Abstract
To gain the insight into the involvement of signaling mediated by the mammalian target of rapamycin (mTOR) in the phenotype and biological profiles of tumors and tumor-like lesions of the bone and soft tissue, we analyzed the expression and phosphorylation (activation) of mTOR and its correlation with the status of upstream and downstream modulator proteins Akt, p70S6-kinase (S6K), and eukaryotic initiation factor 4E-binding protein 1 (4E-BP1), which we refer to collectively as mTOR cassette proteins. Immunohistochemical analysis of 140 cases showed activation of Akt in 55% (61% in malignant and 27% in benign), and mTOR expression in 61% (66% in malignant and 39% in benign). The preponderance of mTOR activation was found in tumors of peripheral nerve sheath (malignant peripheral nerve sheath tumor and schwannoma), skeletal muscle origin (rhabdomyosarcoma), and in those exhibiting epithelial nature (chordoma and synovial sarcoma). Together with the result of immunoblotting analysis, it was shown that many of those particular tumors with mTOR activation exhibited activation of Akt, S6K, and 4E-BP1, suggesting the constitutive activation of the Akt/mTOR pathway. In addition, although activation of the Akt/mTOR pathway was largely independent of activation of epidermal growth factor receptor (EGFR), mutation of EGFR was frequently accompanied by constitutive activation of Akt–mTOR–S6K/4E-BP1. By clinicopathological analysis, activation of Akt correlates with statistically higher probability of metastasis. We conclude that mTOR-mediated signaling proteins function not only in the proliferation of the tumor cells, but also in the differentiation and/or maintenance of morphological phenotypes in tumors of rhabdomyoblastic and nerve sheath cell origin. Furthermore, mTOR signaling may also modulate morphogenesis of tumors exhibiting epithelial nature. Additionally, activated Akt may have a function in metastasis. Overall, these results suggest that inhibitors of mTOR cassette may be useful as novel components of combined chemotherapy for a defined subset of bone and soft tissue sarcomas.
Similar content being viewed by others
Main
Malignant bone and soft tissue tumors, currently represent approximately 1% of adult malignancies and 15–20% of pediatric malignancies.1, 2, 3 Recently, novel approaches targeting the underlying specific molecular events have been developed for a defined subset of bone and soft tissue tumors.
There has been much interest in epidermal growth factor receptor (EGFR) as a target for cancer therapy, and several successful pharmacological inhibitors have been designed that inhibit the tyrosine kinase activity of EGFR. Two such drugs, Gefitinib and Erlotinib, for non-small cell lung carcinoma, are currently being further extended in trials for refractory solid tumors in children.4 There are a growing number of reports describing protein overexpression, gene amplification, and mutation of EGFR in bone and soft tissue tumors. Immunohistochemical analyses by us and others have shown the expression of EGFR in approximately 60%5, 6, 7 and the EGFR phosphorylation (activation) in 27%.7 Furthermore, point mutations in the tyrosine kinase domain, a critical determinant of Gefitinib sensitivity, were found in 13% of the cases.7 Another unique profile was that Akt has a prominent role in mediating intracellular signaling.7 Thus, EGFR and the downstream phosphatidylinositol 3-kinase (PI3-K)/Akt-mediated cascades are candidates for molecular targeting therapy in this tumor group.
Akt transduces signals to various molecules, including tuberous sclerosis complex proteins 1 and 2 (TSC1/2) (Figure 1).8, 9 Akt inhibits the activity of TSC, and thereby relieves inhibition of mammalian target of rapamycin (mTOR), which functions downstream of TSC.8, 10, 11 However, Akt could also directly phosphorylate mTOR on Ser2448.12 mTOR is a 289 kD serine/threonine protein kinase,13, 14 activation of which positively regulates cell proliferation by promoting the G1/S phase transition of the cell cycle. This occurs through phosphorylation of two substrates, p70S6 kinase (S6K) and eukaryotic initiation factor 4E-binding protein 1 (4E-BP1), that cooperate in translational initiation.8, 14, 15 This paradigm of kinase-driven pathway leading to phosphorylation of mTOR and promotion of cell proliferation constitutes a critical mechanism underlying the pathology of tumor growth. Indeed, aberrant activation of mTOR has been shown in several cancers, including kidney, breast, and the head and neck carcinomas.8, 16, 17 Thus, mTOR is considered an attractive target of anticancer agents. One agent, the bacterial macrolide rapamycin, which inhibits mTOR activity, has emerged as a validated novel anticancer therapeutic.8, 14 In vitro studies have already established the potential of rapamycin in inhibiting cellular transformation and several kinds of tumors exhibiting activation of PI3-K/Akt are hypersensitive to rapamycin in vivo.18, 19 Recent preclinical evidence supports the efficiency of mTOR inhibitors in the treatment of sarcomas: rapamycin treatment resulted in reduced phosphorylation of proteins functioning downstream of mTOR and can greatly reduce the growth of cell lines from rhabdomyosarcoma, osteosarcoma, and Ewing sarcoma.1, 2, 19, 20 Thus, rapamycin could be a powerful therapeutic regimen in the treatment of bone and soft tissue tumors, especially if the mTOR pathway is shown to be a critical effector of growth signaling not only from EGFR, but from other growth factor receptors as well. However, the overexpression or activation of mTOR and its association with biological behavior as well as intracellular signaling cascade related to EGFR in this tumor group have not been described in detail. For these reasons, we conducted comprehensive analyses of mTOR signaling molecules in the EGFR cascade using surgical cases of bone and soft tissue tumors. Specifically, we asked (i) whether mTOR cassette proteins, namely Akt—mTOR–S6K/4E-BP1, are constitutively activated in some portion, or in a particular subtype of bone and soft tissue tumors, (ii) whether there is any correlation between aberrations in EGFR gene and activation of mTOR cassette proteins, and (iii) whether the expression levels and activation state in mTOR cassette predict the biological profiles of tumors and/or prognosis of patients.
