinoma in situ (n=83), a complete concordance was observed between the estrogen receptor, progesterone receptor, and HER2 status of both components. Next generation sequencing on a subset of cases with invasive breast cancer and adjacent ductal carcinoma in situ (n=4) showed identical genomic aberrations, including PIK3CA, GATA3, TP53, and MAP2K4 mutations. Next generation sequencing on a subset of cases with invasive breast cancer and an adjacent columnar cell-like lesion showed genomic concordance in two out of three patients. A multivariate Cox model for survival showed a trend that the presence of ductal carcinoma in situ was associated with a better overall survival, in particular in the Luminal B HER2+ subgroup. In conclusion, ductal carcinoma in situ is the most commonly observed precursor lesion in male breast cancer and its presence seems to be associated with a better outcome, in particular in Luminal B HER2+ cases. The rate of lobular carcinoma in situ and columnar cell-like lesions adjacent to male breast cancer is very low, but our findings support the role of columnar cell-like lesions as a precursor of male breast cancer.
Similar content being viewed by others
Main
Male breast cancer is rare, with an estimated incidence of approximately 1.1 per 100 000 a year, representing <1% of all breast cancer cases reported worldwide.1 Male breast cancer seems to resemble hormone receptor-positive postmenopausal female breast cancer, although there is a later age of onset, a more advanced stage at presentation, and consequently an overall worse prognosis.1, 2, 3 Furthermore, there appears to be a markedly lower prevalence of invasive lobular carcinomas in men (1–2%) as compared with women (15%).4
In women, terminal ductal lobular units of the breast are regarded as the origin of invasive breast cancer.5 Ductal carcinoma in situ and lobular carcinoma in situ are seen as precursor lesions of invasive ductal carcinoma and invasive lobular carcinoma, respectively.6 Besides carcinoma in situ, columnar cell lesions are regarded as precursor lesions of (low-grade estrogen receptor positive) female breast cancer.7 Pure ductal carcinoma in situ accounts for up to 15–30% of all breast cancers detected in women nowadays and it is detected adjacent to invasive breast cancer in a substantial proportion of patients.8, 9
In women, coexisting ductal carcinoma in situ has been reported to be associated with lower biological aggressiveness in luminal type breast cancer as compared with pure luminal breast cancer without coexisting ductal carcinoma in situ.10, 11
Obviously, the anatomy of male breasts is different as compared with female breasts since male breasts tissues mainly consist of ducts without the formation of lobules. Based on this difference, one could hypothesize a different pattern of carcinogenesis in men as compared with women. In men, pure ductal carcinoma in situ accounts for about 10% of all breast cancers detected and we could find no published data regarding the frequency of carcinoma in situ adjacent to invasive breast cancer.12, 13 Besides, there are also no published data regarding the biological significance of coexisting ductal carcinoma in situ in male breast cancer, which are estrogen receptor (ER) positive/HER2 negative breast cancers in the vast majority of cases. In literature, there is no consensus regarding the existence of columnar cell-like lesions in males.7, 14 Verschuur-Maes et al.7 found no convincing columnar cell-like lesions at the periphery of 89 male breast cancer cases, but identified Keratine 5 clonally negative ducts, which might indicate that these lesions are breast cancer precursor lesions. In line with this, Ni et al.14 reported the presence of ducts with a columnar cell-like morphology in a small subset of male breast cancer cases. However, both studies were based only on morphology supplemented with immunohistochemistry, lacking additional molecular analyses to evaluate genomic aberrations in these potential male breast cancer precursor lesions.
In this study, we report the presence of various breast cancer precursor lesions in the largest male breast cancer series ever published, supplemented with next generation sequencing on a selected number of cases. Furthermore, we correlated the presence of these lesions with other clinicopathologic features and outcome, in order to increase our understanding of carcinogenesis in this population, which may facilitate future studies regarding prevention and early diagnosis.
Materials and methods
Patients
The International Male Breast Cancer Program is a worldwide collaborative effort, coordinated by the European Organization for Research and Treatment of Cancer (study number 10085), with the help of Translational Breast Cancer Research Consortium in the USA, and run under the Breast International Group and North American Breast Cancer Group networks. It is composed of three parts, where part 1 was a retrospective joint analysis of all male breast cancer cases treated in the participating centers for a period of 20 years (1990–2010). In this part 1, 1822 male breast cancer cases were enrolled in 23 centers from nine countries. A subgroup of this initial population was selected based on eligibility for this male breast cancer program (22 excluded) and availability of a tumor tissue block for central pathology review (446 excluded) for which the precursor lesion status could be assessed (26 excluded). Therefore, the present analysis population consists of 1328 patients. Patient and tumor characteristics studied include age, stage, tumor size, and nodal status.
