Abstract
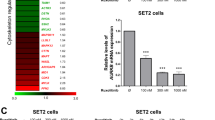
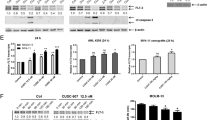
Leukemias bearing CRLF2 and JAK2 gene alterations are characterized by aberrant JAK/STAT signaling and poor prognosis. The HDAC inhibitor givinostat/ITF2357 has been shown to exert anti-neoplastic activity against both systemic juvenile idiopathic arthritis and myeloproliferative neoplasms through inhibition of the JAK/STAT pathway. These findings led us to hypothesize that givinostat might also act against CRLF2-rearranged BCP-ALL, which lack effective therapies. Here, we found that givinostat inhibited proliferation and induced apoptosis of BCP-ALL CRLF2-rearranged cell lines, positive for exon 16 JAK2 mutations. Likewise, givinostat killed primary cells, but not their normal hematopoietic counterparts, from patients carrying CRLF2 rearrangements. At low doses, givinostat downregulated the expression of genes belonging to the JAK/STAT pathway and inhibited STAT5 phosphorylation. In vivo, givinostat significantly reduced engraftment of human blasts in patient-derived xenograft models of CRLF2-positive BCP-ALL. Importantly, givinostat killed ruxolitinib-resistant cells and potentiated the effect of current chemotherapy. Thus, givinostat in combination with conventional chemotherapy may represent an effective therapeutic option for these difficult–to-treat subsets of ALL. Lastly, the selective killing of cancer cells by givinostat may allow the design of reduced intensity regimens in CRLF2-rearranged Down syndrome-associated BCP-ALL patients with an overall benefit in terms of both toxicity and related complications.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Hunger SP, Lu X, Devidas M, Camitta BM, Gaynon PS, Winick NJ et al. Improved survival for children and adolescents with acute lymphoblastic leukemia between 1990 and 2005: A report from the children’s oncology group. J Clin Oncol 2012; 30: 1663–1669.
Parker C, Waters R, Leighton C, Hancock J, Sutton R, Moorman AV et al. Effect of mitoxantrone on outcome of children with first relapse of acute lymphoblastic leukaemia (ALL R3): an open-label randomised trial. Lancet 2010; 376: 2009–2017.
Hof J, Krentz S, van Schewick C, Körner G, Shalapour S, Rhein P et al. Mutations and deletions of the TP53 gene predict nonresponse to treatment and poor outcome in first relapse of childhood acute lymphoblastic leukemia. J Clin Oncol 2011; 29: 3185–3193.
Russell LJ, Capasso M, Vater I, Akasaka T, Bernard OA, Calasanz MJ et al. Deregulated expression of cytokine receptor gene, CRLF2, is involved in lymphoid transformation in B-cell precursor acute lymphoblastic leukemia. Blood 2009; 114: 2688–2698.
Chen I, Harvey RC, Mullighan CG, Gastier-foster J, Wharton W, Kang H et al. Outcome modeling with CRLF2, IKZF1, JAK, and minimal residual disease in pediatric acute lymphoblastic leukemia: a Children’s Oncology Group Study. Blood 2012; 119: 3512–3523.
Harvey RC, Mullighan CG, Wang X, Dobbin KK, Davidson GS, Bedrick EJ et al. Identification of novel cluster groups in pediatric high-risk B-precursor acute lymphoblastic leukemia with gene expression profiling: correlation with genome-wide DNA copy number alterations, clinical characteristics, and outcome Identification of nov. Blood 2014; 116: 4874–4884.
Mullighan CG, Collins-underwood JR, Phillips LAA, Loudin MG, Liu W, Zhang J et al. Rearrangement of CRLF2 in B-progenitor – and Down syndrome – associated acute lymphoblastic leukemia. Nat Genet 2009; 41: 1243–1246.
Hertzberg L, Vendramini E, Ganmore I, Cazzaniga G, Schmitz M, Chalker J et al. Down syndrome acute lymphoblastic leukemia, a highly heterogeneous disease in which aberrant expression of CRLF2 is associated with mutated JAK2: a report from the International BFM Study Group. Blood 2010; 115: 1006–1017.