Materials and methods
Cases and Classification
For this study, 140 cases of tumor and the tumor-like lesions (114 malignant and 25 benign tumors, 1 tumor-like lesions) were obtained from surgeries performed at the Department of Orthopedic Surgery, University of Yamanashi Hospital between 1985 and 2006 under protocols approved by the Institutional Human Tissue Utilization Committee as the project No. 367 in University of Yamanashi and No.07-26 in Jichi Medical University. The case characteristics are outlined with the results of immunohistochemistry in Table 1.
Pathological diagnosis was made by histopathological features on hematoxylin–eosin stain, and by the results of immunohistochemistry according to the suggestive description in WHO classification.21 At the time of surgery, none of the patients had received preoperative chemotherapy or radiation therapy. Twenty-nine cases were pathologically confirmed to have subsequent lymph node or visceral metastasis. One hundred and twenty-eight of these cases had been examined for EGFR protein expression by immunohistochemistry and gene amplification by fluorescence in situ hybridization analysis, as described earlier.5, 7 Therefore, the 12 newly obtained samples were also evaluated for their EGFR status by exactly identical methods and further used for other additional experiments performed in this study.
Immunohistochemistry
Serial sections were prepared from paraffin-embedded tissue and immunostained after microwave treatment. Primary antibodies obtained from Cell Signaling Technology (Beverly, MA, USA) were used with the following dilutions: phosphorylated form of Akt (p-AktSer473 rabbit monoclonal, clone 193H12) 1:50, mTOR (rabbit monoclonal, clone 7C10) 1:200, phosphorylated forms of mTOR (p-mTORSer2448 rabbit monoclonal, clone 49F9) 1:100, S6K (p-S6KThr389 monoclonal, clone 1A5) 1:150, and of 4E-BP1 (p-4E-BP1, rabbit polyclonal) at 1:150 dilution. The sensitivity and the specificity of these antibodies were earlier validated on a variety of cell lines and the tissue specimens by immunohistochemical and immunoblotting analysis.7, 22, 23 Antibodies were visualized by a ChemMate Envision/peroxidase complex kit (DAKO Japan, Kyoto, Japan). Immunohistochemical expression was evaluated by two of us (YD, SS) and the intensity of reactivity was evaluated as ‘significant’ or ‘not-significant’: ‘significant’ was defined as definite staining with higher intensity than that observed in non-neoplastic cells. Furthermore, ‘immunohistochemical expression level’ was scored by three-tier system: negative (score 0), <10%; low (score 1), 10% or more, but <50%; high (score 2), 50% or more tumor cells with ‘significant’ staining.7, 23, 24 When scores were classified into two groups for statistical analysis, ‘low’ and ‘high’ were combined as ‘positive’.
Fresh Surgical Tissues and Immunoblotting Analysis
Fresh fragments of tumor, tumor-like lesions, and non-neoplastic tissues were obtained from 34 surgical specimens. These consisted of 28 cases of malignant bone and soft tissue tumors with paired adjacent non-neoplastic tissue, as well as lesions alone without normal tissue from six cases of benign tumors. Histopathological profiles of these cases are listed together with the experimental results in Table 2.
Lysates were prepared from these fresh tissues as described,7, 22 and immunoblotting analysis was performed. Equal amounts (25 μg of lysates) of protein were used for blotting with anti-p-Akt (1:200 dilutions), anti-mTOR, anti-p-mTOR (both 1:500), anti-p-S6K, and anti-4E-BP1 (both 1:250 dilutions) antibodies. Blotting with an anti-actin antibody (1:2500, monoclonal, Cytoskeleton, Denver, CO, USA) was also performed as a loading control.
Expression levels were quantified by densitometric analysis with Image Gauge (Fujifilm, Tokyo, Japan). Protein levels were standardized by β-actin, which was assigned an arbitrary level of ‘10’, and expression signal relative to this was indicated as the ‘expression value’ for each protein. This ‘expression value’ was used to calculate the ‘protein index’ according to the following criteria: (i) when the protein was detectable in the paired non-neoplastic tissue, the protein index was obtained by dividing the ‘expression value’ in tumor tissue by that in non-neoplastic tissue; (ii) when protein expression was barely detectable in the paired non-neoplastic tissue, the value in tumor tissue was directly used as the ‘protein index’; and (iii) when normal tissue was not available, as in benign lesions, the expression value in tumor tissue was divided by the average expression value of non-neoplastic tissues in all sarcoma cases. In this study, protein expression was interpreted as ‘upregulated’ (for mTOR expression) or ‘activated’ (for p-Akt, p-mTOR, p-S6K, and p-4E-BP1) when (i) detectable expression was observed only in tumor tissue and the level was higher than the highest value in any of the non-neoplastic tissues or (ii) the ‘protein index’ was higher than 1.5.7, 23, 25
Statistical Analysis
Inter-observer agreement for the evaluation of immunohistochemical results was analyzed by kappa (κ) statistics as described.23, 26 Differences in the rate of positive immunostaining between two groups were analyzed by Fisher's test. Correlations between the immunohistochemical score and clinicopathological factors were evaluated by Mann–Whitney U-test or Kruskal–Wallis tests. Differences in the levels of protein expression/activation were analyzed by unpaired comparison t-test. Patients’ survival curves were drawn according to the Kaplan–Meier method, and differences between the survival rates of two or three groups were analyzed.
