Abstract
Only a small number of malignant mesotheliomas with heterologous elements have been described. There are currently no criteria for diagnosis and little data regarding prognosis. We suggest that the term heterologous mesothelioma should be reserved for tumours that show malignant heterologous elements, notably osteosarcomatous, chondrosarcomatous, or rhabdomyoblastic elements but have immunohistochemical and clinical characteristics of mesothelioma. We identified 27 such cases and characterized the clinical and pathological characteristics of these tumours. In our series, 89% originated in the pleura, and 11% from the peritoneal cavity. The median age at diagnosis was 68 years, ranging from 27 to 85 years. Of these cases, 93% occurred in males and 7% in women. Of the 27 mesothelioma cases 16 (59%) were sarcomatoid, 10 (37%) were biphasic, and one was reported as epithelioid; 40% (11 cases) showed osteosarcomatous elements only, 19% showed areas of rhabdomyosarcoma only, 19% contained areas of chondrosarcoma only, and 22% exhibited osteochondromatous elements. Immunohistochemical labelling for cytokeratins was present in the majority of cases. Exposure to asbestos was identified in all the 17 cases for which an exposure history was available (63%). Median survival was 6 months after diagnosis, similar to the survival seen in sarcomatoid mesotheliomas. The differential diagnosis includes primary and secondary pleural sarcomas, including osteosarcomas and chondrosarcomas. Immunohistochemical labelling for cytokeratins is helpful in the distinction, but lack of labelling for cytokeratins in a spindle cell/sarcomatoid tumour does not exclude the diagnosis of mesothelioma, irrespective of the presence of heterologous elements. We suggest that if the anatomical distribution conforms to that of mesothelioma, a diagnosis of heterologous mesothelioma should be made in preference to a diagnosis of primary pleural osteosarcoma or chondrosarcoma, regardless of cytokeratin positivity, as for conventional non-heterologous sarcomatoid mesothelioma.
Similar content being viewed by others
Main
According to the 2004 WHO classification, malignant mesotheliomas can be broadly classified as epithelial, biphasic, and sarcomatoid.1 These groups show significant differences in histological and clinical features. In addition, there are several further subtypes, including desmoplastic2 and lymphohistiocytoid mesothelioma,3 with some of these subtypes carrying a particularly adverse prognosis; for example, sarcomatoid mesotheliomas are typically more rapidly lethal than epithelial mesotheliomas,4 and desmoplastic mesotheliomas, as subtype of sarcomatoid mesothelioma, is characterized by even shorter survival compared with epithelial and sarcomatoid mesothelioma without desmoplastic features.5 For most of these subgroups, criteria for diagnosis are well defined; for example, to qualify for a diagnosis of biphasic mesothelioma, the International Mesothelioma Panel specifies that each component must occupy at least 10% of the tumour in an adequate tissue sample.6 To qualify for a diagnosis of desmoplastic sarcomatoid mesothelioma, the paucicellular collagen-rich tissue must occupy at least 50% of a tissue specimen, and other diagnostic features include invasion, foci of overtly malignant (sarcomatoid) tissue, and areas of ‘bland’ necrosis. In contrast, the criteria for diagnosis of heterologous mesothelioma are less well defined.
Only a handful of cases of mesothelioma containing heterologous elements have been described.7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20 Amongst the heterologous elements that have been recorded, osseous and chondroid elements are most commonly seen, with liposarcomatous differentiation representing an extremely rare finding.20, 21 No case showing leiomyosarcomatous elements in isolation has been described in the literature, and smooth muscle elements in this location are thought to arise from submesothelial cells rather than the mesothelium itself.22 Endothelial differentiation is regarded as inconsistent with a diagnosis of mesothelioma.23 It is worth noting that because currently no clear definition of the term ‘mesothelioma with heterologous elements’ or ‘heterologous mesothelioma’ exists in the literature, some of the reported cases or case series do not distinguish between benign mesenchymal metaplasia and ‘true’ histologically malignant heterologous elements.17 In addition, there are no criteria as to the types of ‘heterologous’ elements that should be included in this diagnostic subgroup, for example, whether smooth muscle differentiation and liposarcomatous elements are acceptable. Mesotheliomas with ‘rhabdoid’ features have been reported separately in a single large series,12 with only two additional cases that have been reported.10, 13 The true incidence of mesothelioma with heterologous differentiation is therefore difficult to ascertain. Also, some cases are reported in radiology or surgery literature,7, 18, 24, 25, 26 sometimes without histopathologic correlation.
Little data has been collected with regards to epidemiologic features and with relationship to previous asbestos exposure. To the best of our knowledge, only two cases of peritoneal mesothelioma with malignant heterologous elements have been reported to date.9
Finally, there are currently conflicting data on survival for these tumours. Some authors describe unusually long survival of up to 3 years for what is most often a subtype of sarcomatoid mesothelioma—highly unusual when one keeps in mind that the mean survival for patients with mesothelioma overall is in the order of 9 months.4
This paper represents the largest series of such tumours of which we are aware, consisting of 27 cases of mesothelioma with heterologous elements, including three arising within the peritoneal cavity. The aims of the series were to propose a definition that provides clear criteria for the recognition of heterologous mesothelioma, based on clinical, radiological, histologic, and immunohistochemical characteristics; to discuss the differential diagnosis; and to ascertain whether the prognosis for heterologous mesothelioma differs significantly from conventional mesothelioma.
Materials and methods
Selection of Cases
Cases were obtained from the departmental archives, consultation cases, and some medicolegal referral files for two of authors spanning the period of 1982–2008 (VLR) and 1998–2008 (DWH) and with two earlier cases from the files of one of the authors (DWH). We did not specify any particular proportion of the heterologous elements for inclusion into this series (specifically, we did not require the heterologous elements to account for 10% or more of the tumour), but to be classified as a heterologous mesothelioma we required the heterologous elements to show sarcomatoid features (osteosarcomatous, chondrosarcomatous, or rhabdomyosarcomatous). In particular, small foci of benign osseous metaplasia were considered insufficient for the diagnosis, because these may be seen in benign calcified pleural plaques and may simply represent involvement of plaque by tumour. We also excluded mesothelioma with stromal osteoclast-like giant cells unless more definite evidence of osteoid/bone production was evident. Taking into account the clinical information regarding the anatomical distribution, a diagnosis of heterologous mesothelioma was made regardless of cytokeratin positivity, as for sarcomatoid mesothelioma.
Immunohistochemical Studies
As many of the cases were received in consultation, only limited immunohistochemical studies were available for much of the material. In addition, some of the immunohistochemical assessment was based on immunohistochemical stains that had been performed in other laboratories. All tissues had been fixed in 10% buffered formalin and had undergone standard processing and embedding in paraffin wax. Sections were cut 4 μm thick, deparaffinized, and rehydrated, if blocks had been received. For all of the cases received at Duke University and the majority of cases reviewed at Flinders Medical Center, the streptavidin-biotin-peroxidase complex method was used (Ultra Strepatavidin Detection System, Signet Laboratories) as a detection system, whereas for one of the cases the DakoCytomation EnVision + Dual Link System (Dako) was used.
Results
The salient case information for the tumours included in this study are summarized in Tables 1 and 2.
Incidence
Among 2694 mesothelioma cases at Duke University Medical Center between 1982 and 2008, 19 (0.6%) cases were identified, and 6 cases were identified among 594 consecutive malignant mesotheliomas accessioned at the Flinders Medical Centre (1%) over a 10-year period. These consecutive Flinders Medical Centre accessions do not include medicolegal referrals to one of the authors (DWH) over the same time for which no additional investigations were carried out and therefore were not included in the department accessions. There were no cases of ‘heterologous MM’ among those medicolegal referrals (estimated at approximately 1000 cases for which confirmation of diagnosis was sought), and had those cases been included, the percentage frequency for heterologous differentiation is estimated at about 0.5%. Two additional cases (Cases 26 and 27) were identified in the files of one of the authors (DWH) and predated the specified 10-year period. For these cases only limited clinical information was available. One of these cases (Case 26 in Table 1) had previously been included in the series by Yousem and Hochholzer.17 This case—a biphasic pleural mesothelioma with osseous differentiation—had also been described earlier by Langlois et al in 1978.26
Clinical Characteristics
Of the total of 27 cases of mesotheliomas with heterologous elements, 24 (89%) originated from the pleural cavity and 3 (11%) from the peritoneal cavity, one of which occurred in a woman. The median age at the time of diagnosis was 68 years, with an age range of 27–85 years. Twenty-five (93%) of the cases occurred in males and two (7%) in women. A history of exposure to asbestos was confirmed in all 17 cases for which an exposure history had been obtained (63% of all cases). Parietal pleural plaques were identified in nine of those patients, with no data available for the remaining patients.
Prognosis
Information regarding the clinical outcome was available for 12 of our 27 cases, with a median survival of 6 months after diagnosis and only one of the patients confirmed to be alive after 1 year.
Histological Features
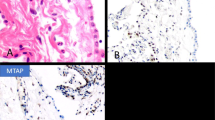
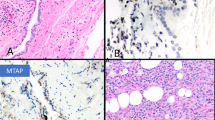
Only 1 of the 27 cases was reported as epithelioid mesothelioma, and in this case the diagnosis was based on a needle biopsy, which may not have adequately represented the tumour. No follow-up biopsy was available for that case. Of the remaining tumours, 16 mesotheliomas (59%) were sarcomatoid and 10 (37%) were biphasic. Of the cases, 11 showed osteosarcomatous elements only, 5 showed areas of rhabdomyosarcoma only, 5 contained areas of chondrosarcoma only, and 6 exhibited a mixture of heterologous elements. The osteosarcomatous areas varied from partly calcified osteoid in direct continuity with malignant spindle cells to large areas indistinguishable from osteosarcoma on H&E-stained sections (Figure 1). Similarly, areas of chondrosarcoma were indistinguishable from chondrosarcomas elsewhere, and generally were located directly adjacent to malignant spindle cell components (Figure 2). Rhabdomyoblastic areas were seen in close proximity to both spindle cell and epithelial components (Figure 3) and showed typical features of rhabdomyoblastic differentiation, including strap cells, large eccentric pleomorphic nuclei and abundant eosinophilic cytoplasm (Figure 3), with cross striations occasionally visible.
(a) Malignant mesothelioma showing an area of osteosarcoma adjacent to a spindle cell area. (b) Nuclear labelling of some of the spindle cells for the mesothelial-related marker calretinin. Not all sarcomatoid mesotheliomas show labelling for calretinin. (c) More extensive labelling of the spindle cells with the pancytokeratin marker AE1/AE3. The majority of sarcomatoid mesotheliomas labels for pancytokeratins.
(a) Area of chondrosarcoma (on the right) adjacent to a spindle cell area of predominantly sarcomatoid mesothelioma with minor epithelial tubular elements (arrow). (b) Higher power view of the transition of spindle cell sarcomatoid area to area of chondrosarcoma. (c) Labelling of the sarcomatoid spindle cell component and some of the chondrosarcoma component with the pancytokeratin marker AE1/AE3.
(a) Area of rhabdomyosarcoma intimately associated with sarcomatoid mesothelioma. (b) Desmin labelling of the rhabdomyoblastic cells. (c) Medium-power view showing typical strap cells (thin arrow) and rhabdomyoblast (short arrow) indicative of rhabdomyoblastic differentiation (d) Low-power view showing the same area (to the left) adjacent to a sarcomatoid area (middle of picture) and a minor epithelial component (right bottom corner of picture). (e) Positive labelling of rhabdomyoblasts for myoglobin. (f) Positive labelling of one of the same cells in an adjacent level for pancytokeratin AE1/AE3.
Immunnohistochemical Characteristics
The results of immunohistochemical studies were available for 26 of the 27 mesotheliomas included in the study and are summarized in Table 2.
No such data was available on case no. 1, where the diagnosis was based on typical clinical and histological appearances. For the remaining 26 cases, immunohistochemical studies for various cytokeratins had been performed on 23 tumours, and there was at least focal labelling in 20 of those cases, which included 12 sarcomatoid mesotheliomas, 7 biphasic mesotheliomas, and 1 epithelial mesothelioma.
Immunohistochemical labelling for calretinin had been performed on 11 cases, and 4 demonstrated positive nuclear staining, 3 of which were biphasic. The staining was predominantly located in the epithelial areas of the tumours. One of the sarcomatoid mesotheliomas labelled for calretinin, and there was equivocal labelling in one further sarcomatoid mesothelioma that was located in the peritoneal cavity. Eight of the nine tumours tested showed labelling for vimentin. Labelling for desmin was present in three of the four tumours where this marker had been studied. Only one of the three mesotheliomas tested showed strong membranous and circumferential staining for EMA typical of epithelial mesothelioma in the epithelial component of a biphasic tumour. None of the mesotheliomas for which immunohistochemical studies for the carcinoma-related markers (CEA, CD15, B72.3, Ber-EP4, and TTF-1) had been performed, labelled with any of these antibodies.
In addition, three mesotheliomas with areas of rhabdomyosarcoma showed labelling of rhabdomyoblasts with antibodies against MyoD1, and for two of these tumours there was also labelling with myogenin. Another case showed positive labelling of rhabdomyoblasts for myoglobin.
Discussion
Mesothelial cells are of mesodermal origin but retain the ability to differentiate towards an epithelial growth pattern during adult life. This multipotent behaviour of the mesothelium with its ability to differentiate into stromal phenotypes has been emphasized in the concept of the ‘mesodermoma’.19, 27, 28, 29 Due to this plasticity of differentiation, which is also evident in the spectrum of recognized histologic types of mesothelioma, mesothelial cells have been invoked as a source of adult stem cells.30 Differential patterns of gene expression have been associated with growth patterns and cellular phenotypes in vitro.31
Heterologous differentiation in mesothelioma is well recognized, mostly as individual case reports, the largest series consisting of the 12 cases being reported by Yousem and Hochholzer in 1987.17 If only the cases containing histologically confirmed malignant heterologous elements are considered, 23 cases of heterologous mesothelioma were identified in a search of the literature (Table 3).7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20 It seems likely that at least some of the cases that have in the past been reported as primary pleural sarcomas may in fact represent heterologous mesothelioma—those where no cytokeratin labelling was performed, the anatomical distribution was indistinguishable from mesothelioma, and where asbestos bodies were found in the lung parenchyma.32 We do not consider ‘rhabdoid’ change (as distinct from rhabdomyosarcomatous) as true heterologous differentiation, and the morphological appearances suggest an overlap with deciduoid13 and pleomorphic mesothelioma.
The incidence of these tumours in our series corresponds to approximately 0.5% of all mesothelioma cases. However, both Duke University and Flinders Medical Centre serve as major referral centres for mesothelioma, so this figure may overestimate the true prevalence.
Epidemiological Data
Of our cases, 11% were located in the peritoneal cavity, compared to 10% of cases in the published literature17 and 6% of all mesotheliomas. The M:F ratio of 13:1 in this series is relatively high, compared to 2:1 in the published cases of mesothelioma with heterologous elements, and approximately 5:1 to 7:1 for all mesotheliomas in HSE33 and Leigh and Driscoll.34 A definite exposure history to asbestos could be elicited in 63% of our cases, compared to 55% of the published cases (Tables 1 and 2). National mesothelioma registries report exposure to asbestos in approximately 90% of male34, 35 and 70% of female patients.36 Our data are likely to underestimate the true exposure to asbestos, because information regarding exposure was unavailable in 35% of our cases. Fibre analysis was performed in three cases and revealed a mixture of fibre types in two cases. The third was within the background range, despite a positive exposure history. The presence of asbestosis was specifically noted in only one of our cases, with four specifically commenting on the lack of asbestosis and no data available for the remaining cases. Our series confirms chondroid and osteoblastic differentiation as the most common patterns. A relatively large proportion of our cases (5/27) showed rhabdomyoblastic features (19% compared to 5% in the literature).
We suggest that the term heterologous mesothelioma should be reserved for those tumours that show malignant heterologous elements, namely osteosarcomatous, chondrosarcomatous, or rhabdomyoblastic elements. Care must be taken to exclude tumours showing benign metaplastic changes.37 Our series did not include any cases showing liposarcomatous differentiation, making it difficult to comment on. However, only few primary pleural liposarcomas38, 39, 40, 41, 42 and even fewer liposarcomas with pleural metastases have been described.43 Lack of labelling for keratin, knowledge of the clinical and radiological characteristics, and absence of a clinical history of soft tissue sarcoma are required for the diagnosis of primary liposarcoma at this site.40, 44
Definition of Heterologous Mesothelioma
We suggest that the criteria for recognition of osteosarcomatous differentiation should mirror those applied for primary osteosarcoma of bone and soft tissue. Recognition of osteoid can be difficult, but the presence of hyaline collagen with a trabecular and lacework pattern, isolating individual neoplastic cells or small groups, and the ‘normalization’ of cell pattern within osteoid trabeculae, whereby those cells are smaller and less pleomorphic than those at periphery, should be required for the diagnosis. If osteoid formation is considered equivocal, consultation with a soft tissue and bone specialist pathologist is recommended. The extent of heterologous differentiation may vary from minor and focal to extensive. We do not suggest a minimal percentage of heterologous elements necessary to make the diagnosis.
Differential Diagnosis and Role of Immunohistochemical Studies
The presence of heterologous elements within a pleural- or peritoneal-based tumour raises a differential diagnosis that includes pulmonary blastoma, primary sarcomas including synovial sarcomas, and metastases. In biopsy tissue, the distinction from primary chest wall osteosarcoma and chondrosarcoma (in contrast to pleura/peritoneum-based mesothelioma) requires correlation with the clinical-radiologic findings—in particular the exact location of the tumour, localized mass lesion as opposed to a confluent pleura-based tumour, and the age of the patient.45 Identification of epithelial components may greatly aid in the diagnosis, particularly if positivity with mesothelial-related markers can be demonstrated.
Primary osteosarcomas and chondrosarcomas of pleura have been described,32, 46 but pleural sarcomas other than mesothelioma, malignant solitary fibrous tumour, and pleural synovial sarcoma are extremely rare.43, 45 Some of the reported cases antedate immunohistochemical labelling for cytokeratins.32, 46, 47 However, even recent reports of extraskeletal oesteosarcoma, one in a 73-year-old patient with known exposure to asbestos and another of a 76-year-old Navy engineer (with no data regarding asbestos exposure available), make no mention of immunohistochemical studies that might have suggested a diagnosis of mesothelioma.48, 49
Heterologous elements have rarely been described in other primary pleural sarcomas, such as malignant fibrous histiocytoma and malignant nerve sheath tumour.50 We are not aware of pleural synovial sarcoma showing heterologous differentiation, but knowledge of the anatomical distribution and molecular studies demonstrating the SYT-SSX fusion gene would be helpful in that instance.51 A pleural localized mass lesion with an anatomical distribution distinct from mesothelioma and lack of labelling for cytokeratins has been reported as malignant pleural mesenchymoma with areas of liposarcoma, leiomyosarcoma, and osteocartilaginous differentiation,52 but it seems possible that this case might represent an example of a localized mesothelioma with heterologous differentiation.
Metastases of sarcomas including osteosarcoma and liposarcoma to the pleura have been described,53, 54, 55 but mimic mesothelioma only rarely, and can usually be recognized from the clinical history of an antecedent sarcoma. Furthermore, the clinical demographics are likely to be different.55, 56 A recent study by Knuuttila et al suggests that comparative genomic hybridization may help to distinguish mesothelioma from other chest wall sarcomas.6, 57
In summary, immunohistochemical findings confirm the usefulness of cytokeratin labelling for the purpose of diagnosing malignant mesothelioma, including sarcomatoid mesotheliomas. However, a proportion of these tumours (approximately 10% of sarcomatoid mesotheliomas) lack detectable expression of cytokeratins.6 Consequently, lack of labelling for cytokeratins in a spindle cell/sarcomatoid tumour does not exclude the diagnosis of mesothelioma, regardless of the presence of heterologous elements.6, 58, 59
Clinical Characteristics and Prognosis
The median survival of 6 months in our series is comparable to the published median survival of 5.5 months for mesothelioma with heterologous elements (data from Table 3) and similar to the 6.2 months survival seen for desmoplastic sarcomatoid mesothelioma.5 The slightly longer survival in our cases may in part reflect the presence of epithelial elements in the biphasic cases, a factor that has been associated with a better prognosis.60 Three cases of mesothelioma with heterologous elements with prolonged survival, including one with a reported survival of 69 months, have been reported.9, 61 Some authors attribute this prolonged survival to the expression of bone morphogenetic protein 2.61 For two of these cases the heterologous elements were described as ‘bland’, and these cases would not fulfill our strict criteria for diagnosis.
Summary
Heterologous mesothelioma is a rare subtype of mesothelioma characterized by the presence of any amount of malignant heterologous elements. These tumours show similar clinical characteristics to other types of mesothelioma, including a relationship to exposure to asbestos. The median survival of 6 months appears to be most akin to the prognosis for sarcomatoid mesothelioma. The differential diagnosis includes rare primary and metastatic pleural sarcomas, and the demonstration of positive labelling for cytokeratins by immunohistochemical studies may aid in that distinction. However, if the anatomical distribution conforms to that of mesothelioma, a diagnosis of heterologous mesothelioma should be made in preference to a diagnosis of primary pleural osteosarcoma or chondrosarcoma, irrespective of cytokeratin positivity, as has previously been recommended for conventional non-heterologous sarcomatoid mesothelioma.6
References
Churg A, Roggli V, Galateau-Salle F, et al. Mesothelioma. In: Travis WD, Brambilla E, Müller-Hermelink HK, Harris C, (eds). Pathology and Genetics of Tumours of the Lung, Pleura, Thymus and Heart. IARC: Lyon, 2004, pp 128–136.
Kannerstein M, Churg J . Desmoplastic diffuse malignant mesothelioma. In: Fenoglio CM, Wolff M, (eds). Progress in Surgical Pathology. Masson: New York, 1980, pp 19–29.
Henderson DW, Attwood HD, Constance TJ, et al. Lymphohistiocytoid mesothelioma: a rare lymphomatoid variant of predominantly sarcomatoid mesothelioma. Ultrastruct Pathol 1988;12:367–384.
Henderson DW, Shilkin KB, Langlois SL, et al. (eds). Malignant Mesothelioma. Hemisphere: New York, 1992, pp 412.
Cantin R, Al-Jabi M, McCaughey WT . Desmoplastic diffuse mesothelioma. Am J Surg Pathol 1982;6:215–222.
Galateau-Sallé F, (ed). International Mesothelioma Panel: Brambilla E, Cagle PT, Churg AM, Colby, TV, Gibbs AR, Hammar SP, Hasleton PS, Henderson DW, Inai K, Praet M, Roggli VL, Travis WD, Vignaud JM. Pathology of Malignant Mesothelioma. Springer: London, 2006.
Chave G, Chalabreysse L, Picaud G, et al. Malignant pleural mesothelioma with osteoblastic heterologous elements: CT and MR imaging findings. AJR Am J Roentgenol 2002;178:949–951.
Goldstein B . Two malignant pleural mesotheliomas with unusual histological features. Thorax 1979;34:375–379.
Kiyozuka Y, Miyazaki H, Yoshizawa K, et al. An autopsy case of malignant mesothelioma with osseous and cartilaginous differentiation: bone morphogenetic protein-2 in mesothelial cells and its tumor. Dig Dis Sci 1999;44:1626–1631.
Matsukuma S, Aida S, Hata Y, et al. Localized malignant peritoneal mesothelioma containing rhabdoid cells. Pathol Int 1996;46:389–391.
Okamoto T, Yokota S, Shinkawa K, et al. Pleural malignant mesothelioma with osseous cartilaginous and rhabdomyogenic differentiation. Nihon Kokyuki Gakkai Zasshi 1998;36:696–701.
Ordonez NG . Mesothelioma with rhabdoid features: an ultrastructural and immunohistochemical study of 10 cases. Mod Pathol 2006;19:373–383.
Puttagunta L, Vriend RA, Nguyen GK . Deciduoid epithelial mesothelioma of the pleura with focal rhabdoid change. Am J Surg Pathol 2000;24:1440–1443.
Rittinghausen S, Ernst H, Muhle H, et al. Atypical malignant mesotheliomas with osseous and cartilaginous differentiation after intraperitoneal injection of various types of mineral fibres in rats. Exp Toxicol Pathol 1992;44:55–58.
Suen HC, Sudholt B, Anderson WM, et al. Malignant mesothelioma with osseous differentiation. Ann Thorac Surg 2002;73:665.
Yoshii C, Imai S, Hoshino H, et al. A case of malignant pleural mesothelioma with osseous and cartilaginous formation. Nihon Kyobu Shikkan Gakkai Zasshi 1992;30:338–342.
Yousem SA, Hochholzer L . Malignant mesotheliomas with osseous and cartilaginous differentiation. Arch Pathol Lab Med 1987;111:62–66.
Raizon A, Schwartz A, Hix W, et al. Calcification as a sign of sarcomatous degeneration of malignant pleural mesotheliomas: a new CT finding. J Comput Assist Tomogr 1996;20:42–44.
Donna A, Betta PG, Bianchi V, et al. A new insight into the histogenesis of ‘mesodermomas’—malignant mesotheliomas. Histopathology 1991;19:239–244.
Krishna J, Haqqani MT . Liposarcomatous differentiation in diffuse pleural mesothelioma. Thorax 1993;48:409–410.
Donna A, Betta PG, Ribotta M . Lipoblastic differentiation in a primary localized fibrous mesothelioma of the peritoneum. Pathol Res Pract 1996;192:1252–1256; discussion 7–8.
Kuo T, London SN, Dinh TV . Endometriosis occurring in leiomyomatosis peritonealis disseminata: ultrastructural study and histogenetic consideration. Am J Surg Pathol 1980;4:197–204.
Attanoos RL, Suvarna SK, Rhead E, et al. Malignant vascular tumours of the pleura in ‘asbestos’ workers and endothelial differentiation in malignant mesothelioma. Thorax 2000;55:860–863.
Salgado RA, Corthouts R, Parizel PM, et al. Malignant pleural mesothelioma with heterologous osteoblastic elements: computed tomography, magnetic resonance, and positron emission tomography imaging characteristics of a rare tumor. J Comput Assist Tomogr 2005;29:653–656.
Quoix E, Chenard MP, Orion B, et al. A left pleural effusion with a calcified tumoral mass and left hemithoracic uptake on bone scan. Lung Cancer 2001;32:203–205.
Langlois SL, Glancy JJ, Henderson DW . The radiology of malignant pleural mesothelioma in Western Australia. Australas Radiol 1978;22:305–314.
Donna A, Betta PG . Mesodermomas: a new embryological approach to primary tumours of coelomic surfaces. Histopathology 1981;5:31–44.
Donna A, Betta PG . Differentiation towards cartilage and bone in a primary tumour of pleura. Further evidence in support of the concept of mesodermoma. Histopathology 1986;10:101–108.
Krismann M, Muller KM, Jaworska M, et al. Severe chromosomal aberrations in pleural mesotheliomas with unusual mesodermal features. Comparative genomic hybridization evidence for a mesothelioma subgroup. J Mol Diagn 2000;2:209–216.
Gotloib L, Gotloib LC, Khrizman V . The use of peritoneal mesothelium as a potential source of adult stem cells. Int J Artif Organs 2007;30:501–512.
Sun X, Gulyas M, Hjerpe A . Mesothelial differentiation as reflected by differential gene expression. Am J Respir Cell Mol Biol 2004;30:510–518.
Goetz SP, Robinson RA, Landas SK . Extraskeletal myxoid chondrosarcoma of the pleura. Report of a case clinically simulating mesothelioma. Am J Clin Pathol 1992;97:498–502.
HSE. Mesothelioma Mortality in Great Britain: Estimating the Future Burden. HSE: London, 2003.
Leigh J, Driscoll T . Malignant mesothelioma in Australia 1945–2002. Int J Occup Environ Health 2003;9:206–217.
Spirtas R, Heineman EF, Bernstein L, et al. Malignant mesothelioma: attributable risk of asbestos exposure. Occup Environ Med 1994;51:804–811.
Spirtas R, Connelly RR, Tucker MA . Survival patterns for malignant mesothelioma: the SEER experience. Int J Cancer 1988;41:525–530.
Fadare O, Bifulco C, Carter D, et al. Cartilaginous differentiation in peritoneal tissues: a report of two cases and a review of the literature. Mod Pathol 2002;15:777–780.
Benchetritt M, Hofman V, Venissac N, et al. Dedifferentiated liposarcoma of the pleura mimicking a malignant solitary fibrous tumor and associated with dedifferentiated liposarcoma of the mediastinum: usefulness of cytogenetic and molecular genetic analyses. Cancer Genet Cytogenet 2007;179:150–155.
Minniti A, Montaundon M, Jougon J, et al. Liposarcoma of the pleural cavity. An exceptional tumour. Monaldi Arch Chest Dis 2005;63:170–172.
Okby NT, Travis WD . Liposarcoma of the pleural cavity: clinical and pathologic features of 4 cases with a review of the literature. Arch Pathol Lab Med 2000;124:699–703.
Wong WW, Pluth JR, Grado GL, et al. Liposarcoma of the pleura. Mayo Clin Proc 1994;69:882–885.
Wouters EF, Greve LH, Visser R, et al. Liposarcoma of the pleura. Neth J Surg 1983;35:192–193.
Toro JR, Travis LB, Wu HJ, et al. Incidence patterns of soft tissue sarcomas, regardless of primary site, in the surveillance, epidemiology and end results program, 1978–2001: an analysis of 26 758 cases. Int J Cancer 2006;119:2922–2930.