In this study we adhered to the Declaration of Helsinki and the Code of Conduct of the Federation of Medical Scientific Societies in the Netherlands (http://www.fmwv.nl). Since this was a retrospective study with coded patient identification without risks, no informed consent was needed.
Pathologic Evaluation
One representative formalin-fixed-paraffin-embedded, hematoxylin and eosin-stained tumor tissue block was selected for central pathology review (performed by CvD or PvD). Tumor characteristics were evaluated, including histological type (according to the WHO), grade (according to the modified Bloom and Richardson grading system),15 and presence of a precursor lesion. The precursor lesions were categorized as columnar cell-like lesions (with or without atypia), atypical lobular neoplasia/lobular carcinoma in situ, atypical ductal hyperplasia or ductal carcinoma in situ. In cases where ductal carcinoma in situ was present, nuclear grade was recorded.16
ER, Progesterone receptor (PR), Ki67, and HER2 expression were assessed on a Tissue Micro Array in a different central lab. ER and PR were reported as Allred scores, using a cutoff point of >2 as positive. HER2 status was reported as per the ASCO-CAP guideline.17 Immunohistochemistry-based surrogate intrinsic breast cancer subtypes were defined according to the 2013 St Gallen consensus guidelines (referred to as surrogate breast cancer subtypes).18 A subset of 83 cases with invasive breast cancer and adjacent ductal carcinoma in situ was selected for additional immunostaining with ER, PR, and HER2 on whole sections. These cases were selected based on the presence of sufficient ductal carcinoma in situ for additional immunostaining.
Molecular Analysis: Microdissection, DNA Extraction, and Next Generation Sequencing
We selected four cases of male breast cancer with a sufficient amount of adjacent ductal carcinoma in situ and three cases with invasive breast cancer and an adjacent lesion resembling columnar cell-like lesions. These cases with a columnar cell-like lesion were selected based on the availability of a tissue block. Additional immunohistochemistry was performed on these three cases with a columnar cell-like lesion, using antibodies against Keratine 5 and ER. Microdissection was performed manually with a sterile scalpel under a stereomicroscope (Zeiss, Oberkochen, Germany). Normal tissue, columnar cell-like lesions, ductal carcinoma in situ, and invasive breast cancer cells were dissected from 10 to 15 hematoxylin-stained sections (6 μm) of formalin-fixed-paraffin-embedded tissue blocks. The percentage of the dissected tumor cells of invasive breast cancer and ductal carcinoma in situ was approximately 80–90%. Of all isolated lesions, DNA was extracted using a lysis buffer (Promega Benelux, Leiden, The Netherlands) with proteinase K and 5% Chelex 100 resin.
We started by analyzing DNA extracted from the invasive breast cancer regions. Next generation sequencing was performed on the Ion Torrent Personal Genome Machine with a broad breast cancer-related panel. Genes listed in this panel included 37 breast cancer-related genes and 9 hotspot-regions as described for female breast cancer, that is, PIK3CA, TP53, AKT1, GATA3, and MAP3K119, 20, 21 (details of genes listed in this breast cancer panel are available in Supplementary Table S1). The minimal DNA input was 10 ng per primer pool. In brief, library and template preparations were performed consecutively with the AmpliSeq Library Kit 2.0-384 LV and the Ion PGM Hi-Q Chef Kit. Templates were sequenced using the Ion PGM Hi-Q Chef Kit on an Ion 318v2 chip. Sequence information was analyzed with Variant Caller v4.4.2.1 (Life Technologies, Carlsbad, CA, USA) and variants were annotated in a local Galaxy pipeline using ANNOVAR.22, 23, 24 Variants were called when the position was covered at least 100 times. Nonsynonymous somatic point mutations, insertions, and deletions that change the protein amino acid sequence and splice site alterations were selected. Variants found in at least 25% of the called reads were considered reliable. Non-reproducible sequence artifacts due to cytosine deamination, G>A, or C>T mutations, were excluded when not listed in the COSMIC database (http://cancer.sanger.ac.uk/cosmic). To find genomic resemblances between breast cancer and the adjacent columnar cell-like lesion and/or ductal carcinoma in situ, we started with next generation sequencing analyses of the invasive component. Based on the selected invasive tumor-specific variants, a specific custom-made panel was designed per patient, which was used for targeted analyses in the adjacent columnar cell-like lesion and/or adjacent ductal carcinoma in situ component. Furthermore, the originally reported variants of the invasive component were validated with this custom-made panel.
