Abstract
Fatty acid synthase (FASN) expression has been reported in many different tumors, including breast cancer. In gene microarray studies, the fatty acid synthase gene co-clustered with cytokeratins 5 and 17 and other genes that defined the basal-like subset of breast cancers. To define the use of this marker in breast pathology, a rabbit polyclonal antiserum (S143) to a peptide fragment of this gene was produced and compared with a commercially available monoclonal antibody by immunohistochemistry on various tissue microarrays and whole tissue sections. The tissue microarrays included 1090 breast cancers and 244 normal breast tissues. Whole tissue sections consisted of benign and malignant tissues from breast resection specimens. In contrast to other ‘basal’ markers identified by gene expression profiling data, the fatty acid synthase (FASN) expression pattern in normal breast was notable for its expression in only a small subset of basal and suprabasal cells. Dual staining experiments revealed that the subpopulation of cells labeling with FASN did not coexpress myoepithelial markers CK5/6 or p63, but did coexpress e-cadherin. In addition to staining a subset of basal and suprabasal cells, the antiserum highlighted apocrine differentiation, and stained 106/144 (74%) cases of columnar cell lesions and five of five cases of flat epithelial atypia. Despite its association with basal keratins in gene array studies, FASN expression did not correlate significantly with the outcome in breast cancer. We describe an expression pattern that highlights only a subset of basal and suprabasal cells in normal breast ducts and we show by dual expression studies that this subset of cells is different from myoepithelial and basal cytokeratin-positive cells. In addition, FASN expression is described in apocrine metaplasia, columnar cell lesions, and flat epithelial atypia.
Similar content being viewed by others
Main
Through gene and tissue microarray analysis, a consistent ‘basal-like’ subtype has been identified in breast carcinoma, which is associated with a poor clinical outcome.1, 2, 3, 4, 5, 6, 7 In gene microarray studies, the fatty acid synthase (FASN) gene co-clustered with genes that defined the ‘basal-like’ subtype, including cytokeratins 5 and 17.1
Fatty acid synthase (FASN) is a heterodimeric enzyme that catalyzes the synthesis of long-chain fatty acids. The gene encoding fatty acid synthase resides on the long arm of chromosome 17 (17q25). In some tumors, including breast cancer, the gene is upregulated, possibly to accommodate the increase in membrane synthesis as a result of cell proliferation.8, 9 In breast cancer, some investigators have reported an association between FASN expression and tumor recurrence.10, 11 There may also be a regulatory effect of FASN on the expression of the nearby (17q21) Her2neu gene12 and a recent study raises the possibility of cancer chemoprevention by inhibition of FASN.13
We have generated a novel rabbit polyclonal antiserum to the FASN gene and describe the immunohistochemical expression in normal, precancerous and cancerous breast. In addition, we compare this antiserum to a commercially available FASN antibody.
Materials and methods
All tissue studies were performed on de-identified specimens with approval of the institutional review boards of all participating institutions (Stanford University Hospital, Vancouver General Hospital and Beth Israel Deaconess Medical Center).
Tissue Microarrays
Tissue microarrays were constructed in the usual fashion14 on a Beecher tissue microarrayer (Beecher Instruments, Silver Spring, MD, USA), using tissue cores varying between 0.6 and 2.0 mm. Tissue microarrays using pathologic material from Stanford University Hospital, Vancouver General Hospital and Beth Israel Deaconess Medical Center, and containing various normal and tumorous tissues were utilized (Table 1). The tissue microarrays contained a variety of control tissues (liver, kidney, placenta, and so on) as reference points and positive controls.
Whole Tissue Sections
It had been noted in initial staining experiments with S143 that the antibody highlighted apocrine metaplasia. For this reason, breast specimens with prominent apocrine metaplasia and cases of columnar cell lesions (because of the frequent appearance of so-called ‘apocrine snouts’ in at least a subset of columnar cell lesions) were selected for the study. Subsequently, cases of flat epithelial atypia were included in the study because of its putative relation to columnar cell lesions.15 All cases were selected through a diagnosis search of the electronic database files of the Stanford University Medical Center Laboratory of Surgical Pathology and Vancouver General Hospital. Diagnoses were verified on reviewing the hematoxylin and eosin-stained sections by two pathologists (KCJ and DFS). Sections were stained with the S143 antiserum and/or FASN antibody as detailed below.
