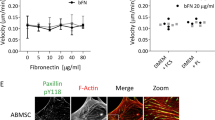
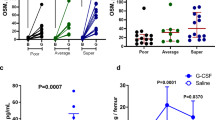
Abstract
We recently demonstrated that acute myeloid leukemia (AML) cell lines and patient-derived blasts release exosomes that carry RNA and protein; following an in vitro transfer, AML exosomes produce proangiogenic changes in bystander cells. We reasoned that paracrine exosome trafficking may have a broader role in shaping the leukemic niche. In a series of in vitro studies and murine xenografts, we demonstrate that AML exosomes downregulate critical retention factors (Scf, Cxcl12) in stromal cells, leading to hematopoietic stem and progenitor cell (HSPC) mobilization from the bone marrow. Exosome trafficking also regulates HSPC directly, and we demonstrate declining clonogenicity, loss of CXCR4 and c-Kit expression, and the consistent repression of several hematopoietic transcription factors, including c-Myb, Cebp-β and Hoxa-9. Additional experiments using a model of extramedullary AML or direct intrafemoral injection of purified exosomes reveal that the erosion of HSPC function can occur independent of direct cell–cell contact with leukemia cells. Finally, using a novel multiplex proteomics technique, we identified candidate pathways involved in the direct exosome-mediated modulation of HSPC function. In aggregate, this work suggests that AML exosomes participate in the suppression of residual hematopoietic function that precedes widespread leukemic invasion of the bone marrow directly and indirectly via stromal components.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Ayala F, Dewar R, Kieran M, Kalluri R . Contribution of bone microenvironment to leukemogenesis and leukemia progression. Leukemia 2009; 23: 2233–2241.
Reynaud D, Pietras E, Barry-Holson K, Mir A, Binnewies M, Jeanne M et al. IL-6 controls leukemic multipotent progenitor cell fate and contributes to chronic myelogenous leukemia development. Cancer Cell 2011; 20: 661–673.
Zhang B, Ho YW, Huang Q, Maeda T, Lin A, Lee SU et al. Altered microenvironmental regulation of leukemic and normal stem cells in chronic myelogenous leukemia. Cancer Cell 2012; 21: 577–592.
Miraki-Moud F, Anjos-Afonso F, Hodby KA, Griessinger E, Rosignoli G, Lillington D et al. Acute myeloid leukemia does not deplete normal hematopoietic stem cells but induces cytopenias by impeding their differentiation. Proc Natl Acad Sci USA 2013; 110: 13576–13581.
Colmone A, Amorim M, Pontier AL, Wang S, Jablonski E, Sipkins DA . Leukemic cells create bone marrow niches that disrupt the behavior of normal hematopoietic progenitor cells. Science 2008; 322: 1861–1865.
Ratajczak J, Miekus K, Kucia M, Zhang J, Reca R, Dvorak P et al. Embryonic stem cell-derived microvesicles reprogram hematopoietic progenitors: evidence for horizontal transfer of mRNA and protein delivery. Leukemia 2006; 20: 847–856.
Huan J, Hornick NI, Shurtleff MJ, Skinner AM, Goloviznina NA, Roberts CT Jr et al. RNA trafficking by acute myelogenous leukemia exosomes. Cancer Res 2013; 73: 918–929.
Chen Y, Jacamo R, Konopleva M, Garzon R, Croce C, Andreeff M . CXCR4 downregulation of let-7a drives chemoresistance in acute myeloid leukemia. J Clin Invest 2013; 123: 2395–2407.
Roberts CT Jr, Kurre P . Vesicle trafficking and RNA transfer add complexity and connectivity to cell-cell communication. Cancer Res 2013; 73: 3200–3205.
Harrison JS, Rameshwar P, Chang V, Bandari P . Oxygen saturation in the bone marrow of healthy volunteers. Blood 2002; 99: 394.
Hatfield KJ, Bedringsaas SL, Ryningen A, Gjertsen BT, Bruserud O . Hypoxia increases HIF- 1alpha expression and constitutive cytokine release by primary human acute myeloid leukaemia cells. Eur Cyt Netw 2010; 21: 154–164.
Lam BS, Adams GB . Blocking HIF1alpha activity eliminates hematological cancer stem cells. Cell Stem Cell 2011; 8: 354–356.
Chen Y, Jacamo R, Shi YX, Wang RY, Battula VL, Konoplev S et al. Human extramedullary bone marrow in mice: a novel in vivo model of genetically controlled hematopoietic microenvironment. Blood 2012; 119: 4971–4980.
Pan D . In situ in vivo gene transfer into murine bone marrow stem cells. Methods Mol Biol 2009; 506: 159–169.
Mazurier F, Doedens M, Gan OI, Dick JE . Rapid myeloerythroid repopulation after intrafemoral transplantation of NOD-SCID mice reveals a new class of human stem cells. Nat Med 2003; 9: 959–963.
