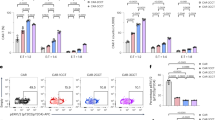
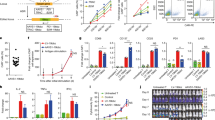
Abstract
Acute myeloid leukemia (AML) is an aggressive malignancy, and development of new treatments to prolong remissions is warranted. Chimeric antigen receptor (CAR) T-cell therapies appear promising but on-target, off-tumor recognition of antigen in healthy tissues remains a concern. Here we isolated a high-affinity (HA) folate receptor beta (FRβ)-specific single-chain variable fragment (2.48 nm KD) for optimization of FRβ-redirected CAR T-cell therapy for AML. T cells stably expressing the HA-FRβ CAR exhibited greatly enhanced antitumor activity against FRβ+ AML in vitro and in vivo compared with a low-affinity FRβ CAR (54.3 nm KD). Using the HA-FRβ immunoglobulin G, FRβ expression was detectable in myeloid-lineage hematopoietic cells; however, expression in CD34+ hematopoietic stem cells (HSCs) was nearly undetectable. Accordingly, HA-FRβ CAR T cells lysed mature CD14+ monocytes, while HSC colony formation was unaffected. Because of the potential for elimination of mature myeloid lineage, mRNA CAR electroporation for transient CAR expression was evaluated. mRNA-electroporated HA-FRβ CAR T cells retained effective antitumor activity in vitro and in vivo. Together, our results highlight the importance of antibody affinity in target protein detection and CAR development and suggest that transient delivery of potent HA-FRβ CAR T cells is highly effective against AML and reduces the risk for long-term myeloid toxicity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Howlader N, Noone AM, Krapcho M, Garshell J, Miller D, Altekruse SF, et al SEER Cancer Statistics Review (CSR) 1975–2012. National Cancer Institute: Bethesda, MD, USA, 2015, based on November 2014 SEER data submission, posted to the SEER web site http://seer.cancer.gov/csr/1975_2012.
Robak T, Wierzbowska A . Current and emerging therapies for acute myeloid leukemia. Clin Ther 2009; 31 (Pt 2): 2349–2370.
Kanate AS, Pasquini MC, Hari PN, Hamadani M . Allogeneic hematopoietic cell transplant for acute myeloid leukemia: current state in 2013 and future directions. World J Stem Cells 2014; 6: 69–81.
Jacobsohn DA, Vogelsang GB . Acute graft versus host disease. Orphanet J Rare Dis 2007; 2: 35.
Hahn T, McCarthy PL Jr, Zhang MJ, Wang D, Arora M, Frangoul H et al. Risk factors for acute graft-versus-host disease after human leukocyte antigen-identical sibling transplants for adults with leukemia. J Clin Oncol 2008; 26: 5728–5734.
Grupp SA, Kalos M, Barrett D, Aplenc R, Porter DL, Rheingold SR et al. Chimeric antigen receptor-modified T cells for acute lymphoid leukemia. N Engl J Med 2013; 368: 1509–1518.
Brentjens RJ, Davila ML, Riviere I, Park J, Wang X, Cowell LG et al. CD19-targeted T cells rapidly induce molecular remissions in adults with chemotherapy-refractory acute lymphoblastic leukemia. Sci Transl Med 2013; 5: 177ra138.
Lee DW, Kochenderfer JN, Stetler-Stevenson M, Cui YK, Delbrook C, Feldman SA et al. T cells expressing CD19 chimeric antigen receptors for acute lymphoblastic leukaemia in children and young adults: a phase 1 dose-escalation trial. Lancet 2015; 385: 517–528.
Gross G, Waks T, Eshhar Z . Expression of immunoglobulin-T-cell receptor chimeric molecules as functional receptors with antibody-type specificity. Proc Natl Acad Sci USA 1989; 86: 10024–10028.
Tettamanti S, Marin V, Pizzitola I, Magnani CF, Giordano Attianese GM, Cribioli E et al. Targeting of acute myeloid leukaemia by cytokine-induced killer cells redirected with a novel CD123-specific chimeric antigen receptor. Br J Haematol 2013; 161: 389–401.
Mardiros A, Dos Santos C, McDonald T, Brown CE, Wang X, Budde LE et al. T cells expressing CD123-specific chimeric antigen receptors exhibit specific cytolytic effector functions and antitumor effects against human acute myeloid leukemia. Blood 2013; 122: 3138–3148.
Gill S, Tasian SK, Ruella M, Shestova O, Li Y, Porter DL et al. Preclinical targeting of human acute myeloid leukemia and myeloablation using chimeric antigen receptor-modified T cells. Blood 2014; 123: 2343–2354.
Dutour A, Marin V, Pizzitola I, Valsesia-Wittmann S, Lee D, Yvon E et al. In vitro and in vivo antitumor effect of anti-CD33 chimeric receptor-expressing EBV-CTL against CD33 acute myeloid leukemia. Adv Hematol 2012; 2012: 683065.
Kenderian SS, Ruella M, Shestova O, Klichinsky M, Aikawa V, Morrissette JJ et al. CD33 specific chimeric antigen receptor T cells exhibit potent preclinical activity against human acute myeloid leukemia. Leukemia 2015; 29: 1637–1647.
Lynn RC, Poussin M, Kalota A, Feng Y, Low PS, Dimitrov DS et al. Targeting of folate receptor-beta on acute myeloid leukemia blasts with chimeric antigen receptor expressing T cells. Blood 2015; 125: 3466–3476.
Chmielewski M, Hombach A, Heuser C, Adams GP, Abken H . T cell activation by antibody-like immunoreceptors: increase in affinity of the single-chain fragment domain above threshold does not increase T cell activation against antigen-positive target cells but decreases selectivity. J Immunol 2004; 173: 7647–7653.
Hudecek M, Lupo-Stanghellini MT, Kosasih PL, Sommermeyer D, Jensen MC, Rader C et al. Receptor affinity and extracellular domain modifications affect tumor recognition by ROR1-specific chimeric antigen receptor T cells. Clin Cancer Res 2013; 19: 3153–3164.
Morgan RA, Yang JC, Kitano M, Dudley ME, Laurencot CM, Rosenberg SA . Case report of a serious adverse event following the administration of T cells transduced with a chimeric antigen receptor recognizing ERBB2. Mol Ther 2010; 18: 843–851.
Lamers CH, Sleijfer S, Vulto AG, Kruit WH, Kliffen M, Debets R et al. Treatment of metastatic renal cell carcinoma with autologous T-lymphocytes genetically retargeted against carbonic anhydrase IX: first clinical experience. J Clin Oncol 2006; 24: e20–e22.
Hinrichs CS, Restifo NP . Reassessing target antigens for adoptive T-cell therapy. Nat Biotechnol 2013; 31: 999–1008.
Feng Y, Zhu Z, Xiao X, Choudhry V, Barrett JC, Dimitrov DS . Novel human monoclonal antibodies to insulin-like growth factor (IGF)-II that potently inhibit the IGF receptor type I signal transduction function. Mol Cancer Ther 2006; 5: 114–120.
Lanitis E, Poussin M, Hagemann IS, Coukos G, Sandaltzopoulos R, Scholler N et al. Redirected antitumor activity of primary human lymphocytes transduced with a fully human anti-mesothelin chimeric receptor. Mol Ther 2012; 20: 633–643.
Godwin AK, Meister A, O'Dwyer PJ, Huang CS, Hamilton TC, Anderson ME . High resistance to cisplatin in human ovarian cancer cell lines is associated with marked increase of glutathione synthesis. Proc Natl Acad Sci USA 1992; 89: 3070–3074.
Schutsky K, Song DG, Lynn R, Smith JB, Poussin M, Figini M et al. Rigorous optimization and validation of potent RNA CAR T cell therapy for the treatment of common epithelial cancers expressing folate receptor. Oncotarget 2015; 6: 28911–28928.
Feng Y, Shen J, Streaker ED, Lockwood M, Zhu Z, Low PS et al. A folate receptor beta-specific human monoclonal antibody recognizes activated macrophage of rheumatoid patients and mediates antibody-dependent cell-mediated cytotoxicity. Arthritis Res Ther 2011; 13: R59.
Ross JF, Wang H, Behm FG, Mathew P, Wu M, Booth R et al. Folate receptor type beta is a neutrophilic lineage marker and is differentially expressed in myeloid leukemia. Cancer 1999; 85: 348–357.
Pan XQ . Strategy for the treatment of acute myelogenous leukemia based on folate receptor beta-targeted liposomal doxorubicin combined with receptor induction using all-trans retinoic acid. Blood 2002; 100: 594–602.
Betts MR, Brenchley JM, Price DA, De Rosa SC, Douek DC, Roederer M et al. Sensitive and viable identification of antigen-specific CD8+ T cells by a flow cytometric assay for degranulation. J Immunol Methods 2003; 281: 65–78.
Reddy JA, Haneline LS, Srour EF, Antony AC, Clapp DW, Low PS . Expression and functional characterization of the beta-isoform of the folate receptor on CD34(+) cells. Blood 1999; 93: 3940–3948.
Shen F, Ross JF, Wang X, Ratnam M . Identification of a novel folate receptor, a truncated receptor, and receptor type beta in hematopoietic cells: cDNA cloning, expression, immunoreactivity, and tissue specificity. Biochemistry 1994; 33: 1209–1215.
Nakashima-Matsushita N, Homma T, Yu S, Matsuda T, Sunahara N, Nakamura T et al. Selective expression of folate receptor beta and its possible role in methotrexate transport in synovial macrophages from patients with rheumatoid arthritis. Arthritis Rheum 1999; 42: 1609–1616.