Results
Immunohistochemistry
To examine the expression and activation of effector molecules downstream of Akt, we initially performed immunohistochemistry and the results are shown in Figures 2–4 and Table 1. Overall inter-observer agreement was ‘nearly perfect’ (lowest κ=0.896: 95% confidence interval, 0.841–0.952 for p-4E-BP1).
p-Akt
p-Akt staining was observed in both the nuclei and the cytoplasm in 77 cases (55%), including 70 malignant (61%) and 7 benign lesions (27%) (Figures 2 and 3). Difference between these two groups was significant (P<0.0001). Positive staining was frequently found, in particular, in myxofibrosarcoma (9/11), osteosarcoma (11/14), and chordoma (4/5). Furthermore, more than 60% of those positive cases showed high-level expression (score 2). In benign lesions, schwannoma (3/5) and giant cell tumor of bone (2/3) showed a higher frequency of positive staining.
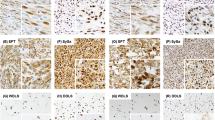
Immunohistochemical staining for proteins functioning up-, and downstream of mTOR. A case of chordoma (case 19) showing positive staining for mTOR, phosphorylated forms of mTOR (p-mTOR), S6K (p-S6K), and 4E-BP1 (p-4E-BP1) with various distribution patterns of positively stained cells. p-Akt staining was observed in the nuclei and the cytoplasm of the tumor cells. Although mTOR and p-4E-BP1 were found in the cytoplasm of tumor cells, p-mTOR and p-S6K were preferentially found in the nucleus. The photo of p-Akt stain shown here was taken in another case, because the result of p-Akt stain in Case 19 was negative.
Immunohistochemical staining in malignant peripheral nerve sheath tumor. Positive p-Akt and p-4E-BP1 stainings were observed in the cytoplasm of tumor cells, mTOR staining in both the nuclei and the cytoplasm, but p-mTOR and p-S6K were predominantly localized in the nucleus. The photo of p-Akt staining was taken in Case 25, and the others were from Case 24, because only the result of p-Akt stain in Case 24 was negative.
mTOR
Positive staining was observed in 75 of 114 (66%) sarcoma cases and in 10 of 26 (39%) benign lesions (P=0.014), with a particularly higher frequency in the following tumors: all the cases of synovial sarcoma (8/8), rhabdomyosarcoma (6/6), Ewing sarcoma (5/5), chordoma (5/5), and malignant peripheral nerve sheath tumor (5/6) (Figures 2 and 3). All of these positive cases gave a positivity score of 2 (high-level expression). mTOR expression was found even in benign lesions, such as schwannoma (5/5, 100%; Figure 4). Positive signal was observed almost exclusively in the cytoplasm, except for a few cases of malignant peripheral nerve sheath tumor.
p-mTOR
p-mTOR staining was observed in 30 of 114 (26%) sarcoma cases and in 6 of 26 (23%) benign lesions (P=0.808). Unlike the staining results for mTOR, p-mTOR staining was often observed in the nuclei of particular types of tumors, such as chordoma (5/5), synovial sarcoma (7/8), rhabdomyosarcoma (5/6), malignant peripheral nerve sheath tumor (4/6), and in schwannoma (4/5) (Figures 2–4). All of these positive cases except for a few cases gave a positive score of 2.
p-S6K
Positive p-S6K staining was found in 86 cases (75%) of sarcoma and 12 cases (46%) of benign lesions (P=0.040), without any obvious differences in expression among histological types. p-S6K staining was observed both in the cytoplasm and the nuclei (Figures 2 and 3). This subcellular localization pattern varies depending on the cases, even within the same histological type of tumor.
p-4E-BP1
Positive p-4E-BP1 staining was observed in 75 (66%) of sarcoma cases and in 13 (50%) of benign lesions (P=0.177), without any obvious differences in expression among histological types. The positive signal was almost exclusively cytoplasmic (Figures 2 and 3).
Immunoblotting Analysis
To evaluate the relative levels of expression or phosphorylation of proteins in tissue samples and to evaluate their correlation with immunohistochemical results, immunoblotting was performed. The results from the 34 cases of fresh tissue specimens are presented in Figure 5 and Table 2.
Protein levels evaluated by immunoblotting analysis in representative cases of tumor and paired normal tissue. The intensity of the bands were quantitated and expressed as ratios relative to β-actin, designated as 10 (‘expression value’). Mutation and amplification of EGFR gene, and immunohistochemical results of EGFR protein, analyzed earlier, were also shown. Asterisks indicate the cases showing metastasis. MFH, malignant fibrous histiocytoma; MyF, Myxofibrosarcoma; SyS, synovial sarcoma; MPNST, malignant peripheral nerve sheath tumor; RMS, rhabdomyosarcoma; Chord, chordoma; Schw, schwannoma; poly, polysomy; low, low-level amplification; IHC, immunohistochemistry; N, normal tissue; T, tumor tissue.
p-Akt
p-Akt was detectable as 60 kD form in all samples obtained from both tumor and normal tissues. Activation of Akt was noted in 17 of 28 cases (61%) of sarcoma and 4 of 6 (67%) benign lesions. Although the total sample number was low, a higher positive frequency was observed in myxofibrosarcoma (3/3), osteosarcoma, synovial sarcoma, and dermatofibrosarcoma protuberans (2/2). Overall, sarcomas and benign lesions showed average p-Akt indices of 2.00 and 1.44, respectively.
mTOR
mTOR expression was observed as the 289 kD form in all cases of tumor and normal tissues. In tumor tissues, mTOR upregulation was observed in 17 of the sarcoma cases (4/5 of malignant fibrous histiocytoma, 2/3 of myxofibrosarcoma, and 2/2 of synovial sarcoma and malignant peripheral nerve sheath tumor, etc) and 3 of the benign lesions. The average mTOR indices in sarcoma and benign lesions were 2.03 and 1.63, respectively.
p-mTOR
mTOR was activated in 9 of 28 sarcoma cases, including both 2 cases of synovial sarcoma, malignant peripheral nerve sheath tumor, one case each of rhabdomyosarcoma, chordoma, etc, and 2 of 6 benign lesions (both schwannoma). The average p-mTOR indices in sarcoma and benign lesions were 1.58 and 0.98, respectively.
In normal tissues, p-mTOR was detectable in 25 of 28 cases, with expression values of up to 0.8, which was lower than the average value in tumor tissue (0.94).
p-S6K
p-S6K was observed as the 70 kD form in 16 of 28 sarcomas and 1 of 6 benign lesions. Higher positive frequency was observed in malignant fibrous histiocytoma (4/5) and osteosarcoma (2/2). The average p-S6K indices in sarcomas and benign lesions were 1.66 and 0.99, respectively. Difference between these two groups was significant (P=0.038).
p-4E-BP1
Phosphorylated forms of 4E-BP1 were observed as the doublet around 17 kD in 16 of 28 sarcomas and 3 of 6 benign lesions: higher positive frequency was observed as 2/2 of osteosarcoma, schwannoma, 4/5 of malignant fibrous histiocytoma, and 2/3 of myxofibrosarcoma. The average p-4E-BP1 indices in malignant and benign lesions were 1.70 and 1.25, respectively.
These overall results correlated well with the results detected by immunohistochemistry.
Pathobiological and Clinicopathological Analyses
Specific correlations in staining patterns of proteins downstream of Akt
We interpreted the results of immunohistochemistry and immunoblotting for sarcomas within the context of the known relationships among effector proteins of EGFR cascade.
Akt and mTOR/p-mTOR.
Expression and activation of mTOR was more frequently observed in the p-Akt positive than in the p-Akt-negative group: mTOR expression was observed in 70% (49/70) of p-Akt-positive vs 59% (26/44) of p-Akt-negative cases, and mTOR activation in 30% (21/70) vs 21% (9/44), respectively (Table 3). However, the differences between these two groups were not statistically significant (P=0.136 and 0.158, respectively). The frequency of Akt activation was not necessarily correlated with those of mTOR expression/activation except for the cases of synovial sarcoma, malignant peripheral nerve sheath tumor, rhabdomyosarcoma, and chordoma (Table 1). Consistently, overlap of positive staining for p-Akt/mTOR or p-Akt/p-mTOR was not frequently observed on a cell-by-cell basis. Thus, direct or constitutive activation of Akt/mTOR may exist in those defined subsets of tumors, but, in general, mTOR activation seems, at least, partially independent of Akt activation.
mTOR and p-mTOR
All p-mTOR-positive cases were also positive for mTOR by both immunohistochemistry and immunoblotting (Tables 2 and 3). In addition, p-mTOR positivity was localized within a larger mTOR-positive area on histology. Therefore, mTOR overexpression seems to be a prerequisite condition for its activation.
p-mTOR and p-S6K/p-4E-BP1
Positive staining for p-S6K and p-4E-BP1 were found at higher frequencies than for p-mTOR, and no correlation between p-mTOR and p-S6K/p-4E-BP1 staining was observed: although 70% (21/30) and 60% (18/30) of the cases showing mTOR activation also stained positive for p-S6K and p-4E-BP1, respectively, 77% (65/84) and 68% (57/84) of the cases that were negative for mTOR activation also showed positive staining for p-S6K and p-4E-BP1 (Table 3). As shown in the representative cases that were positive for p-S6K and p-4E-BP1, p-S6K and p-4E-BP1 staining was present over a broader area than that of p-mTOR. Thus, although direct or constitutive activation of mTOR cassette may exist in particular histological types, such as synovial sarcoma, malignant peripheral nerve sheath tumor, rhabdomyosarcoma, and chordoma, S6K/4E-BP1 activation seems to be largely independent of mTOR activation. Phosphorylation of S6K and 4E-BP1 seem to positively correlate: 86 and 75 cases showed activation of S6K and 4E-BP1, respectively (Table 1), and 64 cases showed both. However, the p-S6K signal was observed in the nuclei as well as cytoplasm, whereas the p-4E-BP1 signal was almost exclusively cytoplasmic.
Correlation among EGFR aberration, protein expression, and phosphorylation of downstream molecules
EGFR amplification and high-level polysomy of chromosome 7 was found in five cases, including one case of high-level amplification (Case 1, malignant fibrous histiocytoma), one case of polysomy (Case 2, malignant fibrous histiocytoma), and three cases of low-level amplification (Case 3, malignant fibrous histiocytoma; Case 6, myxofibrosarcoma; Case 24, malignant peripheral nerve sheath tumor, Table 2).5, 7 Among these five cases, Akt was activated in one of the cases showing low-level amplification (Case 6, myxofibrosarcoma). For mTOR expression levels, four cases exhibited upregulation, including two cases of malignant fibrous histiocytoma (with high-level and low-level amplification) and one case each of myxofibrosarcoma and malignant peripheral nerve sheath tumor (both low-level amplification). With respect to mTOR activation, two cases with low-level amplification (malignant fibrous histiocytoma, malignant peripheral nerve sheath tumor) showed activation, and both of these also showed activation of S6K and 4E-BP1. Overall, the presence of EGFR amplification was frequently, but not always, associated with upregulation of mTOR, and activation of S6K/4E-BP1 (Table 2).
Point mutations were found in four cases, of which three resulted in amino acid substitution (Case 3, malignant fibrous histiocytoma; Case 7, myxofibrosarcoma; Case 19, synovial sarcoma). One of these three cases also exhibited low-level amplification (Case 3).5, 7 All three tumors exhibited markedly increased levels of p-mTOR and two cases exhibited activation of Akt, p-S6K, and/or p-4E-BP1 (Table 2). Therefore, mutation of EGFR may be associated with the constitutive activation of the mTOR cassette.
Of the remaining 21 sarcoma cases not exhibiting EGFR aberrations, no specific correlation with activation of upstream or downstream molecules was found.
Clinicopathological Analysis
Next, overall results were analyzed statistically with respect to their clinicopathological features. First, with regards to metastasis, we examined the correlation between Akt activation determined by immunohistochemistry and metastasis. We observed no statistically significant differences (P=0.383) in metastasis between the cases classified as positive for p-Akt (20/70 cases with score of 1 or 2) and those of negative (9/44 cases with score 0). However, when we compared high-expression cases (17/40 cases with score 2) vs negative cases, we did observe a significantly higher frequency of metastasis in the high-expression group (P=0.0354). This correlation was also observed by immunoblotting analysis. Although 6 of the 9 cases showing metastasis showed activation of Akt, only 11 among the latter 19 cases had activated Akt. Furthermore, in the metastasis-positive group, the average p-Akt index was 2.49, whereas that in the metastasis-negative group was 1.76 (Table 2), and the difference in Akt phosphorylation levels between these two groups was statistically significant in spite of the limited sample size (P=0.031). Thus, activation of Akt may have a role during the process of metastasis. The mutation or expression levels of EGFR, and the upregulation/phosphorylation state of other proteins did not show any correlation with metastasis or cumulative survival rates.
Discussion
Overexpression of EGFR in several types of sarcomas has been reported by us and others,5, 6, 7 and a number of studies have reported that one of mTOR, S6K, and 4E-BP1 were activated in 50–72% of rhabdomyosarcoma.27, 28 Furthermore, it was reported that cases of chordoma and synovial sarcoma displayed strong expression of EGFR,29, 30 and that these cases could be successfully treated by therapies targeting EGFR signaling.30 These observations imply that aberrant EGFR/mTOR signaling may also be involved in the pathology of other defined subsets of bone and soft tissue tumors. However, there are few studies examining the components of this signaling pathway through entire cascade. This study was intended to fill this gap, and our results may offer a number of insights into these pathologies.
First, mTOR expression was detected in wide variety of tumors (61%), and mTOR was activated to higher levels in tumors of specific lineages, namely, those of peripheral nerve sheath and skeletal muscle lineages (malignant peripheral nerve sheath tumor, schwannoma, rhabdomyosarcoma) as well as those showing an epithelial nature (chordoma, synovial sarcoma). These results may shed light on the idea that mTOR activation is involved not only in cell proliferation and growth,13, 14 but also in the differentiation or morphogenesis of particular types of tumors. Consistently, in the past literature, mTOR activation has been described to be prerequisite for myogenic differentiation and growth.31
Second, we observed that mutations in the EGFR were accompanied by constitutive activation of the mTOR cassette proteins, namely mTOR-S6K/4E-BP1. However, these exceptional cases harboring EGFR mutation aside, the data here suggest that in the majority of the cases, mTOR activation may be largely independent of EGFR signaling. Instead, other growth factor receptors have been implicated in pathology of bone and soft tissue tumors, including insulin-like growth factor receptor and fibroblast growth factor receptor.1 Regardless of the particular growth factor receptor pathway involved, mTOR inhibitors may be effective in the cases exhibiting mTOR activation as far as mTOR cassette is activated even in the defined subsets. Indeed, it has been reported from Phase II studies that a rapamycin analogue shows some beneficial response in 28–30% of advanced sarcomas.1, 14, 32
Third, this study showed that mTOR cassette proteins are not activated simultaneously in most tissue sections. Earlier studies also have yielded mixed results: significant association between activation of Akt and mTOR was shown in non-small cell lung carcinoma,33 but not in glioblastoma.9 In the present series, mTOR activation was observed in particular types of tumors in both p-Akt-positive and -negative groups, without any statistically significant differences. This lack of clear correlation in activation among mTOR cassette proteins presumably arises from the convergence of multiple signaling pathways involving mTOR.9 For example, mTOR can be phosphorylated by other protein kinases, in addition to Akt,12, 34 and the mTOR substrates, S6K and 4E-BP1, can be phosphorylated by Erk1/2.35 Moreover, signaling pathways are often not unidirectional and may be subject to feedback signaling. Examples include mTOR activation by p-S6K,34 and Akt inhibition by mTOR.10 It is also important to note that activation of these signaling molecules may be transient and simultaneous activation of linked kinases may be difficult to detect in many tissue specimens.11
Fourth, with regards to subcellular localization, mTOR was observed almost exclusively in the cytoplasm, albeit p-mTOR was frequently in the nucleus. Technically, although anti-mTOR antibody used in this study is supposed to react unphosphorylated and phosphorylated forms of mTOR, nuclear staining was not observed by mTOR staining. The precise reason for this discrepancy was unclear, but small amount of p-mTOR may be detected with higher sensitivity by anti-p-mTOR antibody. Regardless of these speculations, mTOR has been recognized to shuttle between the nucleus and the cytoplasm, and this translocation is required for activation of its target proteins, S6K and 4E-BP1.15, 36 Similar shuttling has also been observed for S6K/4E-BP1,15, 37, 38 and we observe here both nuclear and cytoplasmic localization of p-S6K, whereas p-4E-BP1 was predominantly cytoplasmic. Thus, mTOR and S6K shuttle between the cytoplasm and nucleus, and the latter are activated by p-mTOR in the nucleus and re-localize to the cytoplasm.10 During this process, activation and cytoplasmic re-localization of Akt, mTOR, S6K1, and 4E-BP1 corresponds with movement of the actin arc.39 This coordinated signaling of mTOR leading to the cytoskeleton re-organization by mTOR may have an important function in morphogenesis, and the determination of the epithelial nature of synovial sarcoma and chordoma.
Fifth, mTOR/S6K normally integrates a plethora of signals in response to diverse stimuli that promote an increase in cell size, cytoplasmic volume, and growth of muscle cells,40 as well as growth and differentiation of chondrocyte and adipocyte.41, 42, 43 However, in this study, we did not observe frequent activation of mTOR in tumors of smooth muscle cell, adipocyte, or chondrocyte origin in contrast to rhabdomyosarcoma. Probably, the functional involvement of mTOR is different in tumor vs normal tissue, or the involvement of mTOR in those tumors may be transient in the specific stage during oncogenesis.
Finally, from the clinicopathological viewpoint, there have been some reports of association between phosphorylation of mTOR cassette proteins and tumor development, progression, or prognosis in malignancies of breast, prostate, ovary, and rhabdomyosarcoma.1, 27, 44 However, our analysis did not show clear association between activation of these proteins and clinicopathological factors or prognosis. The only positive result observed was an association between activation of Akt and metastasis. A recent study has shown that Akt/mTOR-related metastasis is linked to an ezrin-mediated pathway that can be inhibited by rapamycin in a murine model of osteosarcoma.45 As Akt was found to function as a predominant effector molecule downstream of growth factor receptors in bone and soft tissue tumors, Akt/mTOR-mediated cascades may be the direct and critical target of drug therapy in this tumor group, aiming at prevention of metastasis.7, 45
Recently, a more viable strategy for using molecularly targeted therapies would be directed toward combined therapy targeting two or more molecules.46 Our current study supports the idea that chemotherapy of bone and soft tissue tumors may be enhanced by using appropriate mTOR cassette inhibitors. However, suppression of the mTOR signaling cassette per se is not necessarily beneficial because of the presence of redundant negative feedback cascades. Indeed, prolonged rapamycin treatment abrogates the negative-feedback pathway from mTOR to Akt and promotes tumor progression.10 On the basis of this interrelationship between the Akt/mTOR pathway and growth factor receptor signaling, the combined use of mTOR inhibitors and inhibitors of PI3-K-Akt signaling or upstream growth factor receptors, may have a synergistic effect to potentiate chemotherapy in the treatment of sarcoma.
In conclusion, this study provides a detailed description of the expression and activation patterns of mTOR cassette proteins. Specifically, we found that (i) Akt was frequently activated in bone and soft tissue tumors and may have a role in metastasis, (ii) mTOR was expressed and activated in tumors of skeletal muscle and nerve sheath origin as well as tumors having an epithelial nature, (iii) tumors harboring EGFR mutation may be accompanied by constitutive activation of mTOR cassette. We believe that this study provides support for future clinical studies using inhibitors targeting the activated kinases of mTOR cassette, in combination with inhibitors of growth factor receptor signaling, including EGFR, to treat bone and soft tissue tumors.
Conflict of interest
The authors declare no conflict of interest.
References
Wan X, Helman LJ . The biology behind mTOR inhibition in sarcoma. Oncologist 2007;12:1007–1018.
Mita MM, Tolcher AW . The role of mTOR inhibitors for treatment of sarcomas. Curr Oncol Rep 2007;9:316–322.
de Alava E . Molecular pathology in sarcomas. Clin Transl Oncol 2007;9:130–144.
Daw NC, Furman WL, Stewart CF, et al. Phase I and pharmacokinetic study of gefitinib in children with refractory solid tumors: a Children's Oncology Group Study. J Clin Oncol 2005;23:6172–6180.
Dobashi Y, Takei N, Suzuki S, et al. Aberration of epidermal growth factor receptor expression in bone and soft-tissue tumors: protein overexpression, gene amplification and activation of downstream molecules. Mod Pathol 2004;17:1497–1505.
Sato O, Wada T, Kawai A, et al. Expression of epidermal growth factor receptor, ERBB2 and KIT in adult soft tissue sarcomas: a clinicopathologic study of 281 cases. Cancer 2005;103:1881–1890.
Dobashi Y, Suzuki S, Sugawara H, et al. Involvement of epidermal growth factor receptor and downstream molecules in bone and soft tissue tumors. Hum Pathol 2007;38:914–925.
Mamane Y, Petroulakis E, LeBacquer O, et al. mTOR, translation initiation and cancer. Oncogene 2006;25:6416–6422.
Riemenschneider MJ, Betensky RA, Pasedag SM, et al. AKT activation in human glioblastomas enhances proliferation via TSC2 and S6 kinase signaling. Cancer Res 2006;66:5618–5623.
O'Reilly KE, Rojo F, She QB, et al. mTOR inhibition induces upstream receptor tyrosine kinase signaling and activates Akt. Cancer Res 2006;66:1500–1508.
Lee CH, Inoki K, Karbowniczek M, et al. Constitutive mTOR activation in TSC mutants sensitizes cells to energy starvation and genomic damage via p53. EMBO J 2007;26:4812–4823.
Bjornsti MA, Houghton PJ . The TOR pathway: a target for cancer therapy. Nat Rev Cancer 2004;4:335–348.
Wullschleger S, Loewith R, Hall MN . TOR signaling in growth and metabolism. Cell 2006;124:471–484.
Petroulakis E, Mamane Y, Le Bacquer O, et al. mTOR signaling: implications for cancer and anticancer therapy. Br J Cancer 2006;94:195–199.
Fingar DC, Richardson CJ, Tee AR, et al. mTOR controls cell cycle progression through its cell growth effectors S6K1 and 4E-BP1/eukaryotic translation initiation factor 4E. Mol Cell Biol 2004;24:200–216.
Rowinsky EK . Targeting the molecular target of rapamycin (mTOR). Curr Opin Oncol 2004;16:564–575.
Molinolo AA, Hewitt SM, Amornphimoltham P, et al. Dissecting the Akt/mammalian target of rapamycin signaling network: emerging results from the head and neck cancer tissue array initiative. Clin Cancer Res 2007;13:4964–4973.
Cao C, Subhawong T, Albert JM, et al. Inhibition of mammalian target of rapamycin or apoptotic pathway induces autophagy and radiosensitizes PTEN null prostate cancer cells. Cancer Res 2006;66:10040–10047.
Petricoin 3rd EF, Espina V, Araujo RP, et al. Phosphoprotein pathway mapping: Akt/mammalian target of rapamycin activation is negatively associated with childhood rhabdomyosarcoma survival. Cancer Res 2007;67:3431–3440.
Wan X, Shen N, Mendoza A, et al. CCI-779 inhibits rhabdomyosarcoma xenograft growth by an antiangiogenic mechanism linked to the targeting of mTOR/Hif-1alpha/VEGF signaling. Neoplasia 2006;8:394–401.
Fletcher CDM, Unni KK, Mertens F . World Health Organization Classification of Tumours. Pathology and Genetic of Tumours of Soft Tissue and Bone. IARC Press: Lyon, 2002.
Suzuki S, Igarashi S, Hanawa M, et al. Diversity of epidermal growth factor receptor-mediated activation of downstream molecules in human lung carcinomas. Mod Pathol 2006;19:986–998.
Dobashi Y, Suzuki S, Matsubara H, et al. Critical and diverse involvement of Akt/mammalian target of rapamycin signaling in human lung carcinomas. Cancer 2009;115:107–118.
Wu F, Liu SY, Tao YM, et al. Decreased expression of methyl methansulfonate and ultraviolet-sensitive gene clone 81 (Mus81) is correlated with a poor prognosis in patients with hepatocellular carcinoma. Cancer 2008;112:2002–2010.
Hoshino R, Chatani Y, Yamori T, et al. Constitutive activation of the 41-/43-kDa mitogen-activated protein kinase signaling pathway in human tumors. Oncogene 1999;18:813–822.
Landis JR, Koch GG . The measurement of observer agreement for categorical data. Biometrics 1977;33:159–174.
Armengol G, Rojo F, Castellvi J, et al. 4E-binding protein 1: a key molecular ‘funnel factor’ in human cancer with clinical implications. Cancer Res 2007;67:7551–7555.
Cen L, Arnoczky KJ, Hsieh FC, et al. Phosphorylation profiles of protein kinases in alveolar and embryonal rhabdomyosarcoma. Mod Pathol 2007;20:936–946.
Nielsen TO, Hsu FD, O'Connell JX, et al. Tissue microarray validation of epidermal growth factor receptor and SALL2 in synovial sarcoma with comparison to tumors of similar histology. Am J Pathol 2003;163:1449–1456.
Hof H, Welzel T, Debus J . Effectiveness of cetuximab/gefitinib in the therapy of a sacral chordoma. Onkologie 2006;29:572–574.
Ohanna M, Sobering AK, Lapointe T, et al. Atrophy of S6K1(−/−) skeletal muscle cells reveals distinct mTOR effectors for cell cycle and size control. Nat Cell Biol 2005;7:286–294.
Okuno S . Mammalian target of rapamycin inhibitors in sarcomas. Curr Opin Oncol 2006;18:360–362.
Balsara BR, Pei J, Mitsuuchi Y, et al. Frequent activation of AKT in non-small cell lung carcinomas and preneoplastic bronchial lesions. Carcinogenesis 2004;25:2053–2059.
Chiang GG, Abraham RT . Phosphorylation of mammalian target of rapamycin (mTOR) at Ser-2448 is mediated by p70S6 kinase. J Biol Chem 2005;280:25485–25490.
Castellvi J, Garcia A, Rojo F, et al. Phosphorylated 4E binding protein 1: a hallmark of cell signaling that correlates with survival in ovarian cancer. Cancer 2006;107:1801–1811.
Kim JE, Chen J . Cytoplasmic-nuclear shuttling of FKBP12-rapamycin-associated protein is involved in rapamycin-sensitive signaling and translation initiation. Proc Natl Acad Sci USA 2000;97:14340–14345.
Rong L, Livingstone M, Sukarieh R, et al. Control of eIF4E cellular localization by eIF4E-binding proteins, 4E-BPs. RNA 2008;14:1318–1327.
Wang ML, Panasyuk G, Gwalter J, et al. Regulation of ribosomal protein S6 kinases by ubiquitination. Biochem Biophys Res Commun 2008;369:382–387.
Liu L, Chen L, Chung J, et al. Rapamycin inhibits F-actin reorganization and phosphorylation of focal adhesion proteins. Oncogene 2008;27:4998–5010.
Fingar DC, Salama S, Tsou C, et al. Mammalian cell size is controlled by mTOR and its downstream targets S6K1 and 4EBP1/eIF4E. Genes Dev 2002;16:1472–1487.
Phornphutkul C, Wu KY, Auyeung V, et al. mTOR signaling contributes to chondrocyte differentiation. Dev Dyn 2008;237:702–712.
Yeh WC, Bierer BE, McKnight SL . Rapamycin inhibits clonal expansion and adipogenic differentiation of 3T3-L1 cells. Proc Natl Acad Sci USA 1995;92:11086–11090.
Kim JE, Chen J . regulation of peroxisome proliferator-activated receptor-gamma activity by mammalian target of rapamycin and amino acids in adipogenesis. Diabetes 2004;53:2748–2756.
Zhou X, Tan M, Stone Hawthorne V, et al. Activation of the Akt/mammalian target of rapamycin/4E-BP1 pathway by ErbB2 overexpression predicts tumor progression in breast cancers. Clin Cancer Res 2004;10:6779–6788.
Wan X, Mendoza A, Khanna C, et al. Rapamycin inhibits ezrin-mediated metastatic behavior in a murine model of osteosarcoma. Cancer Res 2005;65:2406–2411.
Merimsky O, Gorzalczany Y, Sagi-Eisenberg R . Molecular impacts of rapamycin-based drug combinations: combining rapamycin with gemcitabine or imatinib mesylate (Gleevec) in a human leiomyosarcoma model. Int J Oncol 2007;31:225–232.
Acknowledgements
We wish to thank the staff in the Department of Pathology, Saitama Medical Center, for their helpful support. This work is supported by The Japanese Ministry of Education, Sports, Science and Culture No C 20590351 (YD, TY), Young Scientists B 19790256 (SS), B 20791026 (TY), C 19590342 (AO) and the Smoking Research Foundation (YD).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dobashi, Y., Suzuki, S., Sato, E. et al. EGFR-dependent and independent activation of Akt/mTOR cascade in bone and soft tissue tumors. Mod Pathol 22, 1328–1340 (2009). https://doi.org/10.1038/modpathol.2009.104
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/modpathol.2009.104
Keywords
This article is cited by
-
Using biology to guide the treatment of sarcomas and aggressive connective-tissue tumours
Nature Reviews Clinical Oncology (2018)
-
MicroRNA-27a-5p regulation by promoter methylation and MYC signaling in prostate carcinogenesis
Cell Death & Disease (2018)
-
Brain-derived neurotrophic factor promotes VEGF-C-dependent lymphangiogenesis by suppressing miR-624-3p in human chondrosarcoma cells
Cell Death & Disease (2017)
-
The T-box transcription factor 3 is a promising biomarker and a key regulator of the oncogenic phenotype of a diverse range of sarcoma subtypes
Oncogenesis (2016)
-
Activation of the Akt-mTOR and MAPK pathways in dedifferentiated liposarcomas
Tumor Biology (2016)