Abstract
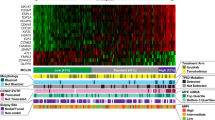
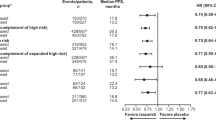
Lenalidomide is an immunomodulatory compound with high clinical activity in multiple myeloma. Lenalidomide binding to the Cereblon (CRBN) E3 ubiquitin ligase results in targeted ubiquitination and degradation of the lymphoid transcription factors Ikaros (IKZF1) and Aiolos (IKZF3) leading to growth inhibition of multiple myeloma cells. Recently, Basigin (BSG) was identified as another protein regulated by CRBN that is involved in the activity of lenalidomide. Here, we analyzed the prognostic value of IKZF1, IKZF3, CRBN and BSG mRNA expression levels in pretreatment plasma cells from 60 patients with newly diagnosed multiple myeloma uniformly treated with lenalidomide in combination with intensive chemotherapy within a clinical trial. We found that IKZF1 mRNA expression levels are significantly associated with progression-free survival (PFS). Patients in the lowest quartile (Q1) of IKZF1 expression had a superior PFS compared with patients in the remaining quartiles (Q2–Q4; 3-year PFS of 86 vs 51%, P=0.01). This translated into a significant better overall survival (100 vs 74%, P=0.03). Subgroup analysis revealed a significant impact of IKZF1, IKZF3 and BSG expression levels on PFS in cytogenetically defined standard-risk but not high-risk patients. Our data suggest a prognostic role of IKZF1, IKZF3 and BSG expression levels in lenalidomide-treated multiple myeloma.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Rajkumar SV, Hayman SR, Lacy MQ, Dispenzieri A, Geyer SM, Kabat B et al. Combination therapy with lenalidomide plus dexamethasone (Rev/Dex) for newly diagnosed myeloma. Blood 2005; 106: 4050–4053.
Richardson PG, Blood E, Mitsiades CS, Jagannath S, Zeldenrust SR, Alsina M et al. A randomized phase 2 study of lenalidomide therapy for patients with relapsed or relapsed and refractory multiple myeloma. Blood 2006; 108: 3458–3464.
Dimopoulos M, Spencer A, Attal M, Prince HM, Harousseau JL, Dmoszynska A et al. Lenalidomide plus dexamethasone for relapsed or refractory multiple myeloma. N Engl J Med 2007; 357: 2123–2132.
Weber DM, Chen C, Niesvizky R, Wang M, Belch A, Stadtmauer EA et al. Lenalidomide plus dexamethasone for relapsed multiple myeloma in North America. N Engl J Med 2007; 357: 2133–2142.
Knop S, Gerecke C, Liebisch P, Topp MS, Platzbecker U, Sezer O et al. Lenalidomide, adriamycin, and dexamethasone (RAD) in patients with relapsed and refractory multiple myeloma: a report from the German Myeloma Study Group DSMM (Deutsche Studiengruppe Multiples Myelom). Blood 2009; 113: 4137–4143.
Benboubker L, Dimopoulos MA, Dispenzieri A, Catalano J, Belch AR, Cavo M et al. Lenalidomide and dexamethasone in transplant-ineligible patients with myeloma. N Engl J Med 2014; 371: 906–917.
Roussel M, Lauwers-Cances V, Robillard N, Hulin C, Leleu X, Benboubker L et al. Front-line transplantation program with lenalidomide, bortezomib, and dexamethasone combination as induction and consolidation followed by lenalidomide maintenance in patients with multiple myeloma: a phase II study by the Intergroupe Francophone du Myelome. J Clin Oncol 2014; 32: 2712–2717.
Attal M, Lauwers-Cances V, Marit G, Caillot D, Moreau P, Facon T et al. Lenalidomide maintenance after stem-cell transplantation for multiple myeloma. N Engl J Med 2012; 366: 1782–1791.
McCarthy PL, Owzar K, Hofmeister CC, Hurd DD, Hassoun H, Richardson PG et al. Lenalidomide after stem-cell transplantation for multiple myeloma. N Engl J Med 2012; 366: 1770–1781.
Palumbo A, Hajek R, Delforge M, Kropff M, Petrucci MT, Catalano J et al. Continuous lenalidomide treatment for newly diagnosed multiple myeloma. N Engl J Med 2012; 366: 1759–1769.
Ito T, Ando H, Suzuki T, Ogura T, Hotta K, Imamura Y et al. Identification of a primary target of thalidomide teratogenicity. Science 2010; 327: 1345–1350.
Krönke J, Udeshi ND, Narla A, Grauman P, Hurst SN, McConkey M et al. Lenalidomide causes selective degradation of IKZF1 and IKZF3 in multiple myeloma cells. Science 2014; 343: 301–305.
Lu G, Middleton RE, Sun H, Naniong M, Ott CJ, Mitsiades CS et al. The myeloma drug lenalidomide promotes the cereblon-dependent destruction of Ikaros proteins. Science 2014; 343: 305–309.
Gandhi AK, Kang J, Havens CG, Conklin T, Ning Y, Wu L et al. Immunomodulatory agents lenalidomide and pomalidomide co-stimulate T cells by inducing degradation of T cell repressors Ikaros and Aiolos via modulation of the E3 ubiquitin ligase complex CRL4(CRBN.). Br J Haematol 2014; 164: 811–821.
