Abstract
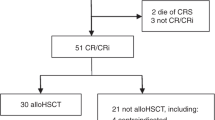
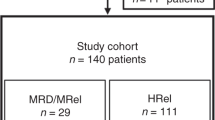
The efficacy of reduced intensity conditioning (RIC) allogeneic hematopoietic cell transplantation (HCT) for Philadelphia chromosome positive (Ph+) acute lymphoblastic leukemia (ALL) is uncertain. We analyzed 197 adults with Ph+ ALL in first complete remission; 67 patients receiving RIC were matched with 130 receiving myeloablative conditioning (MAC) for age, donor type and HCT year. Over 75% received pre-HCT tyrosine kinase inhibitors (TKIs), mostly imatinib; 39% (RIC) and 49% (MAC) were minimal residual disease (MRD)neg pre-HCT. At a median 4.5 years follow-up, 1-year transplant-related mortality (TRM) was lower in RIC (13%) than MAC (36%; P=0.001) while the 3-year relapse rate was 49% in RIC and 28% in MAC (P=0.058). Overall survival (OS) was similar (RIC 39% (95% confidence interval (CI) 27–52) vs 35% (95% CI 27–44); P=0.62). Patients MRDpos pre-HCT had higher risk of relapse with RIC vs MAC (hazard ratio (HR) 1.97; P=0.026). However, patients receiving pre-HCT TKI in combination with MRD negativity pre-RIC HCT had superior OS (55%) compared with a similar MRD population after MAC (33%; P=0.0042). In multivariate analysis, RIC lowered TRM (HR 0.6; P=0.057), but absence of pre-HCT TKI (HR 1.88; P=0.018), RIC (HR 1.891; P=0.054) and pre-HCT MRDpos (HR 1.6; P=0.070) increased relapse risk. RIC is a valid alternative strategy for Ph+ ALL patients ineligible for MAC and MRDneg status is preferred pre-HCT.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Moorman AV, Harrison CJ, Buck GA, Richards SM, Secker-Walker LM, Martineau M et al. Karyotype is an independent prognostic factor in adult acute lymphoblastic leukemia (ALL): analysis of cytogenetic data from patients treated on the Medical Research Council (MRC) UKALLXII/Eastern Cooperative Oncology Group (ECOG) 2993 trial. Blood 2007; 109: 3189–3197.
Mizuta S, Matsuo K, Yagasaki F, Yujiri T, Hatta Y, Kimura Y et al. Pre-transplant imatinib-based therapy improves the outcome of allogeneic hematopoietic stem cell transplantation for BCR-ABL-positive acute lymphoblastic leukemia. Leukemia 2011; 25: 41–47.
Ottmann OG, Wassmann B, Pfeifer H, Giagounidis A, Stelljes M, Duhrsen U et al. Imatinib compared with chemotherapy as front-line treatment of elderly patients with Philadelphia chromosome-positive acute lymphoblastic leukemia (Ph+ALL). Cancer 2007; 109: 2068–2076.
Tanguy-Schmidt A, Rousselot P, Chalandon Y, Cayuela JM, Hayette S, Vekemans MC et al. Long-term follow-up of the imatinib GRAAPH-2003 Study in newly diagnosed patients with de novo Philadelphia chromosome-positive acute lymphoblastic leukemia: a GRAALL Study. Biol Blood Marrow Transplant 2013; 19: 150–155.
Fielding AK, Rowe JM, Richards SM, Buck G, Moorman AV, Durrant IJ et al. Prospective outcome data on 267 unselected adult patients with Philadelphia chromosome-positive acute lymphoblastic leukemia confirms superiority of allogeneic transplantation over chemotherapy in the pre-imatinib era: results from the International ALL Trial MRC UKALLXII/ECOG2993. Blood 2009; 113: 4489–4496.
Dombret H, Gabert J, Boiron JM, Rigal-Huguet F, Blaise D, Thomas X et al. Outcome of treatment in adults with Philadelphia chromosome-positive acute lymphoblastic leukemia—results of the prospective multicenter LALA-94 trial. Blood 2002; 100: 2357–2366.
Laport GG, Alvarnas JC, Palmer JM, Snyder DS, Slovak ML, Cherry AM et al. Long-term remission of Philadelphia chromosome-positive acute lymphoblastic leukemia after allogeneic hematopoietic cell transplantation from matched sibling donors: a 20-year experience with the fractionated total body irradiation-etoposide regimen. Blood 2008; 112: 903–909.
Hunault M, Harousseau JL, Delain M, Truchen-Graczyk M, Cahn JY, Witz F et al. Better outcome of adult acute lymphoblastic leukemia after early genoidentical allogeneic bone marrow transplantation (BMT) than after late high-dose therapy and autologous BMT: a GOELAMS trial. Blood 2004; 104: 3028–3037.