Commercial Antibodies and Generation of a Novel Rabbit Polyclonal Antiserum
Commercial antibodies against p63, e-cadherin, cytokeratin 5/6, calponin, and FASN were used according to the manufacturer's protocols (Table 2). A rabbit polyclonal antiserum was raised using a synthetic peptide derived from the gene sequence of the fatty acid synthase gene (Applied Genomics, Incorporated, Huntsville, AL, USA), as described earlier.16 Briefly, the peptide was synthesized by standard Fluorenylmethoxycarbonyl (Fmoc) chemistry. The peptide was then conjugated to keyhole limpet hemocyanin and injected into two out-bred rabbits. After the rabbits showed a significant anti-peptide titer, serum (S143) was harvested and passed over an affinity column conjugated with the peptide. Bound antibodies were eluted with a pH gradient.
The current study was not a comparison study between the commercial FASN antibody and the S143 antiserum. Representative benign and tumorous tissues were stained by both the methods and qualitatively determined to show similar staining expression patterns, but a head-to-head comparison of the two antibodies was not quantitatively performed.
Immunohistochemical Staining With One or More Antibodies
Sections to be stained were cut at 4-μm thickness, mounted on positively charged glass slides, and baked at 60°C for a minimum of 60 min. Sections were then immersed in xylene, followed by gradated alcohols, at 10 min per solution. Antigen retrieval involved heating slides in a citrate (pH 6.0) buffer in either a microwave or a pressure cooker. Staining was achieved using biotin-free techniques including EnVision+ Kit (Dako, Carpenteria, CA, USA) automated and manual methods for FASN, S143, and p63 and Benchmark automated immunostainer (Ventana Medical Systems, Tucson, AZ, USA) for e-cadherin and cytokeratin 5/6 (Table 2). Dual chromogenic antibody staining was accomplished using Dako's Double-Stain Kit (Dako, Carpenteria, CA, USA) according to manufacturer's instructions. Single stain slides served as positive controls; negative controls were obtained by omitting the primary antibody application step.
All staining with the S143 antiserum and FASN antibody was cytoplasmic and/or membranous. CK5/6 and e-cadherin decorated cells in a predominantly membranous staining pattern; only nuclear reactivity was considered positive labeling for p63. All slides were reviewed by at least two pathologists (Table 2).
Double Immunofluorescence Staining
Sections to be stained were cut at 4-μm thickness, mounted on positively charged glass slides, and baked at 60°C for a minimum of 60 min. They were then immersed in xylene, followed by gradated alcohols, at 10 min per solution. Antigen retrieval involved microwaving slides in an EDTA/TRIS (pH 9.0) buffer. Staining was achieved as described earlier.17 Briefly, sections were incubated with combined primary antibodies, washed, incubated in the dark with combined secondary antibodies bound to fluorochromes (Invitrogen Corporation, Carlsbad, CA, USA), washed and coverslipped with ProLong Gold antifade reagent with DAPI (Invitrogen Corporation, Carlsbad, CA, USA). Fluorescent slides were imaged by using a Nikon E1000 microscope with UV-2E DAPI and Texas red HYQ filter cubes (Nikon) fitted with a CoolSNAP K4 (Photometrics, Tucson, AZ, USA) camera. The same slides were digitized with a commercial image analysis system ARIOL (Applied Imaging Inc., San Jose, CA, USA). The ARIOL scanner is based on an Olympus microscope with a motorized stage and autofocus capabilities, equipped with a black and white video camera. Slides were scanned at × 40 objective magnification with appropriate filters; ARIOL software converted these three-channel images into color images.
Antibody and Fluorescence Scoring
Antibody staining was semi-quantitatively scored using stain intensity as negative (less than 5% of cells staining, regardless of stain intensity), weakly positive (weak stain intensity in greater than or equal to 5% of cells) or strongly positive (strong stain intensity in greater than or equal to 5% of cells). Staining was characterized as ‘diffuse’ if greater than 50% of cells were stained. Scoring of antibody expression by fluorescence was determined to be positive (greater than 5% of cells staining) or negative, with the characterization of diffuse staining reserved for cases with greater than 50% of cell staining.