Skinner AM, Grompe M, Kurre P . Intra-hematopoietic cell fusion as a source of somatic variation in the hematopoietic system. J Cell Sci 2012; 125: 2837–2843.
Wilmarth PA, Tanner S, Dasari S, Nagalla SR, Riviere MA, Bafna V et al. Age-related changes in human crystallins determined from comparative analysis of post-translational modifications in young and aged lens: does deamidation contribute to crystallin insolubility? J Proteome Res 2006; 5: 2554–2566.
McAlister GC, Huttlin EL, Haas W, Ting L, Jedrychowski MP, Rogers JC et al. Increasing the multiplexing capacity of TMTs using reporter ion isotopologues with isobaric masses. Anal Chem 2012; 84: 7469–7478.
Kall L, Canterbury JD, Weston J, Noble WS, MacCoss MJ . Semi-supervised learning for peptide identification from shotgun proteomics datasets. Nat Methods 2007; 4: 923–925.
Robinson MD, McCarthy DJ, Smyth GK . edgeR: a Bioconductor package for differential expression analysis of digital gene expression data. Bioinformatics 2010; 26: 139–140.
Kucharzewska P, Christianson HC, Welch JE, Svensson KJ, Fredlund E, Ringner M et al. Exosomes reflect the hypoxic status of glioma cells and mediate hypoxia-dependent activation of vascular cells during tumor development. Proc Natl Acad Sci USA 2013; 110: 7312–7317.
Schepers K, Pietras EM, Reynaud D, Flach J, Binnewies M, Garg T et al. Myeloproliferative neoplasia remodels the endosteal bone marrow niche into a self-reinforcing leukemic niche. Cell Stem Cell 2013; 13: 285–299.
Ghosh AK, Shanafelt TD, Cimmino A, Taccioli C, Volinia S, Liu CG et al. Aberrant regulation of pVHL levels by microRNA promotes the HIF/VEGF axis in CLL B cells. Blood 2009; 113: 5568–5574.
Ohno Y, Yasunaga S, Janmohamed S, Ohtsubo M, Saeki K, Kurogi T et al. Hoxa9 transduction induces hematopoietic stem and progenitor cell activity through direct down-regulation of geminin protein. PloS One 2013; 8: e53161.
Chong JL, Tsai SY, Sharma N, Opavsky R, Price R, Wu L et al. E2f3a and E2f3b contribute to the control of cell proliferation and mouse development. Mol Cell Biol 2009; 29: 414–424.
Sattler M, Verma S, Pride YB, Salgia R, Rohrschneider LR, Griffin JD . SHIP1 an SH2 domain containing polyinositol-5-phosphatase, regulates migration through two critical tyrosine residues and forms a novel signaling complex with DOK1 and CRKL. J Biol Chem 2001; 276: 2451–2458.
Huang X, Ding L, Bennewith KL, Tong RT, Welford SM, Ang KK et al. Hypoxia-inducible mir-210 regulates normoxic gene expression involved in tumor initiation. Mol Cell 2009; 35: 856–867.
Lee DW, Futami M, Carroll M, Feng Y, Wang Z, Fernandez M et al. Loss of SHIP-1 protein expression in high-risk myelodysplastic syndromes is associated with miR-210 and miR-155. Oncogene 2012; 31: 4085–4094.
Xiao C, Calado DP, Galler G, Thai TH, Patterson HC, Wang J et al. MiR-150 controls B cell differentiation by targeting the transcription factor c-Myb. Cell 2007; 131: 146–159.
Huang da W, Sherman BT, Lempicki RA . Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat Protoc 2009; 4: 44–57.
Huang da W, Sherman BT, Lempicki RA . Bioinformatics enrichment tools: paths toward the comprehensive functional analysis of large gene lists. Nucleic Acids Res 2009; 37: 1–13.
Jensen LJ, Kuhn M, Stark M, Chaffron S, Creevey C, Muller J et al. STRING 8–a global view on proteins and their functional interactions in 630 organisms. Nucleic Acids Res 2009; 37: D412–D416.
Jin L, Hope KJ, Zhai Q, Smadja-Joffe F, Dick JE . Targeting of CD44 eradicates human acute myeloid leukemic stem cells. Nat Med 2006; 12: 1167–1174.
Jacamo R, Chen Y, Wang Z, Ma W, Zhang M, Spaeth EL et al. Reciprocal leukemia-stroma VCAM-1/VLA-4-dependent activation of NF-kappaB mediates chemoresistance. Blood 2014; 123: 2691–2702.
Mizuno S, Chijiwa T, Okamura T, Akashi K, Fukumaki Y, Niho Y et al. Expression of DNA methyltransferases DNMT1, 3A, and 3B in normal hematopoiesis and in acute and chronic myelogenous leukemia. Blood 2001; 97: 1172–1179.
Sugiyama T, Kohara H, Noda M, Nagasawa T . Maintenance of the hematopoietic stem cell pool by CXCL12-CXCR4 chemokine signaling in bone marrow stromal cell niches. Immunity 2006; 25: 977–988.
