Abstract
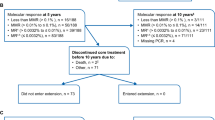
Most clinical trials exclude patients with poor performance or comorbidities. To study whether patients with these characteristics can be treated within a clinical trial, we conducted a study for patients with acute myeloid leukemia (AML) or myelodysplastic syndromes (MDS) with poor performance, organ dysfunction or comorbidities. Primary endpoint was 60-day survival. Study included stopping rules for survival and response. Treatment consisted on a combination of azacitidine and vorinostat. Thirty patients (16 with MDS, 14 with AML) were enrolled. Median follow-up was 7.4 months (0.3–29). Sixty-day survival was 83%. No stopping rules were met. Main adverse events (AEs) were grades 1 and 2 gastrointestinal toxicities. In view of these results, we expanded the study and treated 79 additional patients: 27 with azacitidine (AZA) and 52 with azacitidine and vorinostat (AZA+V). Median follow-up was 22.7 months (12.6–47.5). Sixty-day survival rate was 79% (AZA=67%, AZA+V=85%, P=0.07). Median overall survival was 7.6 months (4.5–10.7). Median event-free survival was 4.5 months (3.5–5.6). Main AEs included grades 1 and 2 gastrointestinal toxicities. Our results suggest this subset of patients can be safely treated within clinical trials and derive clinical benefit. Relaxation of standard exclusion criteria may increase the pool of patients likely to benefit from therapy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Yao B, Zhu L, Jiang Q, Xia HA . Safety monitoring in clinical trials. Pharmaceutics 2013; 5: 94–106.
Yanada M, Okamoto A, Inaguma Y, Tokuda M, Morishima S, Kanie T et al. The fate of patients with acute myeloid leukemia not undergoing induction chemotherapy. Int J Hematol 2015; 102: 35–40.
Bell JA, Balneaves LG . Cancer patient decision making related to clinical trial participation: an integrative review with implications for patients’ relational autonomy. Support Care Cancer 2015; 23: 1169–1196.
Mengis C, Aebi S, Tobler A, Dahler W, Fey MF . Assessment of differences in patient populations selected for excluded from participation in clinical phase III acute myelogenous leukemia trials. J Clin Oncol 2003; 21: 3933–3939.
Kantarjian H, O’Brien S, Cortes J, Giles F, Faderl S, Jabbour E et al. Results of intensive chemotherapy in 998 patients age 65 years or older with acute myeloid leukemia or high-risk myelodysplastic syndrome: predictive prognostic models for outcome. Cancer 2006; 106: 1090–1098.
Kantarjian H, Ravandi F, O’Brien S, Cortes J, Faderl S, Garcia-Manero G et al. Intensive chemotherapy does not benefit most older patients (age 70 years or older) with acute myeloid leukemia. Blood 2010; 116: 4422–4429.
Menzin J, Lang K, Earle CC, Kerney D, Mallick R . The outcomes and costs of acute myeloid leukemia among the elderly. Arch Intern Med 2002; 162: 1597–1603.
Sevinc A, Buyukberber S, Camci C, Koruk M, Savas MC, Turk HM et al. Granulocytic sarcoma of the colon and leukemic infiltration of the liver in a patient presenting with hematochezia and jaundice. Digestion 2004; 69: 262–265.
Wandroo FA, Murray J, Mutimer D, Hubscher S . Acute myeloid leukaemia presenting as cholestatic hepatitis. J Clin Pathol 2004; 57: 544–545.
Ruger W, Kruip MJ, Betjes MG . Reversible renal failure due to bilateral renal sarcoma in a patient with acute myeloid leukemia. Ren Fail 2009; 31: 606–609.
Fenaux P, Mufti GJ, Hellstrom-Lindberg E, Santini V, Finelli C, Giagounidis A et al. Efficacy of azacitidine compared with that of conventional care regimens in the treatment of higher-risk myelodysplastic syndromes: a randomised, open-label, phase III study. Lancet Oncol 2009; 10: 223–232.
Lee YG, Kim I, Yoon SS, Park S, Cheong JW, Min YH et al. Comparative analysis between azacitidine and decitabine for the treatment of myelodysplastic syndromes. Br J Haematol 2013; 161: 339–347.
Thepot S, Itzykson R, Seegers V, Recher C, Raffoux E, Quesnel B et al. Azacitidine in untreated acute myeloid leukemia: a report on 149 patients. Am J Hematol 2014; 89: 410–416.
Silva G, Cardoso BA, Belo H, Almeida AM . Vorinostat induces apoptosis and differentiation in myeloid malignancies: genetic and molecular mechanisms. PLoS One 2013; 8: e53766.
Schaefer EW, Loaiza-Bonilla A, Juckett M, DiPersio JF, Roy V, Slack J et al. A phase 2 study of vorinostat in acute myeloid leukemia. Haematologica 2009; 94: 1375–1382.
Verma A, Odchimar-Reissig R, Feldman EJ, Navada SC, Demakos EP, Baer MR et al. A phase II trial of epigenetic modulators vorinostat in combination with azacitidine (azaC) in patients with the Myelodysplastic Syndrome (MDS): initial results of study 6898 of the New York Cancer Consortium. Blood 2013; 122: 386.
Cameron EE, Bachman KE, Myohanen S, Herman JG, Baylin SB . Synergy of demethylation and histone deacetylase inhibition in the re-expression of genes silenced in cancer. Nat Genet 1999; 21: 103–107.
Esteller M . CpG island methylation and histone modifications: biology and clinical significance. Ernst Schering Res Found Workshop 2006; (57: 115–126.
Yang H, Hoshino K, Sanchez-Gonzalez B, Kantarjian H, Garcia-Manero G . Antileukemia activity of the combination of 5-aza-2′-deoxycytidine with valproic acid. Leuk Res 2005; 29: 739–748.
Greenberg P, Cox C, LeBeau MM, Fenaux P, Morel P, Sanz G et al. International scoring system for evaluating prognosis in myelodysplastic syndromes. Blood 1997; 89: 2079–2088.
Arber DA, Orazi A, Hasserjian R, Thiele J, Borowitz MJ, Le Beau MM et al. The 2016 revision to the World Health Organization (WHO) classification of myeloid neoplasms and acute leukemia. Blood 2016; 127: 2391–2405.
Lo-Coco F, Avvisati G, Vignetti M, Thiede C, Orlando SM, Iacobelli S et al. Retinoic acid and arsenic trioxide for acute promyelocytic leukemia. N Engl J Med 2013; 369: 111–121.
Prebet T, Boissel N, Reutenauer S, Thomas X, Delaunay J, Cahn JY et al. Acute myeloid leukemia with translocation (8;21) or inversion (16) in elderly patients treated with conventional chemotherapy: a collaborative study of the French CBF-AML intergroup. J Clin Oncol 2009; 27: 4747–4753.
Thall PF, Simon RM, Estey EH . Bayesian sequential monitoring designs for single-arm clinical trials with multiple outcomes. Stat Med 1995; 14: 357–379.
Cheson BD, Bennett JM, Kopecky KJ, Buchner T, Willman CL, Estey EH et al. Revised recommendations of the International Working Group for Diagnosis, Standardization of Response Criteria, Treatment Outcomes, and Reporting Standards for Therapeutic Trials in Acute Myeloid Leukemia. J Clin Oncol 2003; 21: 4642–4649.
Cheson BD, Bennett JM, Kantarjian H, Pinto A, Schiffer CA, Nimer SD et al. Report of an international working group to standardize response criteria for myelodysplastic syndromes. Blood 2000; 96: 3671–3674.
Daver N, Naqvi K, Jabbour E, Kadia T, DiNardo C, Cardenas-Turanzas M et al. Impact of comorbidities by ACE-27 in the revised-IPSS for patients with myelodysplastic syndromes. Am J Hematol 2014; 89: 509–516.
Naqvi K, Garcia-Manero G, Sardesai S, Oh J, Vigil CE, Pierce S et al. Association of comorbidities with overall survival in myelodysplastic syndrome: development of a prognostic model. J Clin Oncol 2011; 29: 2240–2246.
Soriano AO, Yang H, Faderl S, Estrov Z, Giles F, Ravandi F et al. Safety and clinical activity of the combination of 5-azacytidine, valproic acid, and all-trans retinoic acid in acute myeloid leukemia and myelodysplastic syndrome. Blood 2007; 110: 2302–2308.
Tapper EB, Luptakova K, Joyce RM, Tzachanis D . A 78-year-old man with acute myeloid leukemia (AML) and acute renal failure. Am J Case Rep 2014; 15: 364–367.
Bhatnagar B, Duong VH, Gourdin TS, Tidwell ML, Chen C, Ning Y et al. Ten-day decitabine as initial therapy for newly diagnosed patients with acute myeloid leukemia unfit for intensive chemotherapy. Leuk Lymphoma 2014; 55: 1533–1537.
Medeiros BC, Satram-Hoang S, Hurst D, Hoang KQ, Momin F, Reyes C . Big data analysis of treatment patterns and outcomes among elderly acute myeloid leukemia patients in the United States. Ann Hematol 2015; 94: 1127–1138.
Oran B, Weisdorf DJ . Survival for older patients with acute myeloid leukemia: a population-based study. Haematologica 2012; 97: 1916–1924.
Ostgard LS, Norgaard JM, Sengelov H, Severinsen M, Friis LS, Marcher CW et al. Comorbidity and performance status in acute myeloid leukemia patients: a nation-wide population-based cohort study. Leukemia 2015; 29: 548–555.
Acknowledgements
Merck Sharp and Dohme Corporation supported this clinical trial (NCT00948064). Support for this project was also provided by a number of other sources, including the MD Anderson Cancer Center Support Grant P30 CA016672, the Dr Kenneth B McCredie Chair in Clinical Leukemia Research endowment, the Edward P Evans Foundation, the Fundacion Ramon Areces, the Cancer Prevention & Research Institute of Texas (CPRIT) award RP140500, and by generous philanthropic contributions to MD Anderson’s MDS/AML Moon Shot Program.
Author contributions
G Garcia-Manero, E Estey, C DiNardo, N Pemmaraju, N Daver, Z Estrov, J Cortes, Y Alvarado, S Verstovsek, N Jain, W Wierda, M Konopleva, E Jabbour, G Borthakur, T Kadia, F Ravandi and H Kantarjian designed the study, analyzed data, contributed patients and participated in writing the manuscript. X Huang and C Hsiang-Chun performed the statistical analysis and participated in statistical design. G Montalban-Bravo participated in data collection, analyzing the data and writing the manuscript. M Brant and S Pierce collected and analyzed data. C Bueso-Ramos performed histopathological analysis. H Yang performed correlative studies. C Foudray and T Sneed participated as the research nurses of the study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
GG-M work has been supported by Celgene, maker of azacitidine, and Merck, maker of vorinostat. Merck supported in part this study. GG-M has served as a consultant and speaker for both Celgene and Merck.
Additional information
Supplementary Information accompanies this paper on the Leukemia website
Supplementary information
Rights and permissions
About this article
Cite this article
Montalban-Bravo, G., Huang, X., Jabbour, E. et al. A clinical trial for patients with acute myeloid leukemia or myelodysplastic syndromes not eligible for standard clinical trials. Leukemia 31, 318–324 (2017). https://doi.org/10.1038/leu.2016.303
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/leu.2016.303
This article is cited by
-
Effect of ECOG performance status on outcomes in patients with acute myeloid leukemia and other high-grade myeloid neoplasms
Leukemia (2023)
-
Age and sex associate with outcome in older AML and high risk MDS patients treated with 10-day decitabine
Blood Cancer Journal (2023)
-
A phase 1b multicenter study of TAS-102 in combination with irinotecan in patients with advanced recurrent or unresectable gastric and gastroesophageal adenocarcinoma after at least one line of treatment with a fluoropyrimidine and platinum-containing regimen
Medical Oncology (2022)
-
Characteristics and outcomes of therapy-related myeloid neoplasms after peptide receptor radionuclide/chemoradionuclide therapy (PRRT/PRCRT) for metastatic neuroendocrine neoplasia: a single-institution series
European Journal of Nuclear Medicine and Molecular Imaging (2019)
-
New drugs in AML: uses and abuses
Leukemia (2018)