Abstract
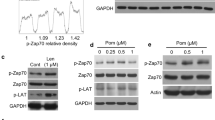
The anti-CD38 monoclonal antibody SAR650984 (SAR) is showing promising clinical activity in treatment of relapsed and refractory multiple myeloma (MM). Besides effector-mediated antibody-dependent cellular cytotoxicity and complement-mediated cytotoxicity, we here define molecular mechanisms of SAR-directed MM cell death and enhanced anti-MM activity triggered by SAR with Pomalidomide (Pom). Without Fc-cross-linking agents or effector cells, SAR specifically induces homotypic aggregation (HA)-associated cell death in MM cells dependent on the level of cell surface CD38 expression, actin cytoskeleton and membrane lipid raft. SAR and its F(ab)’2 fragments trigger caspase 3/7-dependent apoptosis in MM cells highly expressing CD38, even with p53 mutation. Importantly, SAR specifically induces lysosome-dependent cell death (LCD) by enlarging lysosomes and increasing lysosomal membrane permeabilization associated with leakage of cathepsin B and LAMP-1, regardless of the presence of interleukin-6 or bone marrow stromal cells. Conversely, the lysosomal vacuolar H+-ATPase inhibitor blocks SAR-induced LCD. SAR further upregulates reactive oxygen species. Pom enhances SAR-induced direct and indirect killing even in MM cells resistant to Pom/Len. Taken together, SAR is the first therapeutic monoclonal antibody mediating direct cytotoxicity against MM cells via multiple mechanisms of action. Our data show that Pom augments both direct and effector cell-mediated MM cytotoxicity of SAR, providing the framework for combination clinical trials.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Gozzetti A, Candi V, Papini G, Bocchia M . Therapeutic advancements in multiple myeloma. Front Oncol 2014; 4: 241.
Dimopoulos MA, Richardson PG, Moreau P, Anderson KC . Current treatment landscape for relapsed and/or refractory multiple myeloma. Nat Rev Clin Oncol 2015; 12: 42–54.
Castelli R, Orofino N, Losurdo A, Gualtierotti R, Cugno M . Choosing treatment options for patients with relapsed/refractory multiple myeloma. Expert Rev Anticancer Ther 2014; 14: 199–215.
Tai YT, Anderson KC . Antibody-based therapies in multiple myeloma. Bone Marrow Res 2011; 2011: 924058.
Moreau P, Touzeau C . Elotuzumab for the treatment of multiple myeloma. Future Oncol 2014; 10: 949–956.
Tai YT, Dillon M, Song W, Leiba M, Li XF, Burger P et al. Anti-CS1 humanized monoclonal antibody HuLuc63 inhibits myeloma cell adhesion and induces antibody-dependent cellular cytotoxicity in the bone marrow milieu. Blood 2008; 112: 1329–1337.
de Weers M, Tai YT, van der Veer MS, Bakker JM, Vink T, Jacobs DC et al. Daratumumab, a novel therapeutic human CD38 monoclonal antibody, induces killing of multiple myeloma and other hematological tumors. J Immunol 2011; 186: 1840–1848.
Laubach JP, Tai YT, Richardson PG, Anderson KC . Daratumumab granted breakthrough drug status. Expert Opin Investig Drugs 2014; 23: 445–452.
Deckert J, Wetzel MC, Bartle LM, Skaletskaya A, Goldmacher VS, Vallée F et al. SAR650984, a novel humanized CD38-targeting antibody, demonstrates potent antitumor activity in models of multiple myeloma and other CD38+ hematologic malignancies. Clin Cancer Res 2014; 20: 4574–4583.
Berdeja JG . Lorvotuzumab mertansine: antibody-drug-conjugate for CD56+ multiple myeloma. Front Biosci (Landmark Ed) 2014; 19: 163–170.
Sherbenou DW, Behrens CR, Su Y, Wolf JL, Martin TG 3rd, Liu B . The development of potential antibody-based therapies for myeloma. Blood Rev 2014; 29: 81–91.