Zhu YX, Braggio E, Shi CX, Kortuem KM, Bruins LA, Schmidt JE et al. Identification of cereblon-binding proteins and relationship with response and survival after IMiDs in multiple myeloma. Blood 2014; 124: 536–545.
Lopez-Girona A, Mendy D, Ito T, Miller K, Gandhi AK, Kang J et al. Cereblon is a direct protein target for immunomodulatory and antiproliferative activities of lenalidomide and pomalidomide. Leukemia 2012; 26: 2326–2335.
Zhu YX, Braggio E, Shi CX, Bruins LA, Schmidt JE, Van Wier S et al. Cereblon expression is required for the antimyeloma activity of lenalidomide and pomalidomide. Blood 2011; 118: 4771–4779.
Broyl A, Kuiper R, van Duin M, van der Holt B, el Jarari L, Bertsch U et al. High cereblon expression is associated with better survival in patients with newly diagnosed multiple myeloma treated with thalidomide maintenance. Blood 2013; 121: 624–627.
Heintel D, Rocci A, Ludwig H, Bolomsky A, Caltagirone S, Schreder M et al. High expression of cereblon (CRBN) is associated with improved clinical response in patients with multiple myeloma treated with lenalidomide and dexamethasone. Br J Haematol 2013; 161: 695–700.
Schuster SR, Kortuem KM, Zhu YX, Braggio E, Shi CX, Bruins LA et al. The clinical significance of cereblon expression in multiple myeloma. Leuk Res 2014; 38: 23–28.
Eichner R, Heider M, Fernandez-Saiz V, van Bebber F, Garz AK, Lemeer S et al. Immunomodulatory drugs disrupt the cereblon-CD147-MCT1 axis to exert antitumor activity and teratogenicity. Nat Med 2016; 22: 735–743.
Ross FM, Avet-Loiseau H, Ameye G, Gutierrez NC, Liebisch P, O'Connor S et al. Report from the European Myeloma Network on interphase FISH in multiple myeloma and related disorders. Haematologica 2012; 97: 1272–1277.
Durie BG, Harousseau JL, Miguel JS, Blade J, Barlogie B, Anderson K et al. International uniform response criteria for multiple myeloma. Leukemia 2006; 20: 1467–1473.
Sehgal K, Das R, Zhang L, Verma R, Deng Y, Kocoglu M et al. Clinical and pharmacodynamic analysis of pomalidomide dosing strategies in myeloma: impact of immune activation and cereblon targets. Blood 2015; 125: 4042–4051.
Gandhi AK, Mendy D, Waldman M, Chen G, Rychak E, Miller K et al. Measuring cereblon as a biomarker of response or resistance to lenalidomide and pomalidomide requires use of standardized reagents and understanding of gene complexity. Br J Haematol 2014; 164: 233–244.
Acknowledgements
This work was funded by the Deutsche Forschungsgemeinschaft (DFG; Emmy-Noether program Kr3886/2-1 to JK, KFO-216 to CL, RB, HE and LB). FK was supported in part by the Deutsche Krebshilfe (grant 109420, Max-Eder program). FB was supported by the European Research Commission (project BCM-UPS), the Deutsche Forschungsgemeinschaft (SFB 1243) and the Wilhelm Sander Stiftung (#2012.096.1). LB was supported in part by the Deutsche Forschungsgemeinschaft (Heisenberg-Professur BU 1339/8-1).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
Celgene cooperation provided lenalidomide as study medication used in this trial. JK received honoraria from Celgene. SK received honoraria from Celgene, Amgen, Bristol-Myers Squibb, Janssen, Onyx. CL received honoraria from Celgene, Amgen, Bristol-Myers Squibb, Janssen, Novartis, Takeda Oncology.
Additional information
Supplementary Information accompanies this paper on the Leukemia website
Supplementary information
Rights and permissions
About this article
Cite this article
Krönke, J., Kuchenbauer, F., Kull, M. et al. IKZF1 expression is a prognostic marker in newly diagnosed standard-risk multiple myeloma treated with lenalidomide and intensive chemotherapy: a study of the German Myeloma Study Group (DSMM). Leukemia 31, 1363–1367 (2017). https://doi.org/10.1038/leu.2016.384
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/leu.2016.384
This article is cited by
-
High levels of CRBN isoform lacking IMiDs binding domain predicts for a worse response to IMiDs-based upfront therapy in newly diagnosed myeloma patients
Clinical and Experimental Medicine (2023)
-
RNA sequencing identifies novel regulated IRE1-dependent decay targets that affect multiple myeloma survival and proliferation
Experimental Hematology & Oncology (2022)
-
HDAC inhibitor Vorinostat and BET inhibitor Plx51107 epigenetic agents’ combined treatments exert a therapeutic approach upon acute myeloid leukemia cell model
Medical Oncology (2022)
-
Cancer therapies based on targeted protein degradation — lessons learned with lenalidomide
Nature Reviews Clinical Oncology (2021)
-
Cereblon expression is a prognostic marker in newly diagnosed POEMS syndrome treated with lenalidomide plus dexamethasone
Annals of Hematology (2021)