Carpino N, Thierfelder WE, Chang M, Turner SJ, Ziegler SF, Ihle JN et al. Absence of an essential role for thymic stromal lymphopoietin receptor in murine B-cell development absence of an essential role for thymic stromal lymphopoietin receptor in murine B-cell development. Mol Cell Biol 2004; 24: 2584–2592.
Tasian SK, Doral MY, Borowitz MJ, Wood BL, Chen I, Harvey RC et al. Aberrant STAT5 and PI3K / mTOR pathway signaling occurs in human CRLF2 -rearranged B-precursor acute lymphoblastic leukemia. Blood 2012; 2: 833–843.
Mullighan CG, Zhang J, Harvey RC, Collins-Underwood JR, Schulman Ba, Phillips La et al. JAK mutations in high-risk childhood acute lymphoblastic leukemia. Proc Natl Acad Sci USA 2009; 106: 9414–9418.
Yoda A, Yoda Y, Chiaretti S, Bar-Natan M, Mani K, Rodig SJ et al. Functional screening identifies CRLF2 in precursor B-cell acute lymphoblastic leukemia. Proc Natl Acad Sci USA 2010; 107: 252–257.
Heim MH . The Jak-STAT pathway: cytokine signalling from the receptor to the nucleus. J Recept Signal Transduct Res 1999; 19: 75–120.
Baxter EJ, Scott LM, Campbell PJ, East C, Fourouclas N, Swanton S et al. Acquired mutation of the tyrosine kinase JAK2 in human myeloproliferative disorders. Lancet 2005; 365: 1054–1061.
Roberts KG, Morin RD, Zhang J, Hirst M, Zhao Y, Su X et al. Genetic alterations activating kinase and cytokine receptor signaling in high-risk acute lymphoblastic leukemia. Cancer Cell 2012; 22: 153–166.
Weigert O, Lane Aa, Bird L, Kopp N, Chapuy B, van Bodegom D et al. Genetic resistance to JAK2 enzymatic inhibitors is overcome by HSP90 inhibition. J Exp Med 2012; 209: 259–273.
Maude S, Tasian S, Vincent T, Hall J, Roberts K, Collins R et al. Targeting JAK2 and mtor in xenograft models of crlf2-overexpressing acute lymphoblastic leukemia (ALL). Pediatr Blood Cancer 2012; 58: 1014.
Maude SL, Dolai S, Delgado-martin C, Vincent T, Robbins A, Selvanathan A et al. Efficacy of JAK / STAT pathway inhibition in murine xenograft models of early T-cell precursor (ETP) acute lymphoblastic leukemia. Blood 2015; 125: 1759–1768.
Springuel L, Hornakova T, Losdyck E, Lambert F, Leroy E, Constantinescu SN et al. Cooperating JAK1 and JAK3 mutants increase resistance to JAK inhibitors. Blood 2014; 124: 3924–3931.
Rambaldi A, Dellacasa CM, Finazzi G, Carobbio A, Ferrari ML, Guglielmelli P et al. A pilot study of the Histone-Deacetylase inhibitor Givinostat in patients with JAK2V617F positive chronic myeloproliferative neoplasms. Br J Haematol 2010; 150: 446–455.
Finazzi G, Vannucchi AM, Martinelli V, Ruggeri M, Nobile F, Specchia G et al. A phase II study of Givinostat in combination with hydroxycarbamide in patients with polycythaemia vera unresponsive to hydroxycarbamide monotherapy. Br J Haematol 2013; 161: 688–694.
Guerini V, Barbui V, Spinelli O, Salvi A, Dellacasa C, Carobbio A et al. The histone deacetylase inhibitor ITF2357 selectively targets cells bearing mutated JAK2(V617F). Leukemia 2008; 22: 740–747.
Ganai SA . Histone deacetylase inhibitor givinostat: the small-molecule with promising activity against therapeutically challenging haematological malignancies. J Chemother 2016; 28: 247–254.
Galli M, Salmoiraghi S, Golay J, Gozzini A, Crippa C, Pescosta N . A phase II multiple dose clinical trial of histone deacetylase inhibitor ITF2357 in patients with relapsed or progressive multiple myeloma. Ann Hematol 2010; 89: 185–190.