Results
Normal Breast
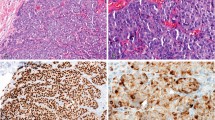
In normal breast tissue, S143 expression was seen in only a subset of basal or suprabasal cells throughout the terminal duct-lobular unit (Figure 1a). Commercially available FASN antibody showed a similar staining pattern and distribution. Significant staining was not seen in larger lactiferous ducts with either reagent.
(a) S143 expression in normal breast. Note that S143 highlights only a subpopulation of basal/suprabasal cells. (b–d) Expression of cytokeratin 5/6, p63 and calponin in normal breast. In contrast to S143, the staining pattern is usually continuous membrane staining in the basal layer for cytokeratin 5/6 and calponin. Relatively continuous nuclear expression along the basal layer is seen with p63.
This staining pattern is in sharp contrast with that seen when normal breast is stained with antibodies to myoepithelial/basal cytokeratin markers, including CK5/6, p63 and calponin, where the staining is typically diffuse and continuous along the basal epithelial/myoepithelial layer (Figure 1b–d). In tissue exhibiting apocrine metaplasia, S143 and/or FASN showed strong diffuse expression (Figure 2a). In double-staining experiments, cells expressing S143 did not coexpress CK5/6 or p63, but did coexpress e-cadherin (Figure 2b–d).
(a) Diffuse S143 expression in an area of apocrine metaplasia. (b) Double-stain immunohistochemistry with CK5/6 (diaminobenzidine, brown) and S143 (fast-red, pink-red) fails to highlight coexpression. Note that S143 expression on high power localizes predominantly to the cytoplasm (see cells at 12 and 6 o'clock), whereas CK5/6 expression localizes predominantly to the membrane (cells along the left side of the image). (c) Double-stain immunohistochemistry with S143 (fast-red, pink-red) and p63 (diaminobenzidine, brown) likewise fails to highlight coexpression. (d) Double immunofluorescence with S143 (FITC, green) and e-cadherin (Cy5, red) shows coexpression of the two antibodies.
Breast Cancer
The antiserum S143 diffusely stained 383 of 743 (52%) scorable tissue cores of invasive breast carcinomas from two large, previously described tissue microarray series.4, 18 Staining was scored on a three-tier scale as negative, weakly positive or strongly positive, with 216 (31% of all scorable invasive breast cancers) exhibiting weak reactivity and 103 (15% of all scorable invasive breast cancers) exhibiting strong reactivity. Staining was diffuse in the overwhelming majority of cases (253/319, or 79%), a pattern markedly different from the subset staining (scattered basal or suprabasal cells only) of FASN commonly observed in normal breast. In contrast, CK5/6 staining, when present (seen in 14% of invasive breast cancers in a previously reported tissue microarray study; see reference 4), often showed a focal staining pattern in carcinomas (67/120, or 56%), although diffuse staining was also observed. Reactivity with S143 did not show any significant correlation with survival in this cohort with a mean follow-up of 17 years (data not shown). Two of two invasive carcinomas with apocrine features showed strong diffuse S143 reactivity (Figure 3, Table 3).
Columnar Cell Lesions
Columnar cell lesions showed FASN expression in 106 of 144 cases (Figure 4a–d, Table 3), using a tissue microarray consisting of 192 scorable tissue cores of normal breast tissue and columnar cell lesions as well as additional full sections.18 This staining distribution was illustrated further by using double stain immunofluorescence (Figure 4c and d).
(a and b) Typical FASN expression in columnar cell lesions (flat epithelial atypia in a). Note the apical snouts and secretions, seen in many columnar cell lesions, in a. (c and d) FASN expression in columnar cell lesions captured through ARIOL system (DAPI (blue) stains nuclei; FITC (green), S143; Cy5 (red), p63).
In lesions characterized as flat epithelial atypia (a category that encompasses columnar cell lesions with atypia), five of five cases showed FASN expression. In all columnar cell lesions and lesions of flat epithelial atypia, FASN expression was relatively diffuse, although focally, rare cells showed more intense staining than the remainder of the lesions.
