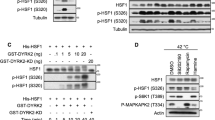
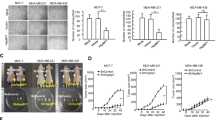
Abstract
Understanding the mechanisms that control stress-induced apoptosis is critical to explain how tumours respond to treatment, as cancer cells frequently escape drug toxicity by regulating stress response through heat shock protein (HSP) expression. The overexpression of Hsp72, in particular, results in increased incidence of cell transformation, and correlates with poor prognosis in a wide range of cancers. We have shown that Hsp72 assists folding of oncogenic NPM–ALK kinase in anaplastic large-cell lymphomas (ALCLs), but its role in the maintenance of the malignant phenotype remains uncertain. Therefore, we assessed Hsp72 expression in ALCLs, investigating more in detail the mechanisms that regulate its status and activity. We found that Hsp72 is unique among the HSPs involved in tumourigenesis to be overexpressed in ALK+ tumours and cell lines and to be induced by stress. Different from other HSPs, Hsp72 prevents cell injury, Bax activation and death by apoptosis in ALK+ cells, acting both upstream and downstream of mitochondria. Conversely, Hsp72 is underexpressed in ALK− ALCL cells, and it is unable to protect cells from apoptosis under stress. Moreover, when Hsp72 expression is reduced following NPM–ALK inhibition, lymphoma cells undergo apoptosis, demonstrating the importance of Hsp72 in regulating ALCL stress response and drug sensitivity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Arya R, Mallik M, Lakhotia SC . Heat shock genes - integrating cell survival and death. J Biosci 2007; 32: 595–610.
Galluzzi L, Giordanetto F, Kroemer G . Targeting HSP70 for cancer therapy. Mol Cell 2009; 36: 176–177.
Awasthi N, Wagner BJ . Upregulation of heat shock protein expression by proteasome inhibition: an antiapoptotic mechanism in the lens. Invest Ophthalmol Vis Sci 2005; 46: 2082–2091.
Kampinga HH, Craig EA . The HSP70 chaperone machinery: J proteins as drivers of functional specificity. Nat Rev Mol Cell Biol 2010; 11: 579–592.
Abravaya K, Myers MP, Murphy SP, Morimoto RI . The human heat shock protein hsp70 interacts with HSF, the transcription factor that regulates heat shock gene expression. Genes Dev 1992; 6: 1153–1164.
Bonvini P, Gastaldi T, Falini B, Rosolen A . Nucleophosmin-anaplastic lymphoma kinase (NPM-ALK), a novel Hsp90-client tyrosine kinase: down-regulation of NPM-ALK expression and tyrosine phosphorylation in ALK(+) CD30(+) lymphoma cells by the Hsp90 antagonist 17-allylamino,17-demethoxygeldanamycin. Cancer Res 2002; 62: 1559–1566.
Lagarrigue F, Dupuis-Coronas S, Ramel D, Delsol G, Tronchere H, Payrastre B et al. Matrix metalloproteinase-9 is upregulated in nucleophosmin-anaplastic lymphoma kinase-positive anaplastic lymphomas and activated at the cell surface by the chaperone heat shock protein 90 to promote cell invasion. Cancer Res 2010; 70: 6978–6987.
Kinney MC, Higgins RA, Medina EA . Anaplastic large cell lymphoma: twenty-five years of discovery. Arch Pathol Lab Med 2011; 135: 19–43.
Bonvini P, Zorzi E, Basso G, Rosolen A . Bortezomib-mediated 26S proteasome inhibition causes cell-cycle arrest and induces apoptosis in CD-30+ anaplastic large cell lymphoma. Leukemia 2007; 21: 838–842.
Bonvini P, Zorzi E, Mussolin L, Monaco G, Pigazzi M, Basso G et al. The effect of the cyclin-dependent kinase inhibitor flavopiridol on anaplastic large cell lymphoma cells and relationship with NPM-ALK kinase expression and activity. Haematologica 2009; 94: 944–955.
Mussolin L, Pillon M, d’Amore ES, Santoro N, Lombardi A, Fagioli F et al. Prevalence and clinical implications of bone marrow involvement in pediatric anaplastic large cell lymphoma. Leukemia 2005; 19: 1643–1647.
Ghisi M, Corradin A, Basso K, Frasson C, Serafin V, Mukherjee S et al. Modulation of microRNA expression in human T-cell development: targeting of Notch3 by miR-150. Blood 2011; 117: 7053–7062.
Joly AL, Wettstein G, Mignot G, Ghiringhelli F, Garrido C . Dual role of heat shock proteins as regulators of apoptosis and innate immunity. J Innate Immun 2010; 2: 238–247.
Zou J, Guo Y, Guettouche T, Smith DF, Voellmy R . Repression of heat shock transcription factor HSF1 activation by HSP90 (HSP90 complex) that forms a stress-sensitive complex with HSF1. Cell 1998; 94: 471–480.
Didelot C, Schmitt E, Brunet M, Maingret L, Parcellier A, Garrido C . Heat shock proteins: endogenous modulators of apoptotic cell death. Handb Exp Pharmacol 2006; 172: 171–198.
Jego G, Hazoume A, Seigneuric R, Garrido C . Targeting heat shock proteins in cancer. Cancer Lett 2010; e-pub ahead of print 13 November 2010; doi:10.1016/j.canlet.2010.10.014.
Pirkkala L, Nykanen P, Sistonen L . Roles of the heat shock transcription factors in regulation of the heat shock response and beyond. FASEB J 2001; 15: 1118–1131.
Dudeja V, Mujumdar N, Phillips P, Chugh R, Borja-Cacho D, Dawra RK et al. Heat shock protein 70 inhibits apoptosis in cancer cells through simultaneous and independent mechanisms. Gastroenterology 2009; 136: 1772–1782.
Bai RY, Ouyang T, Miething C, Morris SW, Peschel C, Duyster J . Nucleophosmin-anaplastic lymphoma kinase associated with anaplastic large-cell lymphoma activates the phosphatidylinositol 3-kinase/Akt antiapoptotic signaling pathway. Blood 2000; 96: 4319–4327.
Chiarle R, Gong JZ, Guasparri I, Pesci A, Cai J, Liu J et al. NPM-ALK transgenic mice spontaneously develop T-cell lymphomas and plasma cell tumors. Blood 2003; 101: 1919–1927.
Chiarle R, Simmons WJ, Cai H, Dhall G, Zamo A, Raz R et al. Stat3 is required for ALK-mediated lymphomagenesis and provides a possible therapeutic target. Nat Med 2005; 11: 623–629.
Ladanyi M . The NPM/ALK gene fusion in the pathogenesis of anaplastic large cell lymphoma. Cancer Surv 1997; 30: 59–75.
Mosse YP, Wood A, Maris JM . Inhibition of ALK signaling for cancer therapy. Clin Cancer Res 2009; 15: 5609–5614.
Palmer RH, Vernersson E, Grabbe C, Hallberg B . Anaplastic lymphoma kinase: signalling in development and disease. Biochem J 2009; 420: 345–361.
Piva R, Chiarle R, Manazza AD, Taulli R, Simmons W, Ambrogio C et al. Ablation of oncogenic ALK is a viable therapeutic approach for anaplastic large-cell lymphomas. Blood 2006; 107: 689–697.
Solimini NL, Luo J, Elledge SJ . Non-oncogene addiction and the stress phenotype of cancer cells. Cell 2007; 130: 986–988.
Tyedmers J, Mogk A, Bukau B . Cellular strategies for controlling protein aggregation. Nat Rev Mol Cell Biol 2010; 11: 777–788.
Young JC . Mechanisms of the Hsp70 chaperone system. Biochem Cell Biol 2010; 88: 291–300.
Morimoto RI, Santoro MG . Stress-inducible responses and heat shock proteins: new pharmacologic targets for cytoprotection. Nat Biotechnol 1998; 16: 833–838.
Garrido C, Schmitt E, Cande C, Vahsen N, Parcellier A, Kroemer G . HSP27 and HSP70: potentially oncogenic apoptosis inhibitors. Cell Cycle 2003; 2: 579–584.
Diakos C, Krapf G, Gerner C, Inthal A, Lemberger C, Ban J et al. RNAi-mediated silencing of TEL/AML1 reveals a heat-shock protein- and survivin-dependent mechanism for survival. Blood 2007; 109: 2607–2610.
Guo F, Sigua C, Bali P, George P, Fiskus W, Scuto A et al. Mechanistic role of heat shock protein 70 in Bcr-Abl-mediated resistance to apoptosis in human acute leukemia cells. Blood 2005; 105: 1246–1255.
Pocaly M, Lagarde V, Etienne G, Ribeil JA, Claverol S, Bonneu M et al. Overexpression of the heat-shock protein 70 is associated to imatinib resistance in chronic myeloid leukemia. Leukemia 2007; 21: 93–101.
Yao Q, Nishiuchi R, Li Q, Kumar AR, Hudson WA, Kersey JH . FLT3 expressing leukemias are selectively sensitive to inhibitors of the molecular chaperone heat shock protein 90 through destabilization of signal transduction-associated kinases. Clin Cancer Res 2003; 9: 4483–4493.
Gotoh T, Terada K, Oyadomari S, Mori M . hsp70-DnaJ chaperone pair prevents nitric oxide- and CHOP-induced apoptosis by inhibiting translocation of Bax to mitochondria. Cell Death Differ 2004; 11: 390–402.
Stankiewicz AR, Lachapelle G, Foo CP, Radicioni SM, Mosser DD . Hsp70 inhibits heat-induced apoptosis upstream of mitochondria by preventing Bax translocation. J Biol Chem 2005; 280: 38729–38739.
Stankiewicz AR, Livingstone AM, Mohseni N, Mosser DD . Regulation of heat-induced apoptosis by Mcl-1 degradation and its inhibition by Hsp70. Cell Death Differ 2009; 16: 638–647.
Beere HM, Wolf BB, Cain K, Mosser DD, Mahboubi A, Kuwana T et al. Heat-shock protein 70 inhibits apoptosis by preventing recruitment of procaspase-9 to the Apaf-1 apoptosome. Nat Cell Biol 2000; 2: 469–475.
Nylandsted J, Brand K, Jaattela M . Heat shock protein 70 is required for the survival of cancer cells. Ann NY Acad Sci 2000; 926: 122–125.
Acknowledgements
We thank Dr G Franceschetto for the statistical analysis; A Cabrelle (Venetian Institute of Molecular Medicine, Padova, Italy) for performing FACS analysis; and E Tosato and A Molin for their technical expertise and support. This work was supported by Fondazione Città della Speranza and MIUR (Ministero Istruzione Università e Ricerca).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Bonvini, P., Zorzi, E., Mussolin, L. et al. Consequences of heat shock protein 72 (Hsp72) expression and activity on stress-induced apoptosis in CD30+ NPM–ALK+ anaplastic large-cell lymphomas. Leukemia 26, 1375–1382 (2012). https://doi.org/10.1038/leu.2011.367
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/leu.2011.367