Shen J, Hilgenbrink AR, Xia W, Feng Y, Dimitrov DS, Lockwood MB et al. Folate receptor-beta constitutes a marker for human proinflammatory monocytes. J Leukoc Biol 2014; 96: 563–570.
Song DG, Ye Q, Poussin M, Liu L, Figini M, Powell DJ Jr . A fully human chimeric antigen receptor with potent activity against cancer cells but reduced risk for off-tumor toxicity. Oncotarget 2015; 6: 21533–21546.
Hombach AA, Schildgen V, Heuser C, Finnern R, Gilham DE, Abken H . T cell activation by antibody-like immunoreceptors: the position of the binding epitope within the target molecule determines the efficiency of activation of redirected T cells. J Immunol 2007; 178: 4650–4657.
James SE, Greenberg PD, Jensen MC, Lin Y, Wang J, Till BG et al. Antigen sensitivity of CD22-specific chimeric TCR is modulated by target epitope distance from the cell membrane. J Immunol 2008; 180: 7028–7038.
Kowolik CM, Topp MS, Gonzalez S, Pfeiffer T, Olivares S, Gonzalez N et al. CD28 costimulation provided through a CD19-specific chimeric antigen receptor enhances in vivo persistence and antitumor efficacy of adoptively transferred T cells. Cancer Res 2006; 66: 10995–11004.
Savoldo B, Ramos CA, Liu E, Mims MP, Keating MJ, Carrum G et al. CD28 costimulation improves expansion and persistence of chimeric antigen receptor-modified T cells in lymphoma patients. J Clin Invest 2011; 121: 1822–1826.
Carpenito C, Milone MC, Hassan R, Simonet JC, Lakhal M, Suhoski MM et al. Control of large, established tumor xenografts with genetically retargeted human T cells containing CD28 and CD137 domains. Proc Natl Acad Sci USA 2009; 106: 3360–3365.
Song DG, Ye Q, Carpenito C, Poussin M, Wang LP, Ji C et al. In vivo persistence, tumor localization, and antitumor activity of CAR-engineered T cells is enhanced by costimulatory signaling through CD137 (4-1BB). Cancer Res 2011; 71: 4617–4627.
Milone MC, Fish JD, Carpenito C, Carroll RG, Binder GK, Teachey D et al. Chimeric receptors containing CD137 signal transduction domains mediate enhanced survival of T cells and increased antileukemic efficacy in vivo. Mol Ther 2009; 17: 1453–1464.
Song DG, Ye Q, Poussin M, Harms GM, Figini M, Powell DJ Jr . CD27 costimulation augments the survival and antitumor activity of redirected human T cells in vivo. Blood 2012; 119: 696–706.
Maus MV, Haas AR, Beatty GL, Albelda SM, Levine BL, Liu X et al. T cells expressing chimeric antigen receptors can cause anaphylaxis in humans. Cancer Immunol Res 2013; 1: 26–31.
Acknowledgements
This work was supported by grants from the NIH (RO1-CA168900; to DJP) and (T32-AI070099; to RCL) and by the Intramural Research Program of the Center for Cancer Research, National Cancer Institute, National Institutes of Health (to DSD). We thank Gwenn Danet-Desnoyers, Tony Secreto, Josh Glover and Winifred Trotman at the SCXC for their assistance with leukemia and BM acquisition and xenograft services. Bioluminescent imaging was supported by the University of Pennsylvania small animal imaging core facility.
Author contributions
RCL and DJP designed experiments and wrote the paper. RCL conducted experiments, analyzed data and prepared the figures. YF and DSD isolated the scFvs and prepared and conducted experiments with soluble FRβ scFvs and IgGs. KS prepared mRNA CAR reagents. MP conducted in vivo imaging. AK scored the CFU. All authors have read and approved the submitted manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Leukemia website
Supplementary information
Rights and permissions
About this article
Cite this article
Lynn, R., Feng, Y., Schutsky, K. et al. High-affinity FRβ-specific CAR T cells eradicate AML and normal myeloid lineage without HSC toxicity. Leukemia 30, 1355–1364 (2016). https://doi.org/10.1038/leu.2016.35
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/leu.2016.35
This article is cited by
-
Preclinical development and evaluation of nanobody-based CD70-specific CAR T cells for the treatment of acute myeloid leukemia
Cancer Immunology, Immunotherapy (2023)
-
CAR-T cell-mediated depletion of immunosuppressive tumor-associated macrophages promotes endogenous antitumor immunity and augments adoptive immunotherapy
Nature Communications (2021)
-
The folate receptor β as a macrophage-mediated imaging and therapeutic target in rheumatoid arthritis
Drug Delivery and Translational Research (2019)
-
Putative cancer stem cells may be the key target to inhibit cancer cell repopulation between the intervals of chemoradiation in murine mesothelioma
BMC Cancer (2018)
-
Targeting FLT3 in acute myeloid leukemia using ligand-based chimeric antigen receptor-engineered T cells
Journal of Hematology & Oncology (2018)