Cosgun KN, Rahmig S, Mende N, Reinke S, Hauber I, Schafer C et al. Kit regulates HSC engraftment across the human-mouse species barrier. Cell Stem Cell 2014; 15: 227–238.
Greenbaum A, Hsu YM, Day RB, Schuettpelz LG, Christopher MJ, Borgerding JN et al. CXCL12 in early mesenchymal progenitors is required for haematopoietic stem-cell maintenance. Nature 2013; 495: 227–230.
Mocsai A, Ruland J, Tybulewicz VL . The SYK tyrosine kinase: a crucial player in diverse biological functions. Nat Rev Immunol 2010; 10: 387–402.
Puissant A, Fenouille N, Alexe G, Pikman Y, Bassil CF, Mehta S et al. SYK is a critical regulator of FLT3 in acute myeloid leukemia. Cancer Cell 2014; 25: 226–242.
Hahn CK, Berchuck JE, Ross KN, Kakoza RM, Clauser K, Schinzel AC et al. Proteomic and genetic approaches identify Syk as an AML target. Cancer Cell 2009; 16: 281–294.
Gross O, Poeck H, Bscheider M, Dostert C, Hannesschlager N, Endres S et al. Syk kinase signalling couples to the Nlrp3 inflammasome for anti-fungal host defence. Nature 2009; 459: 433–436.
Yonemura Y, Ku H, Hirayama F, Souza LM, Ogawa M . Interleukin 3 or interleukin 1 abrogates the reconstituting ability of hematopoietic stem cells. Proc Natl Acad Sci USA 1996; 93: 4040–4044.
Hartwig UF, Keller U, Huber C, Peschel C . Regulation of hematopoietic growth factor production by genetically modified human bone marrow stromal cells expressing interleukin-1beta antisense RNA. J Interferon Cytokine Res 2001; 21: 851–860.
Signer RA, Magee JA, Salic A, Morrison SJ . Haematopoietic stem cells require a highly regulated protein synthesis rate. Nature 2014; 509: 49–54.
Peinado H, Aleckovic M, Lavotshkin S, Matei I, Costa-Silva B, Moreno-Bueno G et al. Melanoma exosomes educate bone marrow progenitor cells toward a pro-metastatic phenotype through MET. Nat Med 2012; 18: 883–891.
Barcellos-Hoff MH, Lyden D, Wang TC . The evolution of the cancer niche during multistage carcinogenesis. Nat Rev Cancer 2013; 13: 511–518.
Acknowledgements
This manuscript is dedicated to the late Dr Zili Zhang, a thoughtful scientist, inspired colleague and, above all, a good friend. We acknowledge the assistance of Dr Muneesh Tewari, Dr Ashok Reddy, John Klimek, Rajani Kaimal, Aurelie Snyder and Jennifer Wherley. Studies were supported in part by the St. Baldrick’s Foundation (PK), the Hyundai Hope on Wheels Foundation (PK), NIH/NIAID T32 grant (5T32AI78903-5) (NH), NIH core grants P30EY10572 and P30CA069533 and a Canary Foundation/American Cancer Society (ACS) Postdoctoral Fellowship (PFTED-09-249-01-SEID) with support by the Hillcrest Committee of Southern Oregon and the ACS Great West Division (JRC).
Author contributions
JH designed and performed experiments, interpreted data and prepared the manuscript; NH designed and performed experiments and interpreted data; NG performed experiments; ANK interpreted data and edited the manuscript; LLD interpreted data and edited the manuscript; PAW performed experiments and interpreted data; TM interpreted data and edited the manuscript; JRC performed experiments; AN performed experiments and interpreted data; CR interpreted data and edited the manuscript; ML interpreted data and edited the manuscript; BHC interpreted data and edited the manuscript; PK designed experiments, interpreted data and edited the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interests.
Additional information
Supplementary Information accompanies this paper on the Leukemia website
Supplementary information
Rights and permissions
About this article
Cite this article
Huan, J., Hornick, N., Goloviznina, N. et al. Coordinate regulation of residual bone marrow function by paracrine trafficking of AML exosomes. Leukemia 29, 2285–2295 (2015). https://doi.org/10.1038/leu.2015.163
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/leu.2015.163
This article is cited by
-
A novel therapeutic strategy: the significance of exosomal miRNAs in acute myeloid leukemia
Medical Oncology (2024)
-
Exosome application in treatment and diagnosis of B-cell disorders: leukemias, multiple sclerosis, and arthritis rheumatoid
Cellular & Molecular Biology Letters (2022)
-
The genesis and evolution of acute myeloid leukemia stem cells in the microenvironment: From biology to therapeutic targeting
Cell Death Discovery (2022)
-
Pathogenic Mechanisms in Acute Myeloid Leukemia
Current Treatment Options in Oncology (2022)
-
Oxidative resistance of leukemic stem cells and oxidative damage to hematopoietic stem cells under pro-oxidative therapy
Cell Death & Disease (2020)