Thomas DA, Faderl S, Cortes J, O’Brien S, Giles FJ, Kornblau SM et al. Treatment of Philadelphia chromosome-positive acute lymphocytic leukemia with hyper-CVAD and imatinib mesylate. Blood 2004; 103: 4396–4407.
de Labarthe A, Rousselot P, Huguet-Rigal F, Delabesse E, Witz F, Maury S et al. Imatinib combined with induction or consolidation chemotherapy in patients with de novo Philadelphia chromosome-positive acute lymphoblastic leukemia: results of the GRAAPH-2003 study. Blood 2007; 109: 1408–1413.
Ribera JM, Oriol A, Gonzalez M, Vidriales B, Brunet S, Esteve J et al. Concurrent intensive chemotherapy and imatinib before and after stem cell transplantation in newly diagnosed Philadelphia chromosome-positive acute lymphoblastic leukemia. Final results of the CSTIBES02 trial. Haematologica 2010; 95: 87–95.
Yanada M, Matsuo K, Suzuki T, Naoe T . Allogeneic hematopoietic stem cell transplantation as part of postremission therapy improves survival for adult patients with high-risk acute lymphoblastic leukemia: a metaanalysis. Cancer 2006; 106: 2657–2663.
Wassmann B, Pfeifer H, Stadler M, Bornhauser M, Bug G, Scheuring UJ et al. Early molecular response to posttransplantation imatinib determines outcome in MRD+ Philadelphia-positive acute lymphoblastic leukemia (Ph+ ALL). Blood 2005; 106: 458–463.
Goldstone AH, Richards SM, Lazarus HM, Tallman MS, Buck G, Fielding AK et al. In adults with standard-risk acute lymphoblastic leukemia, the greatest benefit is achieved from a matched sibling allogeneic transplantation in first complete remission, and an autologous transplantation is less effective than conventional consolidation/maintenance chemotherapy in all patients: final results of the International ALL Trial (MRC UKALL XII/ECOG E2993). Blood 2008; 111: 1827–1833.
Ram R, Storb R, Sandmaier BM, Maloney DG, Woolfrey A, Flowers ME et al. Non-myeloablative conditioning with allogeneic hematopoietic cell transplantation for the treatment of high-risk acute lymphoblastic leukemia. Haematologica 2011; 96: 1113–1120.
Medd PG, Peniket AJ, Littlewood TJ, Pearce R, Perry J, Kirkland KE et al. Evidence for a GVL effect following reduced-intensity allo-SCT in ALL: a British Society of Blood and Marrow Transplantation study. Bone Marrow Transplant 2013; 48: 982–987.
Cho BS, Lee S, Kim YJ, Chung NG, Eom KS, Kim HJ et al. Reduced-intensity conditioning allogeneic stem cell transplantation is a potential therapeutic approach for adults with high-risk acute lymphoblastic leukemia in remission: results of a prospective phase 2 study. Leukemia 2009; 23: 1763–1770.
Stein AS, Palmer JM, O’Donnell MR, Kogut NM, Spielberger RT, Slovak ML et al. Reduced-intensity conditioning followed by peripheral blood stem cell transplantation for adult patients with high-risk acute lymphoblastic leukemia. Biol Blood Marrow Transplant 2009; 15: 1407–1414.
Bachanova V, Verneris MR, DeFor T, Brunstein CG, Weisdorf DJ . Prolonged survival in adults with acute lymphoblastic leukemia after reduced-intensity conditioning with cord blood or sibling donor transplantation. Blood 2009; 113: 2902–2905.
Marks DI, Wang T, Perez WS, Antin JH, Copelan E, Gale RP et al. The outcome of full-intensity and reduced-intensity conditioning matched sibling or unrelated donor transplantation in adults with Philadelphia chromosome-negative acute lymphoblastic leukemia in first and second complete remission. Blood 2010; 116: 366–374.
Mohty M, Labopin M, Volin L, Gratwohl A, Socie G, Esteve J et al. Reduced-intensity versus conventional myeloablative conditioning allogeneic stem cell transplantation for patients with acute lymphoblastic leukemia: a retrospective study from the European Group for Blood and Marrow Transplantation. Blood 2010; 116: 4439–4443.
Radich JP, Kopecky KJ, Boldt DH, Head D, Slovak ML, Babu R et al. Detection of BCR-ABL fusion genes in adult acute lymphoblastic leukemia by the polymerase chain reaction. Leukemia 1994; 8: 1688–1695.
Lee S, Kim DW, Cho B, Kim YJ, Kim YL, Hwang JY et al. Risk factors for adults with Philadelphia-chromosome-positive acute lymphoblastic leukemia in remission treated with allogeneic bone marrow transplantation: the potential of real-time quantitative reverse-transcription polymerase chain reaction. Br J Haematol 2003; 120: 145–153.
Yanada M, Sugiura I, Takeuchi J, Akiyama H, Maruta A, Ueda Y et al. Prospective monitoring of BCR-ABL1 transcript levels in patients with Philadelphia chromosome-positive acute lymphoblastic leukaemia undergoing imatinib-combined chemotherapy. Br J Haematol 2008; 143: 503–510.
Pane F, Cimino G, Izzo B, Camera A, Vitale A, Quintarelli C et al. Significant reduction of the hybrid BCR/ABL transcripts after induction and consolidation therapy is a powerful predictor of treatment response in adult Philadelphia-positive acute lymphoblastic leukemia. Leukemia 2005; 19: 628–635.
Weisdorf D, Spellman S, Haagenson M, Horowitz M, Lee S, Anasetti C et al. Classification of HLA-matching for retrospective analysis of unrelated donor transplantation: revised definitions to predict survival. Biol Blood Marrow Transplant 2008; 14: 748–758.
Bassan R, Rossi G, Pogliani EM, Di Bona E, Angelucci E, Cavattoni I et al. Chemotherapy-phased imatinib pulses improve long-term outcome of adult patients with Philadelphia chromosome-positive acute lymphoblastic leukemia: Northern Italy Leukemia Group protocol 09/00. J Clin Oncol 2010; 28: 3644–3652.
Giralt S, Ballen K, Rizzo D, Bacigalupo A, Horowitz M, Pasquini M et al. Reduced-intensity conditioning regimen workshop: defining the dose spectrum. Report of a workshop convened by the center for international blood and marrow transplant research. Biol Blood Marrow Transplant 2009; 15: 367–369.
Gooley TA, Leisenring W, Crowley J, Storer BE . Estimation of failure probabilities in the presence of competing risks: new representations of old estimators. Stat Med 1999; 18: 695–706.
Sorror ML, Maris MB, Storb R, Baron F, Sandmaier BM, Maloney DG et al. Hematopoietic cell transplantation (HCT)-specific comorbidity index: a new tool for risk assessment before allogeneic HCT. Blood 2005; 106: 2912–2919.
Vignetti M, Fazi P, Cimino G, Martinelli G, Di Raimondo F, Ferrara F et al. Imatinib plus steroids induces complete remissions and prolonged survival in elderly Philadelphia chromosome-positive patients with acute lymphoblastic leukemia without additional chemotherapy: results of the Gruppo Italiano Malattie Ematologiche dell’Adulto (GIMEMA) LAL0201-B protocol. Blood 2007; 109: 3676–3678.
Delannoy A, Delabesse E, Lheritier V, Castaigne S, Rigal-Huguet F, Raffoux E et al. Imatinib and methylprednisolone alternated with chemotherapy improve the outcome of elderly patients with Philadelphia-positive acute lymphoblastic leukemia: results of the GRAALL AFR09 study. Leukemia 2006; 20: 1526–1532.
Pfeifer H, Wassmann B, Pavlova A, Wunderle L, Oldenburg J, Binckebanck A et al. Kinase domain mutations of BCR-ABL frequently precede imatinib-based therapy and give rise to relapse in patients with de novo Philadelphia-positive acute lymphoblastic leukemia (Ph+ ALL). Blood 2007; 110: 727–734.
Watanabe K, Minami Y, Ozawa Y, Miyamura K, Naoe T . T315I mutation in Ph-positive acute lymphoblastic leukemia is associated with a highly aggressive disease phenotype: three case reports. Anticancer Res 2012; 32: 1779–1783.
Soiffer RJ, Lerademacher J, Ho V, Kan F, Artz A, Champlin RE et al. Impact of immune modulation with anti-T-cell antibodies on the outcome of reduced-intensity allogeneic hematopoietic stem cell transplantation for hematologic malignancies. Blood 2011; 117: 6963–6970.
Riva G, Luppi M, Barozzi P, Quadrelli C, Basso S, Vallerini D et al. Emergence of BCR-ABL-specific cytotoxic T cells in the bone marrow of patients with Ph+ acute lymphoblastic leukemia during long-term imatinib mesylate treatment. Blood 2010; 115: 1512–1518.
Topp MS, Kufer P, Gokbuget N, Goebeler M, Klinger M, Neumann S et al. Targeted therapy with the T-cell-engaging antibody blinatumomab of chemotherapy-refractory minimal residual disease in B-lineage acute lymphoblastic leukemia patients results in high response rate and prolonged leukemia-free survival. J Clin Oncol 2011; 29: 2493–2498.
Foa R, Vitale A, Vignetti M, Meloni G, Guarini A, De Propris MS et al. Dasatinib as first-line treatment for adult patients with Philadelphia chromosome-positive acute lymphoblastic leukemia. Blood 2011; 118: 6521–6528.
Ottmann OG, Larson RA, Kantarjian HM, le Coutre PD, Baccarani M, Hochhaus A et al. Phase II study of nilotinib in patients with relapsed or refractory Philadelphia chromosome-positive acute lymphoblastic leukemia. Leukemia 2012; 27: 1411–1413.
Grupp SA, Kalos M, Barrett D, Aplenc R, Porter DL, Rheingold SR et al. Chimeric antigen receptor-modified T cells for acute lymphoid leukemia. N Engl J Med 2013; 368: 1509–1518.
Bassan R, Spinelli O, Oldani E, Intermesoli T, Tosi M, Peruta B et al. Improved risk classification for risk-specific therapy based on the molecular study of minimal residual disease (MRD) in adult acute lymphoblastic leukemia (ALL). Blood 2009; 113: 4153–4162.
Pfeifer H, Wassmann B, Bethge W, Dengler J, Bornhauser M, Stadler M et al. Randomized comparison of prophylactic and minimal residual disease-triggered imatinib after allogeneic stem cell transplantation for BCR-ABL1 positive acute lymphoblastic leukemia. Leukemia 2012; 27: 1254–1262.
Acknowledgements
This work was supported in part by Novartis providing the grant to CIBMTR for supplemental data collection. The CIBMTR is supported by Public Health Service Grant/Cooperative Agreement U24 CA076518 from the National Cancer Institute (NCI), the National Heart, Lung and Blood Institute (NHLBI) and the National Institute of Allergy and Infectious Diseases (NIAID); a Grant/Cooperative Agreement U10 HL069294 from NHLBI and NCI; a contract HHSH250201200016C with Health Resources and Services Administration (HRSA/DHHS); two grants N00014-12-1-0142 and N00014-13-1-0039 from the Office of Naval Research; and grants from Allos Therapeutics, Inc.; Amgen, Inc. Anonymous donation to the Medical College of Wisconsin; Ariad; Be the Match Foundation; Blue Cross and Blue Shield Association; Celgene Corporation; Fresenius-Biotech North America, Inc.; Gamida Cell Teva Joint Venture Ltd; Genentech, Inc.; Gentium SpA; Genzyme Corporation; GlaxoSmithKline; HistoGenetics, Inc.; Kiadis Pharma; the Leukemia and Lymphoma Society; the Medical College of Wisconsin; Merck & Co, Inc.; Millennium: The Takeda Oncology Co.; Milliman USA, Inc.; Miltenyi Biotec, Inc.; National Marrow Donor Program; Onyx Pharmaceuticals; Optum Healthcare Solutions, Inc.; Osiris Therapeutics, Inc.; Otsuka America Pharmaceutical, Inc.; Remedy Informatics; Sanofi US; Seattle Genetics; Sigma-Tau Pharmaceuticals; Soligenix, Inc.; StemCyte, A Global Cord Blood Therapeutics Co.; Stemsoft Software, Inc.; Swedish Orphan Biovitrum; Tarix Pharmaceuticals; TerumoBCT; Teva Neuroscience, Inc.; THERAKOS, Inc.; and Wellpoint, Inc. The views expressed in this article do not reflect the official policy or position of the National Institute of Health, the Department of the Navy, the Department of Defense or any other agency of the US Government.
Author contributions
VB, DJW: designed the study, assisted in supplemental data collection, interpreted data and wrote the manuscript. DIM: assisted in data interpretation, analysis and writing the manuscript. HW; M-JZ: collected and analyzed data, performed statistical analysis. Other authors reviewed the analyses, modified and approved the final manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Scientific Subheading: Transplantation
Rights and permissions
About this article
Cite this article
Bachanova, V., Marks, D., Zhang, MJ. et al. Ph+ ALL patients in first complete remission have similar survival after reduced intensity and myeloablative allogeneic transplantation: impact of tyrosine kinase inhibitor and minimal residual disease. Leukemia 28, 658–665 (2014). https://doi.org/10.1038/leu.2013.253
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/leu.2013.253
Keywords
This article is cited by
-
Measurable residual disease (MRD)-testing in haematological and solid cancers
Leukemia (2024)
-
Reduced-intensity allogenic transplantation for children and adolescents with Philadelphia chromosome-positive acute lymphoblastic leukemia
Annals of Hematology (2024)
-
Myeloablative or reduced-intensity/non-myeloablative hematopoietic cell transplantation for Philadelphia-positive acute lymphoblastic leukemia in adults older than 40 years old — a secondary analysis of a CIBMTR database
Annals of Hematology (2024)
-
Phase II study of myeloablative allogeneic hematopoietic stem cell transplantation for acute lymphoblastic leukemia in adults using fludarabine and total body irradiation 12 Gy
Bone Marrow Transplantation (2022)
-
MRD in ALL: Optimization and Innovations
Current Hematologic Malignancy Reports (2022)