Abstract
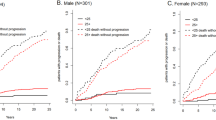
Monoclonal gammopathy of undetermined significance (MGUS) was identified in 3.2% of 21 463 residents of Olmsted County, Minnesota, 50 years of age or older. The risk of progression to multiple myeloma, Waldenstrom's macroglobulinemia, AL amyloidosis or a lymphoproliferative disorder is approximately 1% per year. Low-risk MGUS is characterized by having an M protein <15 g/l, IgG type and a normal free light chain (FLC) ratio. Patients should be followed with serum protein electrophoresis at six months and, if stable, can be followed every 2–3 years or when symptoms suggestive of a plasma cell malignancy arise. Patients with intermediate and high-risk MGUS should be followed in 6 months and then annually for life. The risk of smoldering (asymptomatic) multiple myeloma (SMM) progressing to multiple myeloma or a related disorder is 10% per year for the first 5 years, 3% per year for the next 5 years and 1–2% per year for the next 10 years. Testing should be done 2–3 months after the initial recognition of SMM. If the results are stable, the patient should be followed every 4–6 months for 1 year and, if stable, every 6–12 months.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
International Myeloma Working Group. Criteria for the classification of monoclonal gammopathies, multiple myeloma and related disorders: a report of the International Myeloma Working Group. Br J Haematol 2003; 121: 749–757.
Kyle RA, Therneau TM, Rajkumar SV, Larson DR, Plevak MF, Offord JR et al. Prevalence of monoclonal gammopathy of undetermined significance. N Engl J Med 2006; 354: 1362–1369.
Cohen HJ, Crawford J, Rao MK, Pieper CF, Currie MS . Racial differences in the prevalence of monoclonal gammopathy in a community-based sample of the elderly.[erratum appears in Am J Med 1998 Oct;105(4):362]. Am J Med 1998; 104: 439–444.
Singh J, Dudley Jr AW, Kulig KA . Increased incidence of monoclonal gammopathy of undetermined significance in blacks and its age-related differences with whites on the basis of a study of 397 men and one woman in a hospital setting. J Lab Clin Med 1990; 116: 785–789.
Landgren O, Katzmann JA, Hsing AW, Pfeiffer RM, Kyle RA, Yeboah ED et al. Prevalence of monoclonal gammopathy of undetermined significance among men in Ghana. Mayo Clin Proc 2007; 82: 1468–1473.
Iwanaga M, Tagawa M, Tsukasaki K, Kamihira S, Tomonaga M . Prevalence of monoclonal gammopathy of undetermined significance: study of 52,802 persons in Nagasaki City, Japan. Mayo Clin Proc 2007; 82: 1474–1479.
Landgren O, Kristinsson SY, Goldin LR, Caporaso NE, Blimark C, Mellqvist UH et al. Risk of plasma-cell and lymphoproliferative disorders among 14,621 first-degree relatives of 4,458 patients with monoclonal gammopathy of undetermined significance (MGUS) in Sweden. BLOOD 2009; 114: 791–795.
Vachon CM, Kyle RA, Therneau TM, Foreman BJ, Larson DR, Colby CL et al. Increased risk of monoclonal gammopathy in first-degree relatives of patients with multiple myeloma or monoclonal gammopathy of undetermined significance. Blood 2009; 114: 785–790.
Kyle RA, Therneau TM, Melton III LJ, Dispenzieri A, Larson D, Benson J et al. Monoclonal gammopathy of undetermined significance: estimated incidence and duration prior to recognition. Blood (Abstract) 2007; 110: 79a.
Kyle RA, Therneau TM, Rajkumar SV, Larson DR, Plevak MF, Melton III LJ . Long-term follow-up of 241 patients with monoclonal gammopathy of undetermined significance: the original Mayo Clinic series 25 years later.[see comment]. Mayo Clinic Proc 2004; 79: 859–866.
Kyle RA, Therneau TM, Rajkumar SV, Offord JR, Larson DR, Plevak MF et al. A long-term study of prognosis in monoclonal gammopathy of undetermined significance. [see comment]. New Engl J Med 2002; 346: 564–569.
Rosinol L, Cibeira MT, Montoto S, Rozman M, Esteve J, Filella X et al. Monoclonal gammopathy of undetermined significance: predictors of malignant transformation and recognition of an evolving type characterized by a progressive increase in M protein size. Mayo Clin Proc 2007; 82: 428–434.
Blade J, Lopez-Guillermo A, Rozman C, Cervantes F, Salgado C, Aguilar JL et al. Malignant transformation and life expectancy in monoclonal gammopathy of undetermined significance. Br J Haematol 1992; 81: 391–394.
Cesana C, Klersy C, Barbarano L, Nosari AM, Crugnola M, Pungolino E et al. Prognostic factors for malignant transformation in monoclonal gammopathy of undetermined significance and smoldering multiple myeloma. J Clin Oncol 2002; 20: 1625–1634.
Baldini L, Guffanti A, Cesana BM, Colombi M, Chiorboli O, Damilano I et al. Role of different hematologic variables in defining the risk of malignant transformation in monoclonal gammopathy. Blood 1996; 87: 912–918.
Rajkumar SV, Kyle RA, Therneau TM, Melton III LJ, Bradwell AR, Clark RJ et al. Serum free light chain ratio is an independent risk factor for progression in monoclonal gammopathy of undetermined significance. Blood 2005; 106: 812–817.
Perez-Persona E, Vidriales MB, Mateo G, Garcia-Sanz R, Mateos MV, de Coca AG et al. New criteria to identify risk of progression in monoclonal gammopathy of uncertain significance and smoldering multiple myeloma based on multiparameter flow cytometry analysis of bone marrow plasma cells. Blood 2007; 110: 2586–2592.
Nowakowski GS, Witzig TE, Dingli D, Tracz MJ, Gertz MA, Lacy MQ et al. Circulating plasma cells detected by flow cytometry as a predictor of survival in 302 patients with newly diagnosed multiple myeloma. Blood 2005; 106: 2276–2279.
Anderson KC, Kyle RA, Rajkumar SV, Stewart AK, Weber D, Richardson P . Clinically relevant end points and new drug approvals for myeloma. Leukemia 2008; 22: 231–239.
Kyle RA, Remstein ED, Therneau TM, Dispenzieri A, Kurtin PJ, Hodnefield JM et al. Clinical course and prognosis of smoldering (asymptomatic) multiple myeloma. N Engl J Med 2007; 356: 2582–2590.
Dispenzieri A, Kyle RA, Katzmann JA, Therneau TM, Larson D, Benson J et al. Immunoglobulin free light chain ratio is an independent risk factor for progression of smoldering (asymptomatic) multiple myeloma. Blood 2008; 111: 785–789.
Dimopoulos MA, Moulopoulos LA, Maniatis A, Alexanian R . Solitary plasmacytoma of bone and asymptomatic multiple myeloma. Blood 2000; 96: 2037–2044.
Wang M, Alexanian R, Delasalle K, Weber D . Abnormal MRI of spine is the dominant risk factor for early progression of asymptomatic multiple myeloma. Blood 2003; 102: 687a (abstract).
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Appendix
Appendix
International Myeloma Working Group
Rafat Abonour, Indiana University School of Medicine, Indianapolis, Indiana, USA
Ray Alexanian, MD Anderson, Houston, Texas, USA
Kenneth C Anderson, DFCI, Boston, Massachusetts, USA
Michael Attal, Purpan Hospital, Toulouse, France
Herve Avet-Loiseau, Institute de Biologie, Nantes, France
Ashraf Badros, University of Maryland, Baltimore, Maryland, USA
Bart Barlogie, MIRT UAMS Little Rock, Arkanas, USA
Dalsu Baris, National Cancer Institute, Bethesda, Maryland, USA
Regis Batille, Institute de Biologie, Nantes, France
Meral Beksac, Ankara University, Ankara, Turkey
Andrew Belch, Cross Cancer Institute, Alberta, Canada
Bill Bensinger, Fred Hutchinson Cancer Center, Seattle, Washington, USA
P Leif Bergsagel, Mayo Clinic Scottsdale, Scottsdale, Arizona, USA
Jenny Bird, Bristol Haematology and Oncology Center, Bristol, UK
Joan Bladé, Hospital Clinica, Barcelona, Spain
Mario Boccadoro, University of Torino, Torino, Italy
Michele Cavo, Universita di Bologna, Bologna, Italy
Asher Chanan-Khan, Roswell Park Cancer Institute, Buffalo, New York, USA
Wen Ