Abstract
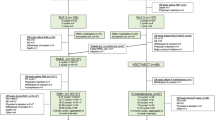
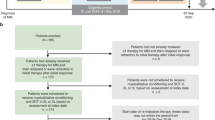
High-dose therapy with autologous stem cell transplantation (HDT–ASCT) has been considered to be the standard frontline treatment for younger, fit patients with multiple myeloma (MM) since the 1990s. Efforts continue to optimize the use of HDT–ASCT with the aim of improving outcomes. One strategy has been the incorporation of novel agents (thalidomide, lenalidomide and bortezomib) in the pre-transplantation setting as an induction therapy or in the post-transplantation setting as a consolidation or maintenance therapy. Given their high response rates, three-drug induction therapy regimens (for example, bortezomib–thalidomide–dexamethasone, lenalidomide–bortezomib–dexamethasone and cyclophosphamide–bortezomib–dexamethasone) are now the standard of care. Thalidomide and bortezomib are well suited for consolidation therapy, and regimens using these agents can improve the depth of response following HDT–ASCT. Lenalidomide is particularly well suited for long-term maintenance therapy following HDT–ASCT, and initial results are promising and have shown improvements in disease outcomes such as progression-free survival and overall survival in some cases, although a low incidence of second primary malignancies have been observed. Further studies are needed to determine the optimal regimen and duration of induction therapy, the impact of maintenance on overall survival and the safety of long-term treatment. Many of the studies currently underway in MM will help address these aspects.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
We are sorry, but there is no personal subscription option available for your country.
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Moreau P, Avet-Loiseau H, Harousseau JL, Attal M . Current trends in autologous stem-cell transplantation for myeloma in the era of novel therapies. J Clin Oncol 2011; 29: 1898–1906.
Attal M, Harousseau JL, Facon T, Guilhot F, Doyen C, Fuzibet JG et al. Single versus double autologous stem-cell transplantation for multiple myeloma. N Engl J Med 2003; 349: 2495–2502.
Macro M, Divine M, Uzunhan Y, Jaccard A, Bouscary D, Leblond V et al. Dexamethasone + thalidomide (Dex/Thal) compared to VAD as pre-transplant treatment in newly diagnosed multiple myeloma (MM): a randomized trial. Blood 2006; 108: (abstract 57).
Lokhorst HM, Schmidt-Wolf I, Sonneveld P, van der Holt B, Martin H, Barge R et al. Thalidomide in induction treatment increases the very good partial remission rate before and after high-dose therapy in previously untreated multiple myeloma. Haematologica 2008; 93: 124–127.
Lokhorst HM, van der Holt B, Zweegman S, Vellenga E, Croockewit S, van Oers MH et al. A randomized phase 3 study on the effect of thalidomide combined with adriamycin, dexamethasone, and high-dose melphalan, followed by thalidomide maintenance in patients with multiple myeloma. Blood 2010; 115: 1113–1120.
Morgan GJ, Davies FE, Owen RG, Rawstron AC, Bell S, Cocks K et al. Thalidomide combinations improve response rates; results from the MRC IX study. Blood 2007; 110: (abstract 3593).
Harousseau JL, Attal M, Avet-Loiseau H, Marit G, Caillot D, Mohty M et al. Bortezomib plus dexamethasone is superior to vincristine plus doxorubicin plus dexamethasone as induction treatment prior to autologous stem-cell transplantation in newly diagnosed multiple myeloma: results of the IFM 2005-01 phase III trial. J Clin Oncol 2010; 28: 4621–4629.
Wang M, Giralt S, Delasalle K, Handy B, Alexanian R . Bortezomib in combination with thalidomide-dexamethasone for previously untreated multiple myeloma. Hematology 2007; 12: 235–239.
Popat R, Oakervee HE, Hallam S, Curry N, Odeh L, Foot N et al. Bortezomib, doxorubicin and dexamethasone (PAD) front-line treatment of multiple myeloma: updated results after long-term follow-up. Br J Haematol 2008; 141: 512–516.
Richardson PG, Weller E, Lonial S, Jakubowiak AJ, Jagannath S, Raje NS et al. Lenalidomide, bortezomib, and dexamethasone combination therapy in patients with newly diagnosed multiple myeloma. Blood 2010; 116: 679–686.
Reeder CB, Reece DE, Kukreti V, Chen C, Trudel S, Hentz J et al. Cyclophosphamide, bortezomib and dexamethasone induction for newly diagnosed multiple myeloma: high response rates in a phase II clinical trial. Leukemia 2009; 23: 1337–1341.
Cavo M, Tacchetti P, Patriarca F, Petrucci MT, Pantani L, Galli M et al. Bortezomib with thalidomide plus dexamethasone compared with thalidomide plus dexamethasone as induction therapy before, and consolidation therapy after, double autologous stem-cell transplantation in newly diagnosed multiple myeloma: a randomised phase 3 study. Lancet 2010; 376: 2075–2085.
Rosiñol L, Oriol A, Teruel AI, Hernandez D, Lopez-Jimenez J, de la Rubia J et al. Superiority of bortezomib, thalidomide, and dexamethasone (VTD) as induction pretransplantation therapy in multiple myeloma: a randomized phase 3 PETHEMA/GEM study. Blood 2012; 120: 1589–1596.
Moreau P, Avet-Loiseau H, Facon T, Attal M, Tiab M, Hulin C et al. Bortezomib plus dexamethasone versus reduced-dose bortezomib, thalidomide plus dexamethasone as induction treatment before autologous stem cell transplantation in newly diagnosed multiple myeloma. Blood 2011; 118: 5752–5758.
Sonneveld P, Schmidt-Wolf I, van der Holt B, el Jarari L, Bertsch U, Salwender H et al. Bortezomib induction and maintenance treatment in patients with newly diagnosed multiple myeloma: results of the randomized phase III HOVON-65/GMMG-HD4 trial. J Clin Oncol 2012; 30: 2946–2955.
Ladetto M, Pagliano G, Ferrero S, Cavallo F, Drandi D, Santo L et al. Major tumor shrinking and persistent molecular remissions after consolidation with bortezomib, thalidomide, and dexamethasone in patients with autografted myeloma. J Clin Oncol 2010; 28: 2077–2084.
Cavo M, Pantani L, Petrucci MT, Patriarca F, Zamagni E, Donnarumma D et al. Bortezomib-thalidomide-dexamethasone is superior to thalidomide-dexamethasone as consolidation therapy following hematopoietic stem cell transplantation in patients with newly diagnosed multiple myeloma. Blood 2012; 120: 9–19.
Terragna C, Zamagni E, Petrucci MT, Durante S, Patriarca F, Narni F et al. Molecular remission after bortezomib-thalidomide-dexamethasone compared with thalidomide-dexamethasone as consolidation therapy following double autologous transplantation for multiple myeloma: results of a qualitative and quantitative analysis. Blood 2010; 116: (abstract 861).
Roussel M, Avet-Loiseau H, Moreau P, Huynh A, Benboubker L, Hulin C et al. Frontline therapy with bortezomib, lenalidomide, and dexamethasone (VRD) induction followed by autologous stem cell transplantation, VRD consolidation and lenalidomide maintenance in newly diagnosed multiple myeloma patients: primary results of the IFM 2008 phase II study. Blood 2010; 116: (abstract 624).
Attal M, Lauwers-Cances V, Marit G, Caillot D, Moreau P, Facon T et al. Lenalidomide maintenance after stem-cell transplantation for multiple myeloma. N Engl J Med 2012; 366: 1782–1791.
Mellqvist UH, Gimsing P, Hjertner O, Lenhoff S, Laane E, Remes K et al. Improved progression free survival with bortezomib consolidation after high dose melphalan; results of a randomized phase III trial. Haematologica 2011; 96 (Suppl 1): (abstract O-11).
Rajkumar SV, Harousseau JL, Durie B, Anderson KC, Dimopoulos M, Kyle R et al. Consensus recommendations for the uniform reporting of clinical trials: report of the International Myeloma Workshop Consensus Panel 1. Blood 2011; 117: 4691–4695.
Attal M, Harousseau JL, Leyvraz S, Doyen C, Hulin C, Benboubker L et al. Maintenance therapy with thalidomide improves survival in patients with multiple myeloma. Blood 2006; 108: 3289–3294.
Barlogie B, Pineda-Roman M, van Rhee F, Haessler J, Anaissie E, Hollmig K et al. Thalidomide arm of Total Therapy 2 improves complete remission duration and survival in myeloma patients with metaphase cytogenetic abnormalities. Blood 2008; 112: 3115–3121.
Spencer A, Prince HM, Roberts AW, Prosser IW, Bradstock KF, Coyle L et al. Consolidation therapy with low-dose thalidomide and prednisolone prolongs the survival of multiple myeloma patients undergoing a single autologous stem-cell transplantation procedure. J Clin Oncol 2009; 27: 1788–1793.
Morgan GJ, Gregory WM, Davies FE, Bell SE, Szubert AJ, Brown JM et al. The role of maintenance thalidomide therapy in multiple myeloma: MRC Myeloma IX results and meta-analysis. Blood 2012; 119: 7–15.
Barlogie B, Tricot G, Anaissie E, Shaughnessy J, Rasmussen E, van Rhee F et al. Thalidomide and hematopoietic-cell transplantation for multiple myeloma. N Engl J Med 2006; 354: 1021–1030.
Barlogie B, Attal M, Crowley J, van Rhee F, Szymonifka J, Moreau P et al. Long-term follow-up of autotransplantation trials for multiple myeloma: update of protocols conducted by the Intergroupe Francophone du Myelome, Southwest Oncology Group, and University of Arkansas for Medical Sciences. J Clin Oncol 2010; 28: 1209–1214.
McCarthy PL, Owzar K, Hofmeister CC, Hurd DD, Hassoun H, Richardson PG et al. Lenalidomide after stem-cell transplantation for multiple myeloma. N Engl J Med 2012; 366: 1770–1781.
Cavo M, Rajkumar SV, Palumbo A, Moreau P, Orlowski R, Bladé J et al. International Myeloma Working Group consensus approach to the treatment of multiple myeloma patients who are candidates for autologous stem cell transplantation. Blood 2011; 117: 6063–6073.
Boccadoro M, Cavallo F, Nagler A, Ben Yehuda D, Omedè P, Cavalli M et al. Melphalan/prednisone/lenalidomide (MPR) versus high-dose melphalan and autologous transplantation (MEL200) in newly diagnosed multiple myeloma (MM) patients: a phase III trial. J Clin Oncol 2011; 29 (Suppl 15): (abstract 8020).
Acknowledgements
We thank Shanthi Jayawardena, PhD, and Eva Polk, PhD (Excerpta Medica), for the linguistic improvement of the manuscript. Editorial support in the preparation of this manuscript was funded by Celgene Corporation. PM and CT were fully responsible for all content and editorial decisions for this manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
PM has participated in the Advisory Boards for Celgene Corporation, Millennium, Janssen and Onyx Pharmaceuticals, and has received honoraria from Celgene Corporation, Millennium and Janssen. CT declares no conflicts of interest.
Rights and permissions
About this article
Cite this article
Moreau, P., Touzeau, C. Optimizing outcomes for patients with newly diagnosed multiple myeloma eligible for transplantation. Leukemia Suppl 2 (Suppl 1), S15–S20 (2013). https://doi.org/10.1038/leusup.2013.4
Published:
Issue Date:
DOI: https://doi.org/10.1038/leusup.2013.4