Abstract
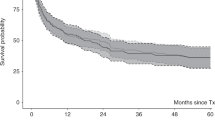
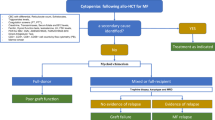
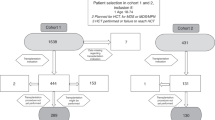
Clinical outcomes after primary graft failure (PGF) remain poor. Here we present a large retrospective analysis (n=23 272) which investigates means to prevent PGF and early detection of patients at high risk. In patients with hematologic malignancies, who underwent their first myeloablative allogeneic hematopoietic cell transplantation, PGF was reported in 1278 (5.5%), and there was a marked difference in PGFs using peripheral blood stem cell compared with bone marrow grafts (2.5 vs 7.3%; P<0.001). A fourfold increase of PGF was observed in myeloproliferative disorders compared with acute leukemia (P<0.001). Other risk factors for PGF included recipient age <30, HLA mismatch, male recipients of female donor grafts, ABO incompatibility, busulfan/cyclophosphamide conditioning and cryopreservation. In bone marrow transplants, total nucleated cell doses ⩽2.4 × 108 per kg were associated with PGF (odds ratio 1.39; P<0.001). The use of tacrolimus-based immunosuppression and granulocyte colony-stimulating factor were associated with decreased PGF risk. These data, allow clinicians to do more informed choices with respect to graft source, donor selection, conditioning and immunosuppressive regimens to reduce the risk of PGF. Moreover, a novel risk score determined on day 21 post transplant may provide the rationale for an early request for additional hematopoietic stem cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Jonson FL, Hartmann JR, Thomas ED, Chard RL, Hersman JA, Buckner CD et al. Marrow transplantation in treatment of children with aplastic anaemia or acute leukaemia. Arch Dis Child 1976; 51: 403–410.
Storb R, Thomas ED, Weiden PL, Buckner CD, Clift RA, Fefer A et al. Aplastic anemia treated by allogeneic bone marrow transplantation: a report on 49 new cases from Seattle. Blood 1976; 48: 817–841.
Remberger M, Mattsson J, Olsson R, Ringden O . Second allogeneic hematopoietic stem cell transplantation: a treatment for graft failure. Clin Transplant 2011; 25: E68–E76.
Schriber J, Agovi MA, Ho V, Ballen KK, Bacigalupo A, Lazarus HM et al. Second unrelated donor hematopoietic cell transplantation for primary graft failure. Biol Blood Marrow Transplant 2010; 16: 1099–1106.
Olsson R, Remberger M, Schaffer M, Berggren DM, Svahn BM, Mattsson J et al. Graft failure in the modern era of allogeneic hematopoietic SCT. Bone Marrow Transplant 2013; 48: 537–543.
Mattsson J, Ringden O, Storb R . Graft failure after allogeneic hematopoietic cell transplantation. Biol Blood Marrow Transplant 2008; 14: 165–170.
Crocchiolo R, Ciceri F, Fleischhauer K, Oneto R, Bruno B, Pollichieni S et al. HLA matching affects clinical outcome of adult patients undergoing haematopoietic SCT from unrelated donors: a study from the Gruppo Italiano Trapianto di Midollo Osseo and Italian Bone Marrow Donor Registry. Bone Marrow Transplant 2009; 44: 571–577.
Remberger M, Watz E, Ringden O, Mattsson J, Shanwell A, Wikman A . Major ABO blood group mismatch increases the risk for graft failure after unrelated donor hematopoietic stem cell transplantation. Biol Blood Marrow Transplant 2007; 13: 675–682.
Cudkowicz G, Bennett M . Peculiar immunobiology of bone marrow allografts. I. Graft rejection by irradiated responder mice. J Exp Med 1971; 134: 83–102.
Kiessling R, Hochman PS, Haller O, Shearer GM, Wigzell H, Cudkowicz G . Evidence for a similar or common mechanism for natural killer cell activity and resistance to hemopoietic grafts. Eur J Immunol 1977; 7: 655–663.
Murphy WJ, Kumar V, Bennett M . Acute rejection of murine bone marrow allografts by natural killer cells and T cells. Differences in kinetics and target antigens recognized. J Exp Med 1987; 166: 1499–1509.
