Abstract
Massive localized lymphedema is a monstrous tumefactive pseudosarcoma seen in middle-aged morbidly obese adults. Since its initial description in 1998, the etiology remains unknown, although associations with trauma, surgeries, and hypothyroidism have been reported. Herein, we report the largest study of massive localized lymphedema and expand upon its clinicopathologic features. Fifty-four cases from 46 patients were retrospectively identified from the institutional archives of The Ohio State University Wexner Medical Center between 2002 and 2015. Forty-six patients (21 males and 25 females, mean age 50 years) presented with large masses developing over a 5–60-month period. The majority of patients were Caucasian (n=39). All patients were obese with a mean weight of 384.7 lb and a mean body mass index of 59.6 kg/m2. Thirty-six patients had a history of atherosclerotic cardiovascular disease and diabetes mellitus type 2 was present in 22 patients. Eight patients had multifocal massive localized lymphedema. The sites included thigh (n=33), abdomen (n=17), suprapubic region (n=1), mons pubis (n=6), scrotum (n=2), perianal region (n=1), and right flank (n=1). Mostly, the clinical impression was benign processes, including pannus or lymphedema pseudotumor. Grossly, the mean weight was 8237 g and the mean size was 53.2 cm. Histologically, eight cases showed a unique pattern of dystrophic calcifications mimicking hyperchromatic, atypical nuclei that might lead to misdiagnosis of liposarcoma, four cases showed focal metaplastic ossification, and three cases showed multinucleated cells in addition to prototypic features of massive localized lymphedema. We report that this is the largest series of massive localized lymphedema. This is a lesion mostly seen in morbidly obese patients and the thigh is the most common site of involvement. We note a marked racial predilection for Caucasians and a tendency towards multiplicity. We suggest that obesity itself and the related metabolic syndrome have an important role in its pathogenesis.
Similar content being viewed by others
Main
Massive localized lymphedema, an emerging complication of morbid obesity, is a monstrous tumefactive pseudosarcoma seen in middle-aged morbidly obese adults. It was first described in 1998 as a distinct clinical-pathologic entity by Farshid and Weiss1 following an analysis of soft tissue masses identified in 14 morbidly obese patients. In 2009, Folpe et al.2 studied a series of 22 patients, where the authors emphasized the challenge in diagnosing massive localized lymphedema as a mimicker of malignant lesions, including atypical lipomatous tumor/well-differentiated liposarcoma, angiosarcoma, low-grade fibroblastic neoplasm, and myxoid liposarcoma. Massive localized lymphedema displays a predilection for the inner thigh, but other sites have been reported, including the lower abdominal wall, suprapubic region, mons pubis, vulva, inguinoscrotal, penis, popliteal fossa, lower leg, and arm.1, 2, 3, 4, 5, 6, 7, 8, 9
The etiology and pathogenesis of massive localized lymphedema is unclear but might be multifactorial. In the original paper of Farshid and Weiss, lymphatic obstruction due to massive adipose tissue was proposed as the main etiological factor.1 Other associations including trauma, surgery, and hypothyroidism have also been reported.1, 9 As massive localized lymphedema mostly occurs in morbidly obese patients, it is unclear to what extent these other factors are etiological.
Herein, we present 46 cases of massive localized lymphedema. We expand upon the existing clinicopathological features while highlighting an enrichment for multiplicity. We report a peculiar pattern of dystrophic calcifications that can mimic the hyperchromatic nuclei seen in atypical lipomatous tumors/well-differentiated liposarcomas, highlighting an important diagnostic pitfall.
Materials and methods
Electronic medical records uncovered 46 consecutive patients with ‘massive localized lymphedema’ from the institutional archives of The Ohio State University Wexner Medical Center for the period of 2002–2015. Patient demographics, clinical histories, with particular emphasis on any antecedent surgery, trauma, or hormonal disorders, and radiographic studies were reviewed, and patient follow-up information was obtained. All available hematoxylin and eosin-stained slides and immunohistochemical stains were re-reviewed by a soft tissue pathologist (OHI) to verify diagnosis. Histologically, all cases showed prototypic features of massive localized lymphedema, including variable degrees of cutaneous reactive changes, alternating edema and fibrosis of the dermis punctuated by foci of lymphangiectasia, and expansion of the fibrous septae encasing lobules of fat in the subcutis with variably atypical spindle cells.
