Abstract
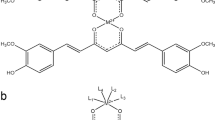
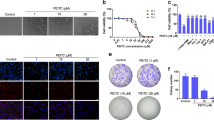
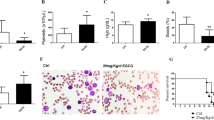
The vitamin E derivative (+)α-tocopheryl succinate (α-TOS) exerts pro-apoptotic effects in a wide range of tumors and is well tolerated by normal tissues. Previous studies point to a mitochondrial involvement in the action mechanism; however, the early steps have not been fully elucidated. In a model of acute promyelocytic leukemia (APL) derived from hCG-PML-RARα transgenic mice, we demonstrated that α-TOS is as effective as arsenic trioxide or all-trans retinoic acid, the current gold standards of therapy. We also demonstrated that α-TOS induces an early dissipation of the mitochondrial membrane potential in APL cells and studies with isolated mitochondria revealed that this action may result from the inhibition of mitochondrial respiratory chain complex I. Moreover, α-TOS promoted accumulation of reactive oxygen species hours before mitochondrial cytochrome c release and caspases activation. Therefore, an in vivo antileukemic action and a novel mitochondrial target were revealed for α-TOS, as well as mitochondrial respiratory complex I was highlighted as potential target for anticancer therapy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Wang Z, Chen Z . Acute promyelocytic leukemia: from highly fatal to highly curable. Blood 2008; 111: 2505–2515.
Sanz MA, Grimwade D, Tallman MS, Lowenberg B, Fenaux P, Estey EH et al. Management of acute promyelocytic leukemia: recommendations from an expert panel on behalf of the European LeukemiaNet. Blood 2009; 113: 1875–1891.
Forghieri F, Luppi M, Morselli M, Favale V, Potenza L, Volzone F et al. Acute promyelocytic leukemia in very elderly patients: still a clinical challenge. Leuk Lymphoma 2009; 50: 119–121.
Tallman M . Treatment of elderly adults with acute promyelocytic leukemia: put the pedal to metal. Leuk Lymphoma 2009; 50: 12–13.
Freitas R, Silva dos Santos G, Gimenes Teixeira H, Scheucher P, Lucena-Araujo A, Lima A et al. Apoptosis induction by (+)alpha-tocopheryl succinate in the absence or presence of all-trans retinoic acid and arsenic trioxide in NB4, NB4-R2 and primary APL cells. Leuk Res 2009; 33: 958–963.
Basu A, Grossie B, Bennett M, Mills N, Imrhan V . Alpha-tocopheryl succinate (alpha-TOS) modulates human prostate LNCaP xenograft growth and gene expression in BALB/c nude mice fed two levels of dietary soybean oil. Eur J Nutr 2007; 46: 34–43.
Quin J, Engle D, Litwiller A, Peralta E, Grasch A, Boley T et al. Vitamin E succinate decreases lung cancer tumor growth in mice. J Surg Res 2005; 127: 139–143.
Neuzil J, Tomasetti M, Zhao Y, Dong L, Birringer M, Wang X et al. Vitamin E analogs, a novel group of ‘mitocans,’ as anticancer agents: the importance of being redox-silent. Mol Pharmacol 2007; 71: 1185–1199.
Jeanne M, Lallemand-Breitenbach V, Ferhi O, Koken M, Le Bras M, Duffort S et al. PML/RARA oxidation and arsenic binding initiate the antileukemia response of As2O3. Cancer Cell 2010; 18: 88–98.
Weber T, Dalen H, Andera L, Nègre-Salvayre A, Augé N, Sticha M et al. Mitochondria play a central role in apoptosis induced by alpha-tocopheryl succinate, an agent with antineoplastic activity: comparison with receptor-mediated pro-apoptotic signaling. Biochemistry 2003; 42: 4277–4291.
Nelson D, Cox M . Lehninger Principles of Biochemistry, Vol. 1. FREEMAN: New York, 2008, 1158pp.
Yamamoto S, Tamai H, Ishisaka R, Kanno T, Arita K, Kobuchi H et al. Mechanism of alpha-tocopheryl succinate-induced apoptosis of promyelocytic leukemia cells. Free Radic Res 2000; 33: 407–418.
Dong L, Low P, Dyason J, Wang X, Prochazka L, Witting P et al. Alpha-tocopheryl succinate induces apoptosis by targeting ubiquinone-binding sites in mitochondrial respiratory complex II. Oncogene 2008; 27: 4324–4335.
Pedersen P, Greenawalt J, Reynafarje B, Hullihen J, Decker G, Soper J et al. Preparation and characterization of mitochondria and submitochondrial particles of rat liver and liver-derived tissues. Methods Cell Biol 1978; 20: 411–481.
Uyemura S, Curti C . Steady-state kinetic properties of FoF1-ATPase: the pH effect. Int J Biochem 1992; 24: 1743–1748.
Trounce I, Kim Y, Jun A, Wallace D . Assessment of mitochondrial oxidative phosphorylation in patient muscle biopsies, lymphoblasts, and transmitochondrial cell lines. Methods Enzymol 1996; 264: 484–509.
Arnoult D . Apoptosis-associated mitochondrial outer membrane permeabilization assays. Methods 2008; 44: 229–234.
He L, Tribioli C, Rivi R, Peruzzi D, Pelicci P, Soares V et al. Acute leukemia with promyelocytic features in PML/RARalpha transgenic mice. Proc Natl Acad Sci USA 1997; 94: 5302–5307.
Rego E, He L, Warrell RJ, Wang Z, Pandolfi P . Retinoic acid (RA) and As2O3 treatment in transgenic models of acute promyelocytic leukemia (APL) unravel the distinct nature of the leukemogenic process induced by the PML-RARalpha and PLZF-RARalpha oncoproteins. Proc Natl Acad Sci USA 2000; 97: 10173–10178.
Weber T, Lu M, Andera L, Lahm H, Gellert N, Fariss M et al. Vitamin E succinate is a potent novel antineoplastic agent with high selectivity and cooperativity with tumor necrosis factor-related apoptosis-inducing ligand (Apo2 ligand) in vivo. Clin Cancer Res 2002; 8: 863–869.
Zamzami N, Marchetti P, Castedo M, Zanin C, Vayssière J, Petit P et al. Reduction in mitochondrial potential constitutes an early irreversible step of programmed lymphocyte death in vivo. J Exp Med 1995; 181: 1661–1672.
Lemasters J, Theruvath T, Zhong Z, Nieminen A . Mitochondrial calcium and the permeability transition in cell death. Biochim Biophys Acta 2009; 1787: 1395–1401.
Wang XF, Xie Y, Wang HG, Zhang Y, Duan XC, Lu ZJ . Tocopheryl succinate induces apoptosis in erbB2-expressing breast cancer cell via NF-κB pathway. Acta Pharmacol Sin 2010; 31: 1604–1610.
Grivennikova V, Vinogradov A . Generation of superoxide by the mitochondrial complex I. Biochim Biophys Acta 2006; 1757: 553–561.
Mukhopadhyay P, Rajesh M, Haskó G, Hawkins BJ, Madesh M, Pacher P . Simultaneous detection of apoptosis and mitochondrial superoxide production in live cells by flow cytometry and confocal microscopy. Nat Protoc 2007; 2: 2295–2301.
Santana B, Pintão M, Abreu e Lima R, Scheucher P, Santos G, Garcia A et al. Asynchronous expression of myeloid antigens in leukemic cells in a PML/RARalpha transgenic mouse model. Braz J Med Biol Res 2006; 39: 615–620.
Le S, Hailer M, Buhrow S, Wang Q, Flatten K, Pediaditakis P et al. Inhibition of mitochondrial respiration as a source of adaphostin-induced reactive oxygen species and cytotoxicity. J Biol Chem 2007; 282: 8860–8872.
Hatefi Y . Resolution of complex II and isolation of succinate dehydrogenase (EC 1.3.99.1). Methods Enzymol 1978; 53: 27–35.
Fariss M, Nicholls-Grzemski F, Tirmenstein M, Zhang J . Enhanced antioxidant and cytoprotective abilities of vitamin E succinate is associated with a rapid uptake advantage in rat hepatocytes and mitochondria. Free Radic Biol Med 2001; 31: 530–541.
Dong L, Freeman R, Liu J, Zobalova R, Marin-Hernandez A, Stantic M et al. Suppression of tumor growth in vivo by the mitocan alpha-tocopheryl succinate requires respiratory complex II. Clin Cancer Res 2009; 15: 1593–1600.
Hirst J, King M, Pryde K . The production of reactive oxygen species by complex I. Biochem Soc Trans 2008; 36 (Pt 5): 976–980.
Martinvalet D, Dykxhoorn D, Ferrini R, Lieberman J . Granzyme A cleaves a mitochondrial complex I protein to initiate caspase-independent cell death. Cell 2008; 133: 681–692.
Ricci J, Muñoz-Pinedo C, Fitzgerald P, Bailly-Maitre B, Perkins G, Yadava N et al. Disruption of mitochondrial function during apoptosis is mediated by caspase cleavage of the p75 subunit of complex I of the electron transport chain. Cell 2004; 117: 773–786.
Urbano A, Lakshmanan U, Choo P, Kwan J, Ng P, Guo K et al. AIF suppresses chemical stress-induced apoptosis and maintains the transformed state of tumor cells. EMBO J 2005; 24: 2815–2826.
Papa S, De Rasmo D, Scacco S, Signorile A, Technikova-Dobrova Z, Palmisano G et al. Mammalian complex I: a regulable and vulnerable pacemaker in mitochondrial respiratory function. Biochim Biophys Acta 2008; 1777: 719–728.