Tai YT, Mayes PA, Acharya C, Zhong MY, Cea M, Cagnetta A et al. Novel anti-B-cell maturation antigen antibody-drug conjugate (GSK2857916) selectively induces killing of multiple myeloma. Blood 2014; 123: 3128–3138.
Zonder JA, Mohrbacher AF, Singhal S, van Rhee F, Bensinger WI, Ding H et al. A phase 1, multicenter, open-label, dose escalation study of elotuzumab in patients with advanced multiple myeloma. Blood 2012; 120: 552–559.
Jurisic V, Srdic T, Konjevic G, Markovic O, Colovic M . Clinical stage-depending decrease of NK cell activity in multiple myeloma patients. Med Oncol 2007; 24: 312–317.
Barbier S, Chatre L, Bras M, Sancho P, Roué G, Virely C et al. Caspase-independent type III programmed cell death in chronic lymphocytic leukemia: the key role of the F-actin cytoskeleton. Haematologica 2009; 94: 507–517.
Ivanov A, Beers SA, Walshe CA, Honeychurch J, Alduaij W, Cox KL et al. Monoclonal antibodies directed to CD20 and HLA-DR can elicit homotypic adhesion followed by lysosome-mediated cell death in human lymphoma and leukemia cells. J Clin Invest 2009; 119: 2143–2159.
Alduaij W, Ivanov A, Honeychurch J, Cheadle EJ, Potluri S, Lim SH et al. Novel type II anti-CD20 monoclonal antibody (GA101) evokes homotypic adhesion and actin-dependent, lysosome-mediated cell death in B-cell malignancies. Blood 2011; 117: 4519–4529.
Breton CS, Nahimana A, Aubry D, Macoin J, Moretti P, Bertschinger M et al. A novel anti-CD19 monoclonal antibody (GBR 401) with high killing activity against B cell malignancies. J Hematol Oncol 2014; 7: 33.
Kim D, Wang J, Willingham SB, Martin R, Wernig G, Weissman IL . Anti-CD47 antibodies promote phagocytosis and inhibit the growth of human myeloma cells. Leukemia 2012; 26: 2538–2545.
Chillemi A, Zaccarello G, Quarona V, Ferracin M, Ghimenti C, Massaia M et al. Anti-CD38 antibody therapy: windows of opportunity yielded by the functional characteristics of the target molecule. Mol Med 2013; 19: 99–108.
Bataille R, Jego G, Robillard N, Barillé-Nion S, Harousseau JL, Moreau P et al. The phenotype of normal, reactive and malignant plasma cells. Identification of ‘many and multiple myelomas’ and of new targets for myeloma therapy. Haematologica 2006; 91: 1234–1240.
Kim D, Park CY, Medeiros BC, Weissman IL . CD19-CD45 low/- CD38 high/CD138+ plasma cells enrich for human tumorigenic myeloma cells. Leukemia 2012; 26: 2530–2537.
Martin T III, Baz R, Benson Jr DM, Lendvai N, Campana F, Charpentier E et al. A Phase Ib dose escalation trial of SAR650984 (Anti-CD-38 mAb) in combination with lenalidomide and dexamethasone in relapsed/refractory multiple myeloma. Blood 2014; 124: 83 (Abstract 83).
Tai YT, Landesman Y, Acharya C, Calle Y, Zhong MY, Cea M et al. CRM1 inhibition induces tumor cell cytotoxicity and impairs osteoclastogenesis in multiple myeloma: molecular mechanisms and therapeutic implications. Leukemia 2014; 28: 155–165.
Deaglio S, Vaisitti T, Billington R, Bergui L, Omede P, Genazzani AA et al. CD38/CD19: a lipid raft-dependent signaling complex in human B cells. Blood 2007; 109: 5390–5398.