Discussion
Gene microarray studies and subsequent confirmatory tissue microarray studies have clearly established the existence of a distinct subgroup of breast cancers defined predominantly by overexpression of a number of basal cytokeratin genes and/or proteins, now relatively well-recognized as the ‘basal-like’ subgroup and known to be associated with a poor clinical outcome.1, 2, 3, 4, 5, 6, 7 In the first gene microarray study to describe this subgroup, the fatty acid synthase (FASN) gene co-clustered with basal cytokeratin genes such as cytokeratins 5,6 14 and 17. Rabbit antisera were raised to six of nine genes in this cluster, including the fatty acid synthase gene (S143); expression patterns of the S143 antiserum and a commercial antibody to FASN are described in this study.
In normal breast, FASN expression is seen in a unique pattern of scattered basal and suprabasal cells. It is yet unclear whether this population of cells represent a distinct functional or biologic subset of epithelial cells. Double-staining experiments indicate that FASN-positive cells do not exhibit a characteristic ‘basal’ (cytokeratin 5/6-positive) or myoepithelial (p63-positive) immunophenotype. Esslimani-Sahla et al19 have also described FASN immunoreactivity through a monoclonal FASN antibody as a scattered expression pattern in normal and pathological structures of epithelial mammary cells. Their findings are comparable to ours, though this group did not describe the suprabasal cell staining seen in the current study. The authors19 suggest that these findings are most likely in keeping with epithelial stem cells because of the topographic location. These results provide endorsement for the proposal by some that FASN expression may reflect increased membrane synthesis and cell growth, as supported by studies of the gastrointestinal tract, brain and genitourinary system.8 We hypothesize that this subset of cells may label a stem cell or progenitor cell (or a proportion of these cells) in normal breast.
The existence of a breast stem cell seems quite likely, but its identification and isolation has proved difficult. Many markers have been postulated to identify breast stem/progenitor cells in both the luminal and myoepithelial compartments.20, 21, 22, 23, 24, 25, 26 Our experience with one of these putative stem cell markers, CK5/6,21 is that its expression in normal breast is typically seen in a continuous portion of the basal epithelial compartment, suggesting a myoepithelial expression phenotype. Other authors have provided some evidence that CK5/6 reactivity may not, indeed, represent a stem cell phenotype.22 The unique subset basal and suprabasal expression pattern we observed with the S143 antiserum (as well as the distinction from CK5/6-positive cells, as shown in our double-staining experiments) would seem a possible expression pattern for a stem/progenitor cell. Certainly, many additional studies are necessary to substantiate this hypothesis.
Benign breast ducts can show a large number of histologic appearances, many of which have been described in detail.27 Apocrine metaplasia represents a physiologic response/change and is not, in and of itself, associated with an increased risk of breast cancer. However, the ability to highlight apocrine differentiation may be of some potential interest in the area of apocrine carcinoma. In particular, reactivity in breast carcinomas may confirm the morphologic impression of apocrine differentiation and may lessen the possibility of overgrading the tumor. Although stage is more significant in determining treatment options, increased nuclear and tumor grade may impact therapy decisions as well, so the importance of avoiding overgrading tumors is clinically relevant. FASN and S143 staining of potential apocrine carcinomas, as was seen in this study, might provide information essential to accurate tumor grading.
Columnar cell lesions and flat epithelial changes of the breast have been increasingly recognized, particularly with the large number of breast biopsies performed for mammographically detected lesions.15, 28, 29 Recent reports have demonstrated that columnar cell lesions of the breast, in particular, lesions that contain characteristic apical snouts and luminal secretions and those with atypia, represent premalignant changes.30 We observed FASN staining in the majority of columnar cell lesions, both with and without atypia, although FASN staining did not distinguish non-atypical from atypical lesions. We could also demonstrate that this cell population coexpressed e-cadherin, which is again in keeping with previous findings showing up to 81% positivity of columnar cell lesions for e-cadherin antibodies.30
Although the FASN gene co-clustered with basal cytokeratins in gene microarray studies, our observation in normal breast and columnar cell lesions is that, immunohistochemically, FASN and basal cytokeratins are not coexpressed. This finding is inconsistent with the results of others that columnar cell lesions and flat epithelial atypia do not typically express basal cytokeratins.31 Also interesting is the observation that FASN stains both apocrine metaplasia and columnar cell lesions, suggesting some similar functional or biologic properties between these lesions. Other biomarkers, including estrogen receptor and bcl2, show different expression patterns in apocrine metaplasia and columnar cell lesions.32 Additional investigation of FASN expression and function may illuminate these preliminary findings.
In vitro studies have demonstrated a close relationship between Her2neu and FASN, showing suppressed Her2neu expression with FASN inhibition.12 This work raises the possibility of pharmacologically targeted inhibition of FASN as a potential breast cancer therapy. If and when such studies in humans become feasible, correlation with FASN protein expression in tumors will need to be closely examined, and may have some significance to therapy response.
Although the data we have demonstrated on fatty acid synthase is intriguing and raises the possibility that this antibody may label columnar cell lesions and flat epithelial atypia, it might also serve with lesser specificity as a label for a stem/progenitor cell population. Many more studies will be needed, including isolation and cell culture experiments, to investigate these possibilities.
References
Perou CM, Sørlie T, Eisen MB, et al. Molecular portraits of human breast tumours. Nature 2000;406:747–752.
Abd El-Rehim DM, Ball G, Pinder SE, et al. High-throughput protein expression analysis using tissue microarray technology of a large well-characterised series identifies biologically distinct classes of breast cancer confirming recent cDNA expression analyses. Int J Cancer 2005;116:340–350.
Abd El-Rehim DM, Pinder SE, Paish CE, et al. Expression of luminal and basal cytokeratins in human breast carcinoma. J Pathol 2004;203:661–671.
Nielsen TO, Hsu FD, Jensen K, et al. Immunohistochemical and clinical characterization of the basal-like subtype of invasive breast carcinoma. Clin Cancer Res 2004;10:5367–5374.
Sørlie T, Perou CM, Tibshirani R, et al. Gene expression patterns of breast carcinomas distinguish tumor subclasses with clinical implications. Proc Natl Acad Sci USA 2001;98:10869–10874.
Sørlie T, Tibshirani R, Parker J, et al. Repeated observation of breast tumor subtypes in independent gene expression data sets. Proc Natl Acad Sci USA 2003;100:8418–8423.
van de Rijn M, Perou CM, Tibshirani R, et al. Expression of cytokeratins 17 and 5 identifies a group of breast carcinomas with poor clinical outcome. Am J Pathol 2002;161:1991–1996.
Kusakabe T, Maeda M, Hoshi N, et al. Fatty acid synthase is expressed mainly in adult hormone-sensitive cells or cells with high lipid metabolism and in proliferating fetal cells. J Histochem Cytochem 2000;48:613–622.
Rossi S, Graner E, Febbo P, et al. Fatty acid synthase expression defines distinct molecular signatures in prostate cancer. Mol Cancer Res 2003;1:707–715.
Alo PL, Visca P, Trombetta G, et al. Fatty acid synthase (FAS) predictive strength in poorly differentiated early breast carcinomas. Tumori 1999;85:35–40.
Kuhajda FP, Piantadosi S, Pasternack GR . Haptoglobin-related protein (Hpr) epitopes in breast cancer as a predictor of recurrence of the disease. N Engl J Med 1989;321:636–641.
Menendez JA, Vellon L, Mehmi I, et al. Inhibition of fatty acid synthase (FAS) suppresses HER2/neu (erbB-2) oncogene overexpression in cancer cells. Proc Natl Acad Sci USA 2004;101:10715–10720.
Alli PM, Pinn ML, Jaffee EM, et al. Fatty acid synthase inhibitors are chemopreventive for mammary cancer in neu-N transgenic mice. Oncogene 2005;24:39–46.
Kononen J, Bubendorf L, Kallioniemi A, et al. Tissue microarrays for high-throughput molecular profiling of tumor specimens. Nat Med 1998;4:844–847.
Tavassoli FA, Hoefler H, Rosai J, et al. Intraductal proliferative lesions. In: Tavassoli FA, Devilee P (eds). Tumours of the breast and female genital organs. IARC Press: Lyon, France, 2003, pp 63–73.