Results
Clinical Features
The 54 cases occurred in 21 males and 25 females, ranging from 23 to 76 years of age (mean 50 years). All patients were morbidly obese with a mean body weight of 384.7 lb (174.5 kg) (range 209–605 lb, 94.8–274.4 kg) and a mean body mass index of 59.6 kg/m2 (range 36–81.4 kg/m2). The majority of patients were Caucasian (n=39) with a Caucasian to African American ratio of 6.5:1. The masses predominantly involved the thigh (n=33, right: 21, left: 12), but also occurred in the abdomen (n=17), suprapubic region (n=1), mons pubis (n=6), scrotum (n=2), perianal region (n=1), and right flank (n=1). The details are summarized in Table 1. Eight patients (patients #6, 11, 16, 22, 23, 28, 30, and 33) presented with multifocal massive localized lymphedema. The mean body mass index of these eight patients was 67.48 kg/m2, which was higher than the average. The tumors were of long duration, reportedly present from 5 months to 5 years before excision.
One patient (patient #10) had a prior radical mastectomy with subsequent right flank massive localized lymphedema. Another patient (patient #1) had a motor vehicle accident with a subsequent diagnosis of right inner thigh massive localized lymphedema. Moreover, 33 of the remaining 44 patients had a history of at least one abdominal/inguinal surgery or major procedure. We noticed a history of blunt trauma in two patients (#5 and 20) and incisional abdominal trauma in one patient (#23). Thirty-six of 46 patients had a history of atherosclerotic cardiovascular disease, including hypertension, coronary artery disease, and congestive heart failure. Endocrinopathies were present in a significant number of patients (n=27), including diabetes mellitus type 2 (n=16), hypothyroidism (n=5), and both diseases (n=6). Eleven patients had a significant weight loss history with/without gastric bypass surgery.
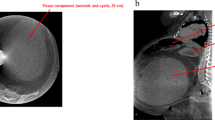
Mostly, the clinical impression was that of pannus or lymphedema pseudotumor. Only two patients (patients #5 and #35) underwent diagnostic studies before excisional surgery because the diagnosis of sarcoma was entertained. One patient (patient #35) had magnetic resonance imaging (Figure 1) showing a mass-like area of soft tissue swelling/edema with multiple internal septations and intervening fat lobules involving the medial aspect of right leg centered at the level of the distal femur, without discrete enhancing masses; these findings were clinically consistent with massive localized lymphedema. The other patient (patient #5) first underwent Fine Needle Aspiration with a diagnosis of ‘suspicious for liposarcoma.’ The subsequent needle core biopsy was consistent with massive localized lymphedema.
Radiologic features of massive localized lymphedema. (a and b) AP radiographs of the right distal femur and medial distal thigh demonstrate a large region of pedunculated, mass-like soft tissue swelling. Multiple septa with areas of fat attenuation are evident. (c–e) MRI of the right femur and medial soft tissues demonstrates a massive lymphedematous medial soft tissue lesion. (c) Coronal T1 showing isointensity to muscle with intervening, expanded, fat-signal lobules. (d) Coronal STIR demonstrates extensive high signal edema throughout the mass. (e) Coronal T1-fat-saturated post-gadolinium. Heterogeneous enhancement after contrast administration can mimic the appearance of well-differentiated liposarcoma.
Pathological Features
The tumors were extremely large at presentation, with a mean weight of 8237 g (range 272–26 940 g) and a mean maximum diameter of 53.2 cm (range 14.2–115.5 cm) (Table 1). Each specimen displayed thickened, indurated skin that was described as nodular, cobblestone or peau d'orange-like in nature (Figure 2a). On a cut surface, the most remarkable finding was the dramatic amount of edema encountered. The dermis was noticeably expanded and often partially fibrotic (Figure 2a). In each specimen, underlying the thickened skin was coarsely lobulated, tan-yellow adipose tissue with interweaving bands of variably fibrotic connective tissue (Figure 2a). Two specimens showed necrosis and 12 specimens showed skin scarring.
Gross and histologic features of massive localized lymphedema. Cross section of surgical specimen shows thickened skin with peau d'orange-like changes, extensive fibrosis and edema (a). Correlating with the gross findings, histologic sections (b–l, Mag × 200) show a thickened epidermis with edema and extensive fibrosis (b). The alternating pattern of edema and fibrosis (c) is easily appreciated in all cases at low power magnification. A common finding is broad fibrous septa that interlaces the mature adipose tissue (d). Ectatic, irregularly shaped lymphatics in the fibrous bands (e) and reactive capillaries aggregated at the junction between adipose and fibrous connective tissue (f) are characteristic vascular proliferations seen in massive localized lymphedema. Calcifications with fat necrosis (g) as well as metaplastic ossifications (h) are observed in several cases that demonstrate the chronicity of the disease. Increased number of ‘atypical’ fibroblasts in the fibrous bands (i), multinucleated cells (j), and microcalcifications mimicking atypical cells are noted in several cases and should not be interpreted as a feature of atypical lipomatous tumor/well-differentiated liposarcoma. In difficult cases, a von Kossa special stain can be a useful diagnostic tool that can highlight the microcalcifications (k).
Histologically, all cases showed prototypic features of massive localized lymphedema as originally described by Farshid and Weiss (Figures 2b–g). Thickened skin with mild hyperkeratosis (Figure 2b) and alternating edema and fibrosis of the dermis (Figure 2c) were identified in each of the cases. Expansion of the fibrous septae encasing lobules of fat in the subcutis (Figure 2d) was also seen in all cases. The dermis and subcutis showed numerous small-to medium-sized, irregularly shaped lymphatic vessels (Figure 2e) and small, thick-walled blood vessels surrounded by scattered to dense lymphocytes, as well as lymphoid follicles. Immunohistochemical staining performed on one specimen revealed that the commonly appreciated perivascular lymphocytes were predominately CD4+ T cells, with occasional CD8+ T cells and B cells. In the case with dense lymphoid follicles, dense populations of both T and B cells were seen. Proliferation of capillaries and small arterioles interface between fat and fibrous septa was observed (Figure 2f). Eight cases showed microscopic calcifications (Figure 2g) and four cases had focal metaplastic ossification (Figure 2h). Variably atypical spindle cells (Figure 2i) were observed in fibrous septa along with multinucleated cells seen in three cases (Figure 2j) akin to those sometimes seen in atypical lipomatous tumor/well-differentiated liposarcoma, serving as a potential diagnostic pitfall. Interestingly, in some cases, microcalcifications could easily mimic hyperchromatic nuclei (Figure 2k). In one case, a von Kossa stain highlighted the calcifications (Figure 2l). However, hyperchromasia, prominent atypia, and features prototypic for atypical lipomatous tumor/well-differentiated liposarcoma, were not appreciated. In all cases, the stroma was extremely hypocellular and edematous. Mature adipocytes within a proliferative vascular network were seen without lipoblasts.
Treatment and Follow-Up
All the patients but one were treated with surgical resection. Only one patient (patient #5) received palliative treatment owing to contraindication of surgery after the core biopsy diagnosis of massive localized lymphedema. One patient (patient #19) died post surgery owing to a complication arising from ventral hernia repair. Follow-up information within 2 months to 9 years of the excision indicated that only two patients (patients #1 and 4) had local recurrence and underwent surgery. Interestingly, these two patients were significantly younger at the time of diagnosis (mean 31 years) and the available body mass index for one of these patients was 79.8 kg/m2. The rest recovered uneventfully without any major complications.
Discussion
Massive localized lymphedema is a relatively recently described pseudosarcomatous tumefactive benign entity that mostly occurs in the obese population.1 Its recognition has become increasingly important because of the phenotypic mimicry with malignant lesions, such as atypical lipomatous tumor/well-differentiated liposarcoma and the variants thereof.1, 2 Though originally thought to be rare, case reports have increasingly been reported.1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21 With the rising incidence of obesity and super morbid obesity throughout the world, particularly in Western countries, the prevalence of massive localized lymphedema is conceivably expected to increase.22, 23 Although the etiology, presentation, and treatment are addressed in the literature, many unanswered questions remain regarding why some morbidly obese patients develop massive localized lymphedema and others seem minimally affected or unaffected.
Primary lymphedema is a disease caused by the congenital anomalous development of the lymphatic system. Secondary lymphedema is more common, and occurs secondary to a variety of factors that cause either lymphatic obstruction or lymphatic interruption, such as infection (filariasis), the most common cause of secondary lymphedema worldwide or more commonly in the developed world-malignancy, radiotherapy, surgery, in particular radical lymph node dissection, trauma, recurrent bacterial infections, chronic venous insufficiency, and thyroid dermopathy.14 In addition to these factors, the link between obesity, metabolic syndrome, and lymphedema has been extensively studied.12 A link between the metabolic syndrome and lymphedema was suggested by a study of rats fed a high fat diet that had significantly smaller diameter mesenteric lymphatic vessels, theoretically reducing their potential load capabilities and impairing the intrinsic contractility required for proper lymph flow.24
Parallel to these animal studies, some clinical studies have also shown an association between obesity and lymphedema. A lymphoscintigraphy study of 15 obese patients with bilateral lower extremity enlargement and without a history primary lymphedema, inguinal lymphadenectomy, radiation, or ulceration of lower extremity showed that only 5 had abnormal results; these patients had a body mass index above 59.25 Patients with a body mass index less than 54 displayed normal lymphatic function. These findings suggest that lymphatic flow becomes impaired as body mass index increases.
Coppack et al.26 compared lymphoscintigraphy results from 22 obese patients (body mass index 49.4±10.4) with bilateral lymphedema, 9 patients with recognized causes of edema (pelvic surgery, deep vein thrombosis, joint replacement), and 18 patients with cyclic edema. The patients with cardiorespiratory disease and lipoedema were excluded. The test was abnormal for all nine patients with recognized causes. However, only 12/22 obese patients had abnormal results and 10/12 were a unilateral abnormality. These studies suggest that although lymphatic obstruction may have a role (at least in a subset of patients), there are other mitigating factors (local or systemic) that are contributory in the etiopathogenesis of massive localized lymphedema.
In our study, all the patients were morbidly obese with a mean body weight of 384.7 lb (174.6 kg) and a mean body mass index of 59.6 kg/m2. The mean body mass index of eight patients with multiple lesions was 67.48 kg/m2, which is higher than the average. Moreover, the available body mass index for one of the patients with recurrences was 79.8 kg/m2. As type 2 diabetes mellitus and atherosclerotic cardiovascular disease appear to be directly promoted by metabolic syndrome,27 we also specifically searched the history of diabetes mellitus and cardiovascular disease. We found that almost 50% (22/46) of our cohort were diagnosed with diabetes mellitus type 2 and 78% (36/46) had a history of atherosclerotic cardiovascular disease. Because hypothyroidism has been associated with massive localized lymphedema as reported by Wu et al.,9 we looked for the history of hypothyroidism in our patients and found that approximately 24% (11/46 patients) had a history of hypothyroidism. These data suggest that obesity and related metabolic syndrome are the main factors in the development of massive localized lymphedema together with other important predisposing factors including previous surgery±lymphadenectomy, trauma, and hypothyroidism (Figure 3).
Noteworthy, in our study, is a Caucasian to African American ratio of 6.5:1. The reasons for this disparity are not clear. Whether the preponderance of Caucasian individuals is due to referral bias of the demographics of the central Ohio region, or whether the massive localized lymphedema has a predilection for Caucasian individuals is unknown and would need to be validated in a large multicenter cohort. Magnetic resonance imaging appearances were consistent with the previous data that demonstrate pendulous masses without the involvement of fascia and muscle, and reflect the histopathologic findings of dermal fibrosis, edema, and fat partitioned by fibrous septae.1, 17
The gross findings were similar to the previous published data.1, 2, 13 Sectioning of the masses demonstrated dramatic amount of edema and abundant mature adipose tissue separated by thick, edematous fibrous septae, a finding reported by others.1, 2, 9 We also noted a unique pattern of dystrophic calcification in seven of our cases that somewhat mimicked hyperchromatic, atypical nuclei that may lead to a misdiagnosis of atypical lipomatous tumor/well-differentiated liposarcoma. In these cases, a von Kossa stain was helpful in highlighting the calcifications. One case was misdiagnosed as suspicious for liposarcoma. Others have noted a similar propensity for misdiagnoses as atypical lipomatous tumor/well-differentiated liposarcoma.1, 2 Helpful diagnostic clues include the history of morbid obesity and prominent skin changes characteristic of massive localized lymphedema, findings not described in atypical lipomatous tumor/well-differentiated liposarcoma. The other differential diagnoses can include desmoid-type fibromatosis, lipomatosis, angiomatosis, lymphangiomatosis, angiosarcoma, angiomyoma, lipoma, and lipomatosis.1, 2, 8 However, careful attention to the clinical history and prototypic histomorphology will facilitate prompt recognition and avoid major mishaps.