Raff RF, Deeg HJ, Loughran Jr TP, Graham TC, Aprile JA, Sale GE et al. Characterization of host cells involved in resistance to marrow grafts in dogs transplanted from unrelated DLA-nonidentical donors. Blood 1986; 68: 861–868.
Raff RF, Sandmaier BM, Graham T, Loughran Jr TP, Pettinger M, Storb R . 'Resistance' to unrelated, DLA-nonidentical canine marrow grafts is unrestricted by the major histocompatibility complex. Exp Hematol 1994; 22: 893–897.
Barge AJ, Johnson G, Witherspoon R, Torok-Storb B . Antibody-mediated marrow failure after allogeneic bone marrow transplantation. Blood 1989; 74: 1477–1480.
Moeller E . Contact-induced cytotoxicity by lymphoid cells containing foreign isoantigens. Science 1965; 147: 873–879.
Storb R, Floersheim GL, Weiden PL, Graham TC, Kolb HJ, Lerner KG et al. Effect of prior blood transfusions on marrow grafts: abrogation of sensitization by procarbazine and antithymocyte serum. J Immunol 1974; 112: 1508–1516.
Taylor PA, Ehrhardt MJ, Roforth MM, Swedin JM, Panoskaltsis-Mortari A, Serody JS et al. Preformed antibody, not primed T cells, is the initial and major barrier to bone marrow engraftment in allosensitized recipients. Blood 2007; 109: 1307–1315.
Warren RP, Storb R, Weiden PL, Su PJ, Thomas ED . Lymphocyte-mediated cytotoxicity and antibody-dependent cell-mediated cytotoxicity in patients with aplastic anemia: distinguishing transfusion-induced sensitization from possible immune-mediated aplastic anemia. Transplant Proc 1981; 13: 245–247.
Nordlander A, Mattsson J, Sundberg B, Sumitran-Holgersson S . Novel antibodies to the donor stem cell population CD34+/VEGFR-2+ are associated with rejection after hematopoietic stem cell transplantation. Transplantation 2008; 86: 686–696.
Cutler C, Kim HT, Sun L, Sese D, Glotzbecker B, Armand P et al. Donor-specific anti-HLA antibodies predict outcome in double umbilical cord blood transplantation. Blood 2011; 118: 6691–6697.
Bacigalupo A, Ballen K, Rizzo D, Giralt S, Lazarus H, Ho V et al. Defining the intensity of conditioning regimens: working definitions. Biol Blood Marrow Transplant 2009; 15: 1628–1633.
Weisdorf D, Spellman S, Haagenson M, Horowitz M, Lee S, Anasetti C et al. Classification of HLA-matching for retrospective analysis of unrelated donor transplantation: revised definitions to predict survival. Biol Blood Marrow Transplant 2008; 14: 748–758.
Korbling M, Anderlini P . Peripheral blood stem cell versus bone marrow allotransplantation: does the source of hematopoietic stem cells matter? Blood 2001; 98: 2900–2908.
Bortin MM, Gale RP, Kay HE, Rimm AA . Bone marrow transplantation for acute myelogenous leukemia. Factors associated with early mortality. JAMA 1983; 249: 1166–1175.
Ringden O, Barrett AJ, Zhang MJ, Loberiza FR, Bolwell BJ, Cairo MS et al. Decreased treatment failure in recipients of HLA-identical bone marrow or peripheral blood stem cell transplants with high CD34 cell doses. Br J Haematol 2003; 121: 874–885.
Ringden O, Nilsson B . Death by graft-versus-host disease associated with HLA mismatch, high recipient age, low marrow cell dose, and splenectomy. Transplantation 1985; 40: 39–44.
Sierra J, Storer B, Hansen JA, Bjerke JW, Martin PJ, Petersdorf EW et al. Transplantation of marrow cells from unrelated donors for treatment of high-risk acute leukemia: the effect of leukemic burden, donor HLA-matching, and marrow cell dose. Blood 1997; 89: 4226–4235.
Storb R, Prentice RL, Thomas ED . Marrow transplantation for treatment of aplastic anemia. An analysis of factors associated with graft rejection. N Engl J Med 1977; 296: 61–66.
Tollemar J, Ringden O, Bostrom L, Nilsson B, Sundberg B . Variables predicting deep fungal infections in bone marrow transplant recipients. Bone Marrow Transplant 1989; 4: 635–641.
Zaucha JM, Gooley T, Bensinger WI, Heimfeld S, Chauncey TR, Zaucha R et al. CD34 cell dose in granulocyte colony-stimulating factor-mobilized peripheral blood mononuclear cell grafts affects engraftment kinetics and development of extensive chronic graft-versus-host disease after human leukocyte antigen-identical sibling transplantation. Blood 2001; 98: 3221–3227.
Nakamura R, Auayporn N, Smith DD, Palmer J, Sun JY, Schriber J et al. Impact of graft cell dose on transplant outcomes following unrelated donor allogeneic peripheral blood stem cell transplantation: higher CD34+ cell doses are associated with decreased relapse rates. Biol Blood Marrow Transplant 2008; 14: 449–457.
Nakamae H, Storer B, Sandmaier BM, Maloney DG, Davis C, Corey L et al. Cytopenias after day 28 in allogeneic hematopoietic cell transplantation: impact of recipient/donor factors, transplant conditions and myelotoxic drugs. Haematologica 2011; 96: 1838–1845.
Delgado J, Thomson K, Russell N, Ewing J, Stewart W, Cook G et al. Results of alemtuzumab-based reduced-intensity allogeneic transplantation for chronic lymphocytic leukemia: a British Society of Blood and Marrow Transplantation Study. Blood 2006; 107: 1724–1730.
Abelsson J, Merup M, Birgegard G, WeisBjerrum O, Brinch L, Brune M et al. The outcome of allo-HSCT for 92 patients with myelofibrosis in the Nordic countries. Bone Marrow Transplant 2012; 47: 380–386.
Ballen KK, Woolfrey AE, Zhu X, Ahn KW, Wirk B, Arora M et al. Allogeneic hematopoietic cell transplantation for advanced polycythemia vera and essential thrombocythemia. Biol Blood Marrow Transplant 2012; 18: 1446–1454.
Akpek G, Pasquini MC, Logan B, Agovi MA, Lazarus HM, Marks DI et al. Effects of spleen status on early outcomes after hematopoietic cell transplantation. Bone Marrow Transplant 2013; 48: 825–831.
Champlin RE, Horowitz MM, van Bekkum DW, Camitta BM, Elfenbein GE, Gale RP et al. Graft failure following bone marrow transplantation for severe aplastic anemia: risk factors and treatment results. Blood 1989; 73: 606–613.
Gupta V, Hari P, Hoffman R . Allogeneic hematopoietic cell transplantation for myelofibrosis in the era of JAK inhibitors. Blood 2012; 120: 1367–1379.
Kang HJ, Shin HY, Park JE, Chung NG, Cho B, Kim HK et al. Successful engraftment with fludarabine, cyclophosphamide, and thymoglobulin conditioning regimen in unrelated transplantation for severe aplastic anemia: A phase II prospective multicenter study. Biol Blood Marrow Transplant 2010; 16: 1582–1588.
Petersdorf EW, Gooley TA, Anasetti C, Martin PJ, Smith AG, Mickelson EM et al. Optimizing outcome after unrelated marrow transplantation by comprehensive matching of HLA class I and II alleles in the donor and recipient. Blood 1998; 92: 3515–3520.
Fleischhauer K, Locatelli F, Zecca M, Orofino MG, Giardini C, De Stefano P et al. Graft rejection after unrelated donor hematopoietic stem cell transplantation for thalassemia is associated with nonpermissive HLA-DPB1 disparity in host-versus-graft direction. Blood 2006; 107: 2984–2992.
Spellman S, Bray R, Rosen-Bronson S, Haagenson M, Klein J, Flesch S et al. The detection of donor-directed, HLA-specific alloantibodies in recipients of unrelated hematopoietic cell transplantation is predictive of graft failure. Blood 2010; 115: 2704–2708.
Morishima Y, Sasazuki T, Inoko H, Juji T, Akaza T, Yamamoto K et al. The clinical significance of human leukocyte antigen (HLA) allele compatibility in patients receiving a marrow transplant from serologically HLA-A, HLA-B, and HLA-DR matched unrelated donors. Blood 2002; 99: 4200–4206.
Petersdorf EW, Hansen JA, Martin PJ, Woolfrey A, Malkki M, Gooley T et al. Major-histocompatibility-complex class I alleles and antigens in hematopoietic-cell transplantation. N Engl J Med 2001; 345: 1794–1800.
Daniel-Johnson J, Schwartz J . How do I approach ABO-incompatible hematopoietic progenitor cell transplantation? Transfusion 2011; 51: 1143–1149.
Kelton JG, Bebenek G . Granulocytes do not have surface ABO antigens. Transfusion 1985; 25: 567–569.
Davies SM, Kollman C, Anasetti C, Antin JH, Gajewski J, Casper JT et al. Engraftment and survival after unrelated-donor bone marrow transplantation: a report from the national marrow donor program. Blood 2000; 96: 4096–4102.
Nash RA, Pepe MS, Storb R, Longton G, Pettinger M, Anasetti C et al. Acute graft-versus-host disease: analysis of risk factors after allogeneic marrow transplantation and prophylaxis with cyclosporine and methotrexate. Blood 1992; 80: 1838–1845.
Saliba RM, de Lima M, Giralt S, Andersson B, Khouri IF, Hosing C et al. Hyperacute GVHD: risk factors, outcomes, and clinical implications. Blood 2007; 109: 2751–2758.
Rufer N, Starobinski M, Chapuis B, Gratwohl A, Jeannet M, Helg C et al. Clinical consequences of sensitisation to minor histocompatibility antigens before allogeneic bone marrow transplantation. Bone Marrow Transplant 1998; 22: 895–898.
Stern M, Passweg JR, Locasciulli A, Socie G, Schrezenmeier H, Bekassy AN et al. Influence of donor/recipient sex matching on outcome of allogeneic hematopoietic stem cell transplantation for aplastic anemia. Transplantation 2006; 82: 218–226.
Marmont AM, Horowitz MM, Gale RP, Sobocinski K, Ash RC, van Bekkum DW et al. T-cell depletion of HLA-identical transplants in leukemia. Blood 1991; 78: 2120–2130.
Soderling CC, Song CW, Blazar BR, Vallera DA . A correlation between conditioning and engraftment in recipients of MHC-mismatched T cell-depleted murine bone marrow transplants. J Immunol 1985; 135: 941–946.
Seidel MG, Fritsch G, Matthes-Martin S, Lawitschka A, Lion T, Potschger U et al. In vitro and in vivo T-cell depletion with myeloablative or reduced-intensity conditioning in pediatric hematopoietic stem cell transplantation. Haematologica 2005; 90: 1405–1414.
Devine SM, Carter S, Soiffer RJ, Pasquini MC, Hari PN, Stein A et al. Low risk of chronic graft-versus-host disease and relapse associated with T cell-depleted peripheral blood stem cell transplantation for acute myelogenous leukemia in first remission: results of the blood and marrow transplant clinical trials network protocol 0303. Biol Blood Marrow Transplant 2011; 17: 1343–1351.
Small TN, Avigan D, Dupont B, Smith K, Black P, Heller G et al. Immune reconstitution following T-cell depleted bone marrow transplantation: effect of age and posttransplant graft rejection prophylaxis. Biol Blood Marrow Transplant 1997; 3: 65–75.
Meijer E, Cornelissen JJ, Lowenberg B, Verdonck LF . Antithymocyteglobulin as prophylaxis of graft failure and graft-versus-host disease in recipients of partially T-cell-depleted grafts from matched unrelated donors: a dose-finding study. Exp Hematol 2003; 31: 1026–1030.
Fernyhough LJ, Buchan VA, McArthur LT, Hock BD . Relative recovery of haematopoietic stem cell products after cryogenic storage of up to 19 years. Bone Marrow Transplant 2013; 48: 32–35.