Fujinami K, Kondoh K, Kondoh I, et al. A case of advanced retroperitoneal dedifferentiated liposarcoma treated effectively with high-dose ifosfamide. Hinyokika Kiyo 1999;45:463–466.
Gladish GW, Sabloff BM, Munden RF, et al. Primary thoracic sarcomas. Radiographics 2002;22:621–637.
Cohn L, Hall AD . Extraosseous osteogenic sarcoma of the pleura. Ann Thorac Surg 1968;5:545–549.
Pearson KD, Rubin D, Szemes GC, et al. Extraosseous osteogenic sarcoma of the chest. Br J Dis Chest 1969;63:231–234.
Sabloff B, Munden RF, Melhem AI, et al. Radiologic-pathologic conferences of the University of Texas M. D. Anderson Cancer Center: extraskeletal osteosarcoma of the pleura. AJR Am J Roentgenol 2003;180:972.
Chandak P, Hunt I, Rawlins R, et al. Bone or pleura? Primary pleural osteosarcoma. J Thorac Cardiovasc Surg 2007;133:587–588.
Ordonez NG, Tornos C . Malignant peripheral nerve sheath tumor of the pleura with epithelial and rhabdomyoblastic differentiation: report of a case clinically simulating mesothelioma. Am J Surg Pathol 1997;21:1515–1521.
Ten Heuvel SE, Hoekstra HJ, Suurmeijer AJ . Diagnostic Accuracy of FISH and RT-PCR in 50 routinely processed synovial sarcomas. Appl Immunohistochem Mol Morphol 2008;16:246–250.
Deslee G, Guillou PJ, Baehrel B, et al. Malignant mesenchymoma of the pleura. Interact Cardiovasc Thorac Surg 2003;2:376–378.
Shanley DJ, Mulligan ME . Osteosarcoma with isolated metastases to the pleura. Pediatr Radiol 1991;21:226.
Eskenasy A . Histopathology of tumorous ribs: a review of 130 cases. Morphol Embryol (Bucur) 1985;31:35–50.
Mori T, Yoshioka M, Iwatani K, et al. Kissing pleural metastases from metastatic osteosarcoma of the lung. Ann Thorac Cardiovasc Surg 2006;12:129–132.
Bai H, Aswad BI, Gaissert H, et al. Malignant solitary fibrous tumor of the pleura with liposarcomatous differentiation. Arch Pathol Lab Med 2001;125:406–409.
Knuuttila A, Jee KJ, Taskinen E, et al. Spindle cell tumours of the pleura: a clinical, histological and comparative genomic hybridization analysis of 14 cases. Virchows Arch 2006;448:135–141.
Attanoos RL, Dojcinov SD, Webb R, et al. Anti-mesothelial markers in sarcomatoid mesothelioma and other spindle cell neoplasms. Histopathology 2000;37:224–231.
Lucas DR, Pass HI, Madan SK, et al. Sarcomatoid mesothelioma and its histological mimics: a comparative immunohistochemical study. Histopathology 2003;42:270–279.
Antman K, Shemin R, Ryan L, et al. Malignant mesothelioma: prognostic variables in a registry of 180 patients, the Dana-Farber Cancer Institute and Brigham and Women's Hospital experience over two decades, 1965–1985. J Clin Oncol 1988;6:147–153.
Demirag F, Unsal E, Tastepe I . Biphasic malignant mesothelioma cases with osseous differentiation and long survival: a review of the literature. Lung Cancer 2007;57:233–236.
Acknowledgements
Commercial relationships policy: none.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Klebe, S., Mahar, A., Henderson, D. et al. Malignant mesothelioma with heterologous elements: clinicopathological correlation of 27 cases and literature review. Mod Pathol 21, 1084–1094 (2008). https://doi.org/10.1038/modpathol.2008.125
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/modpathol.2008.125
Keywords
This article is cited by
-
Malignant peritoneal mesothelioma: prognostic significance of clinical and pathologic parameters and validation of a nuclear-grading system in a multi-institutional series of 225 cases
Modern Pathology (2021)
-
Heterologous sarcomatoid pleural mesothelioma with osteosarcomatous differentiation: a report of autopsy case that accomplished trimodality therapy and review of the literature
General Thoracic and Cardiovascular Surgery (2020)
-
A Subset of Malignant Mesothelioma Tumors Retain Osteogenic Potential
Scientific Reports (2016)