Honeychurch J, Alduaij W, Azizyan M, Cheadle EJ, Pelicano H, Ivanov A et al. Antibody-induced nonapoptotic cell death in human lymphoma and leukemia cells is mediated through a novel reactive oxygen species-dependent pathway. Blood 2012; 119: 3523–3533.
van Bueren JL, Jakobs D, Kaldenhoven N, Roza M, Hiddingh S, Meesters J et al. Direct in vitro comparison of daratumumab with surrogate analogs of CD38 antibodies MOR03087, SAR650984 and Ab79. Blood 2014; 124: 3474 (Abstract 3474).
Aits S, Jaattela M . Lysosomal cell death at a glance. J Cell Sci 2013; 126: 1905–1912.
Conus S, Perozzo R, Reinheckel T, Peters C, Scapozza L, Yousefi S et al. Caspase-8 is activated by cathepsin D initiating neutrophil apoptosis during the resolution of inflammation. J Exp Med 2008; 205: 685–698.
Droga-Mazovec G, Bojic L, Petelin A, Ivanova S, Romih R, Repnik U et al. Cysteine cathepsins trigger caspase-dependent cell death through cleavage of bid and antiapoptotic Bcl-2 homologues. J Biol Chem 2008; 283: 19140–19150.
Deans JP, Li H, Polyak MJ . CD20-mediated apoptosis: signalling through lipid rafts. Immunology 2002; 107: 176–182.
Drenou B, Amiot L, Setterblad N, Taque S, Guilloux V, Charron D et al. MHC class II signaling function is regulated during maturation of plasmacytoid dendritic cells. J Leukoc Biol 2005; 77: 560–567.
Kumar S, Fonseca R, Ketterling RP, Dispenzieri A, Lacy MQ, Gertz MA et al. Trisomies in multiple myeloma: impact on survival in patients with high-risk cytogenetics. Blood 2012; 119: 2100–2105.
Avet-Loiseau H, Attal M, Campion L, Caillot D, Hulin C, Marit G et al. Long-term analysis of the IFM 99 trials for myeloma: cytogenetic abnormalities [t(4;14), del(17p), 1q gains] play a major role in defining long-term survival. J Clin Oncol 2012; 30: 1949–1952.
Reece D, Song KW, Fu T, Roland B, Chang H, Horsman DE et al. Influence of cytogenetics in patients with relapsed or refractory multiple myeloma treated with lenalidomide plus dexamethasone: adverse effect of deletion 17p13. Blood 2009; 114: 522–525.
Kapoor P, Fonseca R, Rajkumar SV, Sinha S, Gertz MA, Stewart AK et al. Evidence for cytogenetic and fluorescence in situ hybridization risk stratification of newly diagnosed multiple myeloma in the era of novel therapie. Mayo Clin Proc 2010; 85: 532–537.
Teoh PJ, Chung TH, Sebastian S, Choo SN, Yan J, Ng SB et al. p53 haploinsufficiency and functional abnormalities in multiple myeloma. Leukemia 2014; 28: 2066–2074.
Matsuo Y, Drexler HG, Harashima A, Okochi A, Hasegawa A, Kojima K et al. Induction of CD28 on the new myeloma cell line MOLP-8 with t(11;14)(q13;q32) expressing delta/lambda type immunoglobulin. Leuk Res 2004; 28: 869–877.
Erdal H, Berndtsson M, Castro J, Brunk U, Shoshan MC, Linder S . Induction of lysosomal membrane permeabilization by compounds that activate p53-independent apoptosis. Proc Natl Acad Sci U S A 2005; 102: 192–197.
Groth-Pedersen L, Jaattela M . Combating apoptosis and multidrug resistant cancers by targeting lysosomes. Cancer Lett 2013; 332: 265–274.
Villamil Giraldo AM, Appelqvist H, Ederth T, Ollinger K . Lysosomotropic agents: impact on lysosomal membrane permeabilization and cell death. Biochem Soc Trans 2014; 42: 1460–1464.