Statistics
The association between the presence of ductal carcinoma in situ and lobular carcinoma in situ with histological type of the tumor was assessed, as was the association between the presence of ductal carcinoma in situ and M stage, HER2 status, breast cancer subtype, and nodal status (for patients who were free of metastases at diagnosis (M0 patients)). Also, the relationship between grade of the ductal carcinoma in situ component vs grade of the adjacent invasive breast cancer was explored. For all the aforementioned contingency tables, Fisher exact tests for association were performed.
The relationship between the presence of ductal carcinoma in situ and outcome, as measured by relapse-free survival for M0 patients and overall survival, was investigated. Subgroup analyses were added for the three breast cancer subtypes with a prevalence of at least 50 patients: Luminal A, Luminal B (HER2 negative), and Luminal B (HER2 positive). A multivariate model for overall survival was fitted to assess the effect of the presence of ductal carcinoma in situ when adjusting for the baseline factors included in Table 3. Patients with missing information on one of the aforementioned factors, or with a different breast cancer subtype than the ones mentioned above were excluded from the analysis.
Relapse-free survival was defined as the time from diagnosis until one of the following events: local recurrence, distant relapse, or death due to any cause. Overall survival constitutes the time interval from diagnosis until death due to any cause. Patients without an event of interest for the above end points are censored at their last follow-up date. Patients with missing data on (any of) the events of interest for relapse-free survival or overall survival are excluded from the analyses on that end point. Outcome data are analyzed per the Kaplan–Meier method, reported P-values correspond to the logrank test, and the hazard ratio was estimated from the Cox proportional hazards model (95% confidence intervals are per Wald test).
The reported analyses should be considered exploratory. No multiple testing adjustments were implemented.
Results
General Patient and Treatment Characteristics
We collected a total number of 1328 primary male breast cancers. Median age was 67 years. The majority of patients were treated with a mastectomy (60.1%). A small subset of patients underwent either breast-conserving surgery (2.6%) or no surgery (0.6%). The remaining cases (36.6%) missed data regarding breast surgery. About half of the patients with known data regarding adjuvant radiotherapy received radiation (29.9% with radiation vs 29.7% without radiation). The majority of patients (43.2%) did not receive chemotherapy (only 16.6% of the patients did receive chemotherapy and 40.2% of the patients had missing data). In contrast, the majority of patients (43.9%) received endocrine therapy (14.5% did not receive endocrine therapy and remaining data were missing). The majority of Her2-positive patients received Trastuzumab from 2006 onwards (43.3% vs 16.7% who did not receive Trastuzumab, remaining data were missing).
Patients with Precursor Lesions
Out of 1328 cases, 613 (46.2%) had a precursor lesion adjacent to the invasive component. In the remaining 715 cases (53.8%), no precursor lesion was detected within the selected tissue block. The majority of precursor lesions consisted of ductal carcinoma in situ (97.9%), mainly grade 2 (64%). The observed frequency of lobular carcinoma in situ, atypical ductal hyperplasia, and columnar cell-like lesions was very low (<1%). Table 1 provides an overview of subtypes of precursor lesions. A total of 13 patients had a combination of precursor lesions. The majority of these cases (11 out of 13) had a combination of ductal carcinoma in situ with a columnar cell-like lesion, one patient had atypical ductal hyperplasia with a columnar cell-like lesion and one case had a combination of lobular carcinoma in situ with ductal carcinoma in situ grade 1.
Presence of Precursor Lesions According to Other Clinicopathological Features
Table 2 provides an overview of patient and tumor characteristics by the presence of a precursor lesion. The majority of breast cancers were classified as invasive ductal carcinoma (84.6%), mainly grade 2 (49.8%). The prevalence of invasive lobular carcinoma was low (1.4%). Most carcinomas were classified by immunohistochemistry as luminal-like subtype, either luminal A (35.9%) or luminal B (49.3%). There was no significant association between surrogate breast cancer subtype or HER2 status and the presence of a precursor lesion (P=0.14 and 0.31 respectively). More detailed patient and tumor characteristics were presented before.25
Comparison of Ductal Carcinoma In Situ and Lobular Carcinoma In Situ with Adjacent Invasive Breast Cancer
We observed a significant correlation between the presence of ductal carcinoma in situ and the histology of the invasive breast cancer (P=0.02). The prevalence of ductal carcinoma in situ adjacent to invasive ductal carcinoma was the highest (46.6%), as compared with lobular or other subtypes (27.8% and 36.8% respectively). Similarly, there was a significant correlation between the presence of lobular carcinoma in situ and histologic breast cancer subtype (P<0.01). Although the prevalence of lobular carcinoma in situ was low (n=5), it was mainly seen adjacent to invasive lobular carcinoma (3 out of 5). The remaining two cases with lobular carcinoma in situ were associated with a mixed ductal and lobular carcinoma.
In cases with invasive breast cancer and adjacent ductal carcinoma in situ, there was a positive correlation between nuclear grade of ductal carcinoma in situ and nuclear grade of invasive breast cancer, where grade was frequently similar in both components (Trend test for association P<0.01). In line with this, there was a strong correlation of ER, PR, and HER2 status between ductal carcinoma in situ and the adjacent invasive breast cancer where tested. Regarding ER, the majority of cases (82 out of 83 cases) were positive for ER in both the ductal carcinoma in situ and the invasive component. One case was negative in both components. PR status was positive in both components in 81 out of 83 patients. The remaining two cases were negative in both components. Regarding HER2 status, no discrepancies were detected between ductal carcinoma in situ and the adjacent invasive component. The majority of cases (78 out of 83) were not overexpressed in both components; the remaining cases (n=5) were overexpressed in both components.
For four patients, we performed targeted next generation sequencing of invasive ductal carcinoma and adjacent ductal carcinoma in situ. The results of these analyses are presented in Table 3. In three out of four patients, well-known breast cancer mutations noted in the COSMIC database were found in both the ductal carcinoma in situ component and the adjacent invasive component, which supports the hypothesis that ductal carcinoma in situ is indeed a precursor lesion of male breast cancer. In one out of these four cases, no specific somatic mutation was found in either the invasive or the in situ component within this focused panel of genes.
Presence of Columnar Cell-Like Lesions
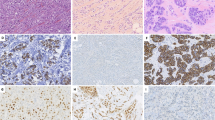
In 13 patients, a lesion resembling columnar cell-like lesions in women was detected adjacent to invasive breast cancer. The majority of these cases (11 out of 13) also had adjacent ductal carcinoma in situ. These columnar cell-like lesions mainly consisted of dilated ducts with apical snouting and cytonuclear atypia. Notably, the cytonuclear aspects resembled the cellular aspects of the adjacent invasive component (Figure 1). However, a convincing morphologic architecture of these lesions, as seen in women, is missing.
Two cases with distended ducts lined by myoepithelial cells and an inner layer of columnar cells with apical snouting, rounded nuclei, and prominent nucleoli (a, c), interpreted as a columnar cell-like lesion and an adjacent invasive component with similar cytonuclear features (b, d, respectively) (original magnification × 40).
Next generation sequencing was performed for three of these cases. The selection was based on availability of tissue. In two out of three cases, we found similar mutations in the columnar cell-like lesion and the adjacent invasive component (Figure 2). In case 1 (Figure 2a–f), the invasive breast cancer was associated with both a ductal carcinoma in situ component and a columnar cell-like lesion. These three components showed similar mutations, including a PIK3CA and a GATA3 mutation. Case 2 (Figure 2g–l) showed a PIK3CA mutation in both the invasive breast cancer and the adjacent columnar cell-like lesion. In the remaining case, we identified a TP53 mutation in the invasive component, which could not be found in the adjacent columnar cell-like lesion. These findings are in line with the overlapping morphology and support the hypothesis that columnar cell-like lesions are a putative precursor lesion of male breast cancer.