Warburg O, Wind F, Negelein E . The metabolism of tumors in the body. J Gen Physiol 1927; 8: 519–530.
Samudio I, Fiegl M, Andreeff M . Mitochondrial uncoupling and the Warburg effect: molecular basis for the reprogramming of cancer cell metabolism. Cancer Res 2009; 69: 2163–2166.
Campbell A, Chan S . Mitochondrial membrane cholesterol, the voltage dependent anion channel (VDAC), and the Warburg effect. J Bioenerg Biomembr 2008; 40: 193–197.
Bonnet S, Archer S, Allalunis-Turner J, Haromy A, Beaulieu C, Thompson R et al. A mitochondria-K+ channel axis is suppressed in cancer and its normalization promotes apoptosis and inhibits cancer growth. Cancer Cell 2007; 11: 37–51.
Schmidt W, Alam M . Controversies on new animal models of Parkinson's disease pro and con: the rotenone model of Parkinson's disease (PD). J Neural Transm Suppl 2006; 70: 273–276.
Neuzil J, Wang X, Dong L, Low P, Ralph S . Molecular mechanism of ‘mitocan’-induced apoptosis in cancer cells epitomizes the multiple roles of reactive oxygen species and Bcl-2 family proteins. FEBS Lett 2006; 580: 5125–5129.
Kang Y, Lee E, Choi M, Ku J, Kim S, Park Y et al. Role of reactive oxygen species in the induction of apoptosis by alpha-tocopheryl succinate. Int J Cancer 2004; 112: 385–392.
Zhang XW, Yan XJ, Zhou ZR, Yang FF, Wu ZY, Sun HB et al. Arsenic trioxide controls the fate of the PML-RARalpha oncoprotein by directly binding PML. Science 2010; 328: 240–243.
Guibal F, Alberich-Jorda M, Hirai H, Ebralidze A, Levantini E, Di Ruscio A et al. Identification of a myeloid committed progenitor as the cancer initiating cell in acute promyelocytic leukemia. Blood 2009; 114: 5415–5425.
Ravandi F, Estey E, Jones D, Faderl S, O’Brien S, Fiorentino J et al. Effective treatment of acute promyelocytic leukemia with all-trans-retinoic acid, arsenic trioxide, and gemtuzumab ozogamicin. J Clin Oncol 2009; 27: 504–510.
Diaz Z, Colombo M, Mann K, Su H, Smith K, Bohle D et al. Trolox selectively enhances arsenic-mediated oxidative stress and apoptosis in APL and other malignant cell lines. Blood 2005; 105: 1237–1245.
Diaz Z, Laurenzana A, Mann K, Bismar T, Schipper H, Miller WJ . Trolox enhances the anti-lymphoma effects of arsenic trioxide, while protecting against liver toxicity. Leukemia 2007; 21: 2117–2127.
Acknowledgements
We are grateful to AB Garcia and DA Gallo for technical assistance in flow cytometry and to CL Silva and LL Alves for handling of transgenic mice. RSAL, PSS, BAASL, ARLA and FPR received financial support from Fundação de Amparo a Pesquisa do Estado de São Paulo (FAPESP); HLGT and CRP from Coordenação de Aperfeiçoamento de pessoal de nível superior (CAPES). This work was supported by the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq—grant # 573754/2008-0) and Fundação de Apoio à Pesquisa do Estado de São Paulo (FAPESP—grant # 1998/14247-6).
Author contributions
GASS designed and performed research, analyzed data and wrote the paper. RSAL performed research and analyzed data of the ‘in vivo experiments’. BAAS-L performed research of the ‘in vivo experiments’. SAU, PPP and HT contributed vital new reagents or analytical tools and discussed data. CC and EMR designed experiments and wrote the paper. All other co-authors performed experiments and analyzed data.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Leukemia website
Rights and permissions
About this article
Cite this article
dos Santos, G., Abreu e Lima, R., Pestana, C. et al. (+)α-Tocopheryl succinate inhibits the mitochondrial respiratory chain complex I and is as effective as arsenic trioxide or ATRA against acute promyelocytic leukemia in vivo. Leukemia 26, 451–460 (2012). https://doi.org/10.1038/leu.2011.216
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/leu.2011.216
Keywords
This article is cited by
-
Novel inhibitor of hematopoietic cell kinase as a potential therapeutic agent for acute myeloid leukemia
Cancer Immunology, Immunotherapy (2022)
-
Mitochondrial metabolism as a target for acute myeloid leukemia treatment
Cancer & Metabolism (2021)
-
(–)-Epigallocatechin-3-gallate induces apoptosis and differentiation in leukaemia by targeting reactive oxygen species and PIN1
Scientific Reports (2021)
-
α-Tocopherol Acetate Attenuates Mitochondrial Oxygen Consumption and Maintains Primitive Cells within Mesenchymal Stromal Cell Population
Stem Cell Reviews and Reports (2021)
-
Targeting the mitochondria in acute myeloid leukemia
Applied Cancer Research (2017)