Petersen NH, Olsen OD, Groth-Pedersen L, Ellegaard AM, Bilgin M, Redmer S et al. Transformation-associated changes in sphingolipid metabolism sensitize cells to lysosomal cell death induced by inhibitors of acid sphingomyelinase. Cancer Cell 2013; 24: 379–393.
Kirkegaard T, Roth AG, Petersen NH, Mahalka AK, Olsen OD, Moilanen I et al. Hsp70 stabilizes lysosomes and reverts Niemann-Pick disease-associated lysosomal pathology. Nature 2010; 463: 549–553.
Singh AT, Evens AM, Prachand SN, Gordon LI . Motexafin gadolinium enhances p53-Mdm2 interactions, reducing p53 and downstream targets in lymphoma cell lines. Anticancer Res 2010; 30: 1131–1136.
Leleu X, Karlin L, Macro M, Hulin C, Garderet L, Roussel M et al. Pomalidomide plus low-dose dexamethasone in multiple myeloma with deletion 17p and/or translocation (4;14): IFM 2010-02 trial results. Blood 2015; 125: 1411–1417.
Acknowledgements
We thank Dr Ti Cai (former employee at Sanofi, currently at EMD Serono, Inc.) for helpful input and all clinical and laboratory members of the Jerome Lipper Multiple Myeloma Center of the Dana-Farber Cancer Institute for support and help for this study. Funding: National Institutes of Health Grants RO1050947, PO1-CA078378 and DF/HCC SPORE in Multiple Myeloma P50CA100707; KCA is an American Cancer Society Clinical Research Professor.
Author Contributions
HJ, Y-TT and KCA conceptualized research and formed the hypothesis of this paper; HJ, GA, CA, XF, MZ, LW, ND and LQ designed, performed experiments, collected and analyzed data; HJ and LW performed confocal microscopy; ZS, GY and FA provided transfectants, reagents and analytic tools; NCM, PR and KCA provided MM patient samples; HJ and Y-TT wrote the manuscript; Y-TT and KCA critically evaluated and edited the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
ZS, GY and FA are employees of Sanofi whose product was used in this research. Y-TT is a consultant for Onyx. PR serves on advisory boards to Millennium, Celgene, Novartis, Johnson & Johnson and Bristol-Myers Squibb. NCM serves on advisory boards to Millennium, Celgene and Novartis. KCA serves on advisory boards to Onyx, Celgene, Gilead, Bristol-Myers Squibb and Sanofi-Aventis and is a scientific founder of Acetylon and Oncopep. The remaining authors declare no competing financial interest.
Additional information
Supplementary Information accompanies this paper on the Leukemia website
Supplementary information
Rights and permissions
About this article
Cite this article
Jiang, H., Acharya, C., An, G. et al. SAR650984 directly induces multiple myeloma cell death via lysosomal-associated and apoptotic pathways, which is further enhanced by pomalidomide. Leukemia 30, 399–408 (2016). https://doi.org/10.1038/leu.2015.240
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/leu.2015.240
This article is cited by
-
Belantamab mafodotin, pomalidomide and dexamethasone in refractory multiple myeloma: a phase 1/2 trial
Nature Medicine (2024)
-
Efficacy and safety of isatuximab plus bortezomib, lenalidomide, and dexamethasone in patients with newly diagnosed multiple myeloma ineligible/with no immediate intent for autologous stem cell transplantation
Leukemia (2023)
-
Mechanism research and treatment progress of NAD pathway related molecules in tumor immune microenvironment
Cancer Cell International (2022)
-
Anti-CD38 antibody therapy for patients with relapsed/refractory multiple myeloma: differential mechanisms of action and recent clinical trial outcomes
Annals of Hematology (2022)
-
Isatuximab plus pomalidomide and dexamethasone in relapsed/refractory multiple myeloma patients with renal impairment: ICARIA-MM subgroup analysis
Leukemia (2021)