Two cases (a–f: case 1, g–l: case 2) with a columnar cell-like lesion and adjacent invasive breast cancer. Hematoxylin and eosin staining of the columnar cell-like lesions adjacent to invasive breast cancer in (a,g) (original magnification × 20). A detailed hematoxylin and eosin staining of columnar cell-like lesion ducts (b, h) and an adjacent invasive component with similar cytonuclear features (c,i) (original magnification × 40). The luminal columnar cells show strong nuclear staining with ER (d,j) while only a few cells are positive for CK5 (e,k). Next generation sequencing showed identical mutations in both the columnar cell-like lesions and the adjacent invasive component (f: GATA3 deletion mutation and a PIK3CA missense mutation, l: PIK3CA missense mutation).
Association Between the Presence of Ductal Carcinoma In Situ and Clinical Outcome
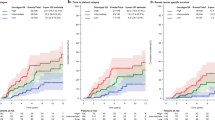
There was no significant association between the presence of ductal carcinoma in situ and metastatic or nodal status (P=0.17 and P=0.41, respectively). Relapse-free survival for M0 patients with ductal carcinoma in situ vs patients without ductal carcinoma in situ was not statistically different (HR=0.84; 95% CI 0.65, 1.08; P=0.18). Subgroup analyses for Luminal A, Luminal B HER2−, and Luminal B HER2+ did also not indicate an effect for ductal carcinoma in situ vs no ductal carcinoma in situ (Luminal A: HR=0.81, 95% CI 0.52–1.24, P=0.33; Luminal B HER2−: HR=0.96, 95% CI 0.67–1.37, P=0.80; Luminal B HER2+: HR=0.43, 95% CI 0.09–2.07,P=0.29). For overall survival, however, there was a difference between patients with ductal carcinoma in situ as compared with patients without ductal carcinoma in situ (HR=0.74, 95% CI 0.63–0.87, P<0.01; Figure 3a). Subgroup analyses showed that this effect is mainly driven by the Luminal A cases (HR=0.64, 95% CI 0.49–0.84, P<0.01; Figure 3b) and Luminal B HER2+ patients (HR=0.34, 95% CI 0.15-0.79, P<0.01; Figure 2d) and was not seen in the Luminal B HER2− cases (HR=0.91, 95% CI 0.72–1.16, P=0.44; Figure 2c). A multivariate Cox model for overall survival was fitted including potential confounding covariates (Table 4). After adjusting for these factors in this model, there was a trend that the presence of ductal carcinoma in situ was associated with a better overall survival, in the Luminal A but in particular in the Luminal B HER2+ subgroup.
Discussion
In our series, a substantial proportion (46%) of patients with invasive breast cancer also had adjacent ductal carcinoma in situ. Although we cannot draw conclusions regarding the exact frequency of ductal carcinoma in situ adjacent to male breast cancer (since we only received one block/patient), we can conclude that ductal carcinoma in situ is present in a large proportion of male breast cancer. There was a strong positive correlation between nuclear grade, ER, PR, and HER2 status of ductal carcinoma in situ and the adjacent invasive breast cancer. In line with this, molecular analysis confirmed similarities on the genomic level, including identical PIK3CA, GATA3, TP53, and MAP2K4 mutations in both components. These data are supportive but not definitive evidence that ductal carcinoma in situ represents a precursor lesion of male breast cancer.
The frequency of lobular carcinoma in situ in our series was very low (<1%), which is in line with the very low incidence of invasive lobular carcinoma previously reported in male breast cancer patients. No classic columnar cell-like lesions were reported in this large series of male breast cancer patients, which is in line with a previous smaller series.7 However, we reported a few cases with columnar cell-like lesions adjacent to invasive breast cancer, including dilated, twisted ducts with apical snouts, and morphological resemblance with the adjacent invasive component. These ducts lacked the classical morphology of female columnar cell-like lesions, including rounded ducts with intraluminal calcifications, which limits the ability to recognize these lesions. Therefore, since distinct morphological criteria to define columnar cell-like lesions in male are lacking, the incidence remains unknown. In our series, we reported several identical genomic alterations, including PIK3CA and GATA3 mutations, in two out of three patients with a columnar cell-like lesion and an adjacent invasive component.
A limitation of this study is that next generation sequencing analysis was performed on only a small subset of cases with a columnar cell-like lesion, due to the low detection rate and the lack of available tissue blocks to perform additional analyses. Regarding ductal carcinoma in situ, there was not such a restriction regarding availability of tissue, but performing next generation sequencing on more samples would not have changed the conclusion that ductal carcinoma in situ is indeed a precursor of male breast cancer. Another limitation is that we only sequenced a panel of selected tumor-specific variants and, therefore, we were not able to evaluate the full spectrum of mutational events. A larger panel could have identified additional genes. However, the goal of this part of the study was to support the morphological finding of resemblance of the columnar cell-like lesions and the adjacent invasive component by providing additional information on the genetic level, rather than providing an overview of all mutations present in these lesions.
In women, ductal carcinoma in situ is more often detected adjacent to ER, PR, and/or HER2 positive invasive breast cancer. In this male breast cancer series, there was no significant association between the presence of ductal carcinoma in situ and surrogate breast cancer subtype. A potential explanation for this difference is the different distribution of breast cancer surrogate subtypes in men as compared with women, including a low frequency of HER2+ and triple negative cases.
In the literature, no data exist regarding the association between the presence of ductal carcinoma in situ and outcome of male breast cancer. In our series, Luminal A and Luminal B HER2+ patients with an adjacent ductal carcinoma in situ component were observed to have a better overall survival compared with those without a ductal carcinoma in situ component, also after adjustment for potential confounders, which suggests that coexisting ductal carcinoma in situ could represent an earlier or biologically less aggressive form of disease. However, the observed associations between the presence of ductal carcinoma in situ with clinical outcome in this study should be interpreted cautiously since the treatments these patients received were not highly standardized and not controlled by protocols. Therefore, the reported analysis is informative and hypothesis generating but cannot be considered a classical prognostic factor analysis.
In conclusion, this is the first and largest study describing the presence and significance of breast cancer precursor lesions in male breast cancer, supplemented with next generation sequencing. Ductal carcinoma in situ seems to be the most common precursor lesion in male breast cancer, as in female patients. The frequency of lobular carcinoma in situ was very low, which is in line with the low frequency of lobular carcinomas in male patients. Based on our data, no definite conclusion can be drawn regarding the prevalence of columnar cell-like lesions in men, but the morphological and genetic overlap between columnar cell-like lesions and adjacent invasive breast cancer suggest a possible causal relationship between these lesions.
References
Anderson WF, Jatoi I, Tse J et al, Male breast cancer: population-based comparison with female breastcancer. J Clin Oncol 2010; 28: 232–239.
Anderson WF, Althuis D . Is male breast cancer similar or different than female breast cancer? Breast Cancer Res Treat 2004; 83: 77–86.
Korde AL, Zujewski JA . Multidisciplinary meeting on male breast cancer: summary and research recommendations. J Clin Oncol 2010; 28: 2114–2122.
Ottini L, Palli D . Male breast cancer. Crit Rev Oncol Hematol 2010; 73: 141–155.
Wellings SR, Jensen HM, Marcum RG . An atlas of subgross pathology of the human breast with special reference to possible precancerous lesions. J Natl Cancer Inst 1975; 55: 231–273.
Burstein HJ, Polyak K, Wong JS et al, Ductal carcinoma in situ of the breast. N Engl J Med 2004; 350: 1430–1441.
Verschuur-Maes AH, Kornegoor R, de Bruin PC et al, Do columnar cell lesions exist in the male breast? Histopathology 2014; 64: 818–825.
Virnig BA, Tuttle TM . Ductal carcinoma in situ of the breast: a systematic review of incidence, treatment, and outcomes. J Natl Cancer Inst 2010; 102: 170–178.
Doebar SC, van den Broek EC, Koppert LB et al, Extent of ductal carcinoma in situ according to breast cancer subtypes: a population based cohort study. Breast cancer Res Treat 2016; 185: 179–187.
Dieterich M, Hartwig F, Stubert J et al, Accompanying ductal carcinoma in situ in breast cancer patients with invasive ductal carcinoma is predictive of improved local recurrence- free survival. Breast 2014; 23: 346–351.
Wong H, Lau S, Leung R et al, Coexisting ductal carcinoma in situ independently predicts lower tumor aggressiveness in node-positive luminal breast cancer. Med Oncol 2012; 29: 1536–1542.
Cutuli B, Dilhuydy JM, De Lafontan B . Ductal carcinoma in situ of the male breast. Analysis of 31 cases. Eur J Cancer 1997; 33: 35–38.