The initial step of chronic lymphedema management in obese patients is conservative, including weight reduction, physical therapy, comprehensive garments, intensive bandaging, lymphatic massage service, and good skin hygiene;14 referral to a bariatric weight loss center should also be considered. However, in the treatment of massive localized lymphedema, surgery is the standard treatment in order to restore mobility, prevent recurrent infections, and confirm the diagnosis.13
The clinical course of massive localized lymphedema is mostly benign, marked by recurrence or persistence and occasional mortality from other comorbid risk factors.1, 13 However, there are sentinel reports of transformation of massive localized lymphedema to angiosarcoma, but these are exceptionally rare events.28, 29, 30
In summary, we report that massive localized lymphedema is a benign, reactive, superficial pseudoneoplastic lesion seen mostly in morbidly obese patients and most commonly involving the thigh, similar to others.1, 2 Unique to this study is the tendency for multiplicity, a novel case of involvement of perianal region, and a peculiar pattern of dystrophic calcifications that can lead to a misdiagnosis of atypical lipomatous lesions/well-differentiated liposarcomas. In these cases, a von Kossa special stain can be reassuring. While the etiology of massive localized lymphedema in obese patients seems to be multifactorial, we suggest that obesity itself and the related metabolic syndrome are the main factors in the development of massive localized lymphedema.
References
Farshid G, Weiss SW . Massive localized lymphedema in the morbidly obese: a histologically distinct reactive lesion simulating liposarcoma. Am J Surg Pathol 1998;22:1277–1283.
Manduch M, Oliveira AM, Nascimento AG et al. Massive localised lymphoedema: a clinicopathological study of 22 cases and review of the literature. J Clin Pathol 2009;62:808–811.
Barr J . Massive localized lymphedema of suprapubic origin. Plast Reconstr Surg 2000;106:1663–1664.
Brewer MB, Singh DP . Massive localized lymphedema: review of an emerging problem and report of a complex case in the mons pubis. Ann Plast Surg 2012;68:101–104.
Champaneria MC, Workman A, Kao H et al. Reconstruction of massive localised lymphoedema of the scrotum with a novel fasciocutaneous flap: A rare case presentation and a review of the literature. J Plast Reconstr Aesthet Surg 2013;66:281–286.
Fadare O, Brannan SM, Arin-Silasi D et al. Localized lymphedema of the vulva: a clinicopathologic study of 2 cases and a review of the literature. Int J Gynecol Pathol 2011;30:306–313.
Lee S, Han JS, Ross HM et al. Massive localized lymphedema of the male external genitalia: a clinicopathologic study of 6 cases. Hum Pathol 2013;44:277–281.
McCluggage WG, Nielsen GP, Young RH . Massive vulval edema secondary to obesity and immobilization: a potential mimic of aggressive angiomyxoma. Int J Gynecol Pathol 2008;27:447–452.
Wu D, Gibbs J, Corral D et al. Massive localized lymphedema: additional locations and association with hypothyroidism. Hum Pathol 2000;31:1162–1168.
Berenji M, Kalani A, Kim J et al. Massive localized lymphedema of the thigh in a morbidly obese patient. Eur J Surg Oncol 2010;36:104–106.
Bogusz AM, Hussey SM, Kapur P et al. Massive localized lymphedema with unusual presentations: report of 2 cases and review of the literature. Int J Surg Pathol 2011;19:212–216.
Chakraborty S, Zawieja S, Wang W et al. Lymphatic system: a vital link between metabolic syndrome and inflammation. Ann N Y Acad Sci 2010;1207 (Suppl 1):E94–102.
Chopra K, Tadisina KK, Brewer M et al. Massive localized lymphedema revisited: a quickly rising complication of the obesity epidemic. Ann Plast Surg 2015;74:126–132.
Fife CE, Carter MJ . Lymphedema in the morbidly obese patient: unique challenges in a unique population. Ostomy Wound Manag 2008;54:44–56.
Gilmore T, Fields JM . A patient with massive localized lymphedema. J Emerg Med 2012;43:e481–e482.
Goshtasby P, Dawson J, Agarwal N . Pseudosarcoma: massive localized lymphedema of the morbidly obese. Obes Surg 2006;16:88–93.
Khanna M, Naraghi AM, Salonen D et al. Massive localised lymphoedema: clinical presentation and MR imaging characteristics. Skeletal Radiol 2011;40:647–652.
McCrystal DJ, O’Loughlin BS . Massive localized lymphoedema of the thigh. ANZ J Surg 2007;77:91–92.
Narayanarao T, Suvarchala A, Krishnababu G . Pseudosarcoma - massive localized lymphoedema in morbidly obese - a rare entity: Case report. Int J Surg Case Rep 2012;3:389–391.
Rosenberg AE . Pseudosarcomas of soft tissue. Arch Pathol Lab Med 2008;132:579–586.
Evans RJ, Scilley C . Massive localized lymphedema: A case series and literature review. Can J Plast Surg 2011;19:e30–e31.
Flegal KM, Carroll MD, Kit BK et al. Prevalence of obesity and trends in the distribution of body mass index among US adults, 1999-2010. JAMA 2012;307:491–497.
Flegal KM, Carroll MD, Ogden CL et al. Prevalence and trends in obesity among US adults, 1999-2008. JAMA 2010;303:235–241.
Zawieja SD, Wang W, Wu X et al. Impairments in the intrinsic contractility of mesenteric collecting lymphatics in a rat model of metabolic syndrome. Am J Physiol Heart Circ Physiol 2012;302:H643–H653.
Greene AK, Grant FD, Slavin SA . Lower-extremity lymphedema and elevated body-mass index. N Engl J Med 2012;366:2136–2137.
Vasileiou AM, Bull R, Kitou D et al. Oedema in obesity; role of structural lymphatic abnormalities. Int J Obes (Lond) 2011;35:1247–1250.
Fife CE, Carter M . Lymphoedema in bariatric patients. J Lymphoedema 2009;4:29–37.
Krause KI, Hebert AA, Sanchez RL et al. Anterior abdominal wall angiosarcoma in a morbidly obese woman. J Am Acad Dermatol 1986;15:327–330.
Salas S, Stock N, Stoeckle E et al. Chronic lymphedema due to morbid obesity: an exceptional cause of abdominal wall angiosarcoma. Virchows Arch 2008;453:217–219.
Shon W, Ida CM, Boland-Froemming JM et al. Cutaneous angiosarcoma arising in massive localized lymphedema of the morbidly obese: a report of five cases and review of the literature. J Cutan Pathol 2011;38:560–564.
Acknowledgements
We gratefully acknowledge Dr Gary Barnett of the Department of Pathology and Laboratory Medicine, The Ohio State University Wexner Medical Center for proof-reading the manuscript and providing editorial assistance.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
This paper was presented in part at the 102nd Annual Meeting of United States and Canadian Academy of Pathology (USCAP), Baltimore, Maryland, March 2nd-8th, 2013.
Rights and permissions
About this article
Cite this article
Kurt, H., Arnold, C., Payne, J. et al. Massive localized lymphedema: a clinicopathologic study of 46 patients with an enrichment for multiplicity. Mod Pathol 29, 75–82 (2016). https://doi.org/10.1038/modpathol.2015.135
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/modpathol.2015.135
This article is cited by
-
Scrotal massive localized lymphedema: a case report
European Journal of Plastic Surgery (2021)
-
Angiosarcoma arising in massive localized lymphedema
Skeletal Radiology (2020)
-
Massive Localized Lymphedema in an Unreported Location (Retroperitoneum)
Diagnostic Pathology (2018)