West RB, Corless CL, Chen X, et al. The novel marker, DOG1, is expressed ubiquitously in gastrointestinal stromal tumors irrespective of KIT or PDGFRA mutation status. Am J Pathol 2004;165:107–113.
Mason DY, Micklem K, Jones M . Double immunofluorescence labelling of routinely processed paraffin sections. J Pathol 2000;191:452–461.
Porter D, Lahti-Domenici J, Keshaviah A, et al. Molecular markers in ductal carcinoma in situ of the breast. Mol Cancer Res 2003;1:362–375.
Esslimani-Sahla M, Thezenas S, Simony-Lafontaine J, et al. Increased expression of fatty acid synthase and progesterone receptor in early steps of human mammary carcinogenesis. Int J Cancer 2007;120:224–229.
Al-Hajj M, Wicha MS, Benito-Hernandez A, et al. Prospective identification of tumorigenic breast cancer cells. Proc Natl Acad Sci USA 2003;100:3983–3988.
Bocker W, Moll R, Poremba C, et al. Common adult stem cells in the human breast give rise to glandular and myoepithelial cell lineages: a new cell biological concept. Lab Invest 2002;82:737–746.
Clarke CL, Sandle J, Parry SC, et al. Cytokeratin 5/6 in normal human breast: lack of evidence for a stem cell phenotype. J Pathol 2004;204:147–152.
Gudjonsson T, Villadsen R, Nielsen HL, et al. Isolation, immortalization, and characterization of a human breast epithelial cell line with stem cell properties. Genes Dev 2002;16:693–706.
Jones C, Nonni AV, Fulford L, et al. CGH analysis of ductal carcinoma of the breast with basaloid/myoepithelial cell differentiation. Br J Cancer 2001;85:422–427.
Lakhani SR, Chaggar R, Davies S, et al. Genetic alterations in ‘normal’ luminal and myoepithelial cells of the breast. J Pathol 1999;189:496–503.
Pechoux C, Gudjonsson T, Ronnov-Jessen L, et al. Human mammary luminal epithelial cells contain progenitors to myoepithelial cells. Dev Biol 1999;206:88–99.
Rosen PP . Rosen's breast pathology, 2nd edn. Lippincott Williams and Wilkins: Philadelphia, 2001.
Fraser JL, Raza S, Chorny K, et al. Columnar alteration with prominent apical snouts and secretions: a spectrum of changes frequently present in breast biopsies performed for microcalcifications. Am J Surg Pathol 1998;22:1521–1527.
Schnitt SJ, Vincent-Salomon A . Columnar cell lesions of the breast. Adv Anat Pathol 2003;10:113–124.
Dessauvagie BF, Zhao W, Heel-Miller KA, et al. Characterization of columnar cell lesions of the breast: immunophenotypic analysis of columnar alteration of lobules with prominent apical snouts and secretions. Hum Pathol 2007;38:284–292.
Turashvili G, Hayes M, Gilks B, et al. Are columnar cell lesions the earliest histologically detectable non-obligate precursor of breast cancer? Virchows Arch print copy in press 2008 Available from: URL: www.springerlink.com/content/k03kh8172647tn20/.
Luna-Moré S, Weil B, Bautista D, et al. Bcl-2 protein in normal, hyperplastic and neoplastic breast tissues. A metabolite of the putative stem-cell subpopulation of the mammary gland. Histol Histopathol 2004;19:457–463.
Acknowledgements
We thank Torsten O Nielsen of the Genetic Pathology Evaluation Centre in Vancouver, British Columbia, for helpful advice in tissue microarray scoring.
Author information
Authors and Affiliations
Corresponding author
Additional information
Data from this paper was presented as a poster at the 28th Annual San Antonio Breast Cancer Symposium, December 8–11, 2005, San Antonio, Texas and as a platform at the 97th Annual Meeting of the United States and Canadian Academy of Pathology, March 1–7, 2008.
Disclosure/conflict of interest
The authors have none to report.
Rights and permissions
About this article
Cite this article
Jensen, K., Schaeffer, D., Cheang, M. et al. Characterization of a novel anti-fatty acid synthase (FASN) antiserum in breast tissue. Mod Pathol 21, 1413–1420 (2008). https://doi.org/10.1038/modpathol.2008.163
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/modpathol.2008.163