Fentiman IS, Fourquet A . Male breast cancer. Lancet 2006; 367: 595–604.
Ni YB, Mujtaba S, Shao MM et al, Columnar cell-like changes in the male breast. J Clin Pathol 2014; 67: 45–48.
Elston CW, Ellis IO . Pathological prognostic factors in breast cancer. The value of histological grade in breast cancer: experience from a large study with long-term follow-up. Histopathology 1991; 19: 403–410.
The Consensus Conference Committee (1997).. Consensus conference on the classification of ductal carcinoma. Cancer 1997; 80: 1798–1802.
Wolf AC, Hammond MEH, Hicks DG . Recommendations for human epidermal growth factor receptor 2 testing in breast cancer: American Society of Clinical Oncology/College of American Pathologists Clinical Practice Guideline Update. J Clin Oncol 2013; 31: 3997–4013.
Untch M, Gerber B, Harbeck N et al. 13th St. Gallen International Breast Cancer Conference 2013: Primary Therapy of Early Breast cancer Evidence, Controversies, Consensus – Opinion of a German Team of Experts (Zurich 2013). Breast Care (Basel) 2013; 8: 1–29.
Shah SP, Roth A, Goya R et al, The clonal and mutational evolution spectrum of primary triple-negative breast cancers. Nature 2012; 486: 395–399.
Cancer Genome Atlas Network. Comprehensive molecular portraits of human breast tumours. Nature 2012; 490: 61–70.
Banerji S, Cibulskis K, Rangel-Escareno C et al, Sequence analysis of mutations and translocations across breast cancer subtypes. Nature 2012; 486: 405–409.
Giardine B, Riemer C, Hardison RC et al, Galaxy: a platform for interactive large-scale genome analysis. Genome Res 2005; 15: 1451–1455.
Goecks J, Nekrutenko A, Taylor J . Galaxy: a comprehensive approach for supporting accessible, reproducible, and transparent computational research in the life sciences. Genome Biol 2010; 11: R86.
Wang K, Li M, Hakonarson H . ANNOVAR: functional annotation of genetic variants from high-throughput sequencing data. Nucleic Acids Res 2010; 38: e164.
Cardoso F, Bartlett J, Slaets L . Characterization of male breast cancer: first results of the EORTC10085/TBCRC/BIG/NABCG International Male Breast Cancer Program. Cancer Res 2015; 75: S6-05.
Acknowledgements
We are grateful to all patients, investigators, and pathologists who participated in the study, to all national coordinating centers and groups (The European Organisation for Research and Treatment of Cancer-Breast Cancer Group, Borstkanker Onderzoek Groep, Swedish Association of Breast Oncologists, Ireland Cooperative Oncology Research Group, Schweizerisches Arbeitsgemeinschaft Klin. Krebsforschung, Pathologisch Anatomisch Landelijk Geautomatiseerd Archief), their centers and to many independent sites from USA, UK, and Spain. The International Male Breast cancer Program and this work is supported by grants from the Breast cancer Research Foundation, the Dutch Pink Ribbon, the European Breast cancer Council, the Susan G. Komen for the Cure, the Swedish Pink Ribbon, the Swedish Breast cancer Association, and the Erasmus MC Cancer Institute. This Program is also supported by the The European Organisation for Research and Treatment of Cancer Research Fund.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on Modern Pathology website
Supplementary information
Rights and permissions
About this article
Cite this article
Doebar, S., Slaets, L., Cardoso, F. et al. Male breast cancer precursor lesions: analysis of the EORTC 10085/TBCRC/BIG/NABCG International Male Breast Cancer Program. Mod Pathol 30, 509–518 (2017). https://doi.org/10.1038/modpathol.2016.229
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/modpathol.2016.229
This article is cited by
-
Male breast cancer: an update
Virchows Archiv (2022)
-
Male breast cancer: a disease distinct from female breast cancer
Breast Cancer Research and Treatment (2019)
-
Characterization and prognosis of estrogen receptor-positive/progesterone receptor-negative male breast cancer: a population-based study
World Journal of Surgical Oncology (2018)
-
Preferential expression of NY-BR-1 and GATA-3 in male breast cancer
Journal of Cancer Research and Clinical Oncology (2018)
-
Characterisation of male breast cancer: a descriptive biomarker study from a large patient series
Scientific Reports (2017)