Khoury HJ, Loberiza Jr FR, Ringden O, Barrett AJ, Bolwell BJ, Cahn JY et al. Impact of posttransplantation G-CSF on outcomes of allogeneic hematopoietic stem cell transplantation. Blood 2006; 107: 1712–1716.
Acknowledgements
RFO thanks the Swedish Society for Medical Research (SSMF), the Swedish Medical Society, the Centre for Clinical Research Sörmland, the David and Astrid Hagelén’s Foundation and the Karolinska Institutet for their financial support. The CIBMTR is supported by Public Health Service Grant/Cooperative Agreement U24-CA076518 from the National Cancer Institute (NCI), the National Heart, Lung and Blood Institute (NHLBI) and the National Institute of Allergy and Infectious Diseases (NIAID); a Grant/Cooperative Agreement 5U10HL069294 from NHLBI and NCI; a contract HHSH250201200016C with Health Resources and Services Administration (HRSA/DHHS); two Grants N00014-12-1-0142 and N00014-13-1-0039 from the Office of Naval Research; and grants from Actinium Pharmaceuticals; Allos Therapeutics, Inc.; Amgen, Inc.; Anonymous donation to the Medical College of Wisconsin; Ariad; Be the Match Foundation; Blue Cross and Blue Shield Association; Celgene Corporation; Chimerix, Inc.; Fred Hutchinson Cancer Research Center; Fresenius-Biotech North America, Inc.; Gamida Cell Teva Joint Venture Ltd.; Genentech, Inc.; Gentium SpA; Genzyme Corporation; GlaxoSmithKline; Health Research, Inc. Roswell Park Cancer Institute; HistoGenetics, Inc.; Incyte Corporation; Jeff Gordon Children’s Foundation; Kiadis Pharma; The Leukemia & Lymphoma Society; Medac GmbH; The Medical College of Wisconsin; Merck & Co, Inc.; Millennium: The Takeda Oncology Co.; Milliman USA, Inc.; Miltenyi Biotec, Inc.; National Marrow Donor Program; Onyx Pharmaceuticals; Optum Healthcare Solutions, Inc.; Osiris Therapeutics, Inc.; Otsuka America Pharmaceutical, Inc.; Perkin Elmer, Inc.; Remedy Informatics; Sanofi US; Seattle Genetics; Sigma-Tau Pharmaceuticals; Soligenix, Inc.; St. Baldrick’s Foundation; StemCyte, A Global Cord Blood Therapeutics Co.; Stemsoft Software, Inc.; Swedish Orphan Biovitrum; Tarix Pharmaceuticals; TerumoBCT; Teva Neuroscience, Inc.; THERAKOS, Inc.; University of Minnesota; University of Utah; and Wellpoint, Inc.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Disclaimer
The views expressed in this article do not reflect the official policy or position of the National Institute of Health, the Department of the Navy, the Department of Defense, Health Resources and Services Administration (HRSA) or any other agency of the U.S. Government
Supplementary Information accompanies this paper on the Leukemia website
Supplementary information
Rights and permissions
About this article
Cite this article
Olsson, R., Logan, B., Chaudhury, S. et al. Primary graft failure after myeloablative allogeneic hematopoietic cell transplantation for hematologic malignancies. Leukemia 29, 1754–1762 (2015). https://doi.org/10.1038/leu.2015.75
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/leu.2015.75
This article is cited by
-
Pretransplant hepatomegaly is linked to relapse in patients with leukemia and myelodysplastic syndrome not in remission
International Journal of Hematology (2024)
-
Salvage haploidentical transplantation for graft failure after first haploidentical allogeneic stem cell transplantation: an updated experience
Bone Marrow Transplantation (2024)
-
Salvage single-unit unrelated cord blood transplantation for graft failure following initial allogeneic transplantation in adult acute myeloid leukemia: trends in outcomes over the past 20 years
Bone Marrow Transplantation (2022)
-
Haemophagocytic lymphohistiocytosis following allogeneic hematopoietic cell transplantation from mismatched unrelated donors associated with low CD34 and CD3 cell counts in the graft
Bone Marrow Transplantation (2022)
-
Non-relapse cytopenias following allogeneic stem cell transplantation, a case based review
Bone Marrow Transplantation (2022)