Vojinovic J, Damjanov N, D’Urzo C, Furlan A, Susic G, Pasic S et al. Safety and efficacy of an oral histone deacetylase inhibitor in systemic-onset juvenile idiopathic arthritis. Arthritis Rheum 2011; 63: 1452–1458.
Bettica P, Petrini S, Oria VD, Amico AD, Catteruccia M, Pane M et al. Histological effects of givinostat in boys with Duchenne muscular dystrophy. Neuromuscul Disord 2016; 26: 643–649.
Pinz S, Unser S, Buob D, Fischer P, Jobst B, Rascle A . Deacetylase inhibitors repress STAT5-mediated transcription by interfering with bromodomain and extra-terminal (BET) protein function. Nucleic Acids Res 2015; 43: 3524–3545.
Amaru Calzada A, Todoerti K, Donadoni L, Pellicioli A, Tuana G, Gatta R et al. The HDAC inhibitor Givinostat modulates the hematopoietic transcription factors NFE2 and C-MYB in JAK2 V617F myeloproliferative neoplasm cells. Exp Hematol 2012; 40: 634–645.
Krutzik PO, Nolan GP . Intracellular phospho-protein staining techniques for flow cytometry: monitoring single cell signaling events. Cytom Part A 2003; 55A: 61–70.
Greco WR, Bravo G, Parsons JC . The search for synergy: a critical review from a response surface perspective. Pharmacol Rev 1995; 47: 331–385.
Butler LM, Agus DB, Scher HI, Higgins B, Rose A, Cordon-cardo C et al. Suberoylanilide hydroxamic acid, an inhibitor of histone deacetylase, suppresses the growth of prostate cancer cells in vitro and in vivo 1. Cancer Res 2000; 60: 5165–5170.
Mann BS, Johnson JR, Cohen MH, Justice R, Pazdur R . FDA approval summary: vorinostat for treatment of advanced primary cutaneous T-cell lymphoma. Oncologist 2007; 12: 1247–1252.
Bugarin C, Sarno J, Palmi C, Savino AM, te Kronnie G, Dworzak M et al. Fine tuning of surface CRLF2 expression and its associated signaling profile in childhood B cell precursor acute lymphoblastic leukemia. Haematologica 2015; 100: 229–232.
Bercovich D, Ganmore I, Scott LM, Wainreb G, Birger Y, Elimelech A et al. Mutations of JAK2 in acute lymphoblastic leukaemias associated with Down’s syndrome. Lancet 2008; 372: 1484–1492.
Conter V, Bartram CR, Valsecchi MG, Schrauder A, Panzer-Grümayer R, Möricke A et al. Molecular response to treatment redefines all prognostic factors in children and adolescents with B-cell precursor acute lymphoblastic leukemia: results in 3184 patients of the AIEOP-BFM ALL 2000 study. Blood 2010; 115: 3206–3214.
Spijkers-Hagelstein JaP, Pinhanços SS, Schneider P, Pieters R, Stam RW . Chemical genomic screening identifies LY294002 as a modulator of glucocorticoid resistance in MLL-rearranged infant ALL. Leukemia 2014; 28: 761–769.
Evrot E, Ebel N, Romanet V, Roelli C, Andraos R, Qian Z et al. JAK1/2 and pan-deacetylase inhibitor combination therapy yields improved efficacy in preclinical mouse models of JAK2V617F-driven disease. Clin Cancer Res 2013; 19: 6230–6241.
Koppikar P, Bhagwat N, Kilpivaara O, Manshouri T, Adli M, Hricik T et al. Heterodimeric JAK-STAT activation as a mechanism of persistence to JAK2 inhibitor therapy. Nature 2012; 489: 155–159.
Stumpel D, Schneider P, Seslija L, Osaki H, Williams O, Pieters R et al. Connectivity mapping identifies HDAC inhibitors for the treatment of t(4;11)-positive infant acute lymphoblastic leukemia. Leukemia 2012; 26: 682–692.
Bhatla T, Wang J, Morrison DJ, Raetz EA, Burke MJ, Brown P et al. Epigenetic reprogramming reverses the relapse-specific gene expression signature and restores chemosensitivity in childhood B-lymphoblastic leukemia. Blood 2012; 119: 5201–5210.
Stubbs MC, Kim W, Bariteau M, Davis T, Vempati S, Minehart J et al. Selective inhibition of HDAC1 and HDAC2 as a potential therapeutic option for B-ALL. Clin Cancer Res 2015; 21: 2348–2358.
Beagle BR, Nguyen DM, Mallya S, Tang SS, Lu M, Zeng Z et al. mTOR kinase inhibitors synergize with histone deacetylase inhibitors to kill B-cell acute lymphoblastic leukemia cells. Oncotarget 2014; 6: 2088–2100.
Pinazza M, Borga C, Agnusdei V, Minuzzo S, Fossati G, Paganin M et al. An immediate transcriptional signature associated with response to the histone deacetylase inhibitor Givinostat in T acute lymphoblastic leukemia xenografts. Cell Death Dis 2016; 6: e2047.
Loh ML, Tasian SK, Rabin KR, Brown P, Magoon D, Reid JM et al. A phase 1 dosing study of ruxolitinib in children with relapsed or refractory solid tumors, leukemias, or myeloproliferative neoplasms: A Children’s Oncology Group phase 1 consortium study (ADVL1011). Pediatr Blood Cancer 2015; 62: 1717–1724.
Izraeli S, Vora A, Zwaan CM, Whitlock J . How I treat all in down’s syndrome: pathobiology and management. Blood 2014; 123: 35–40.
Stankov MV, El Khatib M, Kumar Thakur B, Heitmann K, Panayotova-Dimitrova D, Schoening J et al. Histone deacetylase inhibitors induce apoptosis in myeloid leukemia by suppressing autophagy. Leukemia 2014; 28: 577–588.
Acknowledgements
AM Savino is supported by a grant from Associazione Italiana per la Ricerca sul Cancro (AIRC and FIRC, grant no. 16645) from the parents’ association ‘Insieme ad Andrea si può, ONLUS’ and from the family of Alessandra Aloisi. This study was supported by grants from: Fondazione Tettamanti, Fondazione Città della Speranza, Associazione Italiana Ricerca sul Cancro (AIRC) (to GteK, GC, AB), CARIPARO project of excellence (to GteK), Fondazione Cariplo (to AB, GC and GteK), Beat Leukemia Foundation (to CP) The Israel Science Foundation, Legacy program and the Israeli Health Ministry (to SI) the European Union’s Seventh Framework Program (FP7/2007-2013) under the project European Network for Cancer research in Children and Adolescents (ENCCA, grant agreement HEALTH-F2-2011-261474) (to AB, GC and GteK). We thank Dr Andrea Ballerini for many helpful discussions and Riccardo Milan for graphic support.
Author contributions
AMS, JS, LT, MV, GF, MB, CB and CP performed the experiments; AMS, JS, LT and CP analyzed the data; AMS, LT, KD, CP and GC wrote the manuscript; GF, KD, GG, SI, LHM, GPN, AB and GTK supervised the research; CP and GC designed the study and supervised the research.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
GF is an employee of Italfarmaco SpA. GPN is a paid consultant for Fluidigm, the manufacturer that produced some of the reagents and instrumentation used in this manuscript. The remaining authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Leukemia website
Supplementary information
Rights and permissions
About this article
Cite this article
Savino, A., Sarno, J., Trentin, L. et al. The histone deacetylase inhibitor givinostat (ITF2357) exhibits potent anti-tumor activity against CRLF2-rearranged BCP-ALL. Leukemia 31, 2365–2375 (2017). https://doi.org/10.1038/leu.2017.93
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/leu.2017.93
This article is cited by
-
Chromatin modifiers in human disease: from functional roles to regulatory mechanisms
Molecular Biomedicine (2024)
-
Cancer epigenetics: from laboratory studies and clinical trials to precision medicine
Cell Death Discovery (2024)
-
HDAC inhibitor ITF2357 reduces resistance of mutant-KRAS non-small cell lung cancer to pemetrexed through a HDAC2/miR-130a-3p-dependent mechanism
Journal of Translational Medicine (2023)
-
Epigenetic regulation in hematopoiesis and its implications in the targeted therapy of hematologic malignancies
Signal Transduction and Targeted Therapy (2023)
-
Exploring the oncogenic and therapeutic target potential of the MYB-TYK2 fusion gene in B-cell acute lymphoblastic leukemia
Cancer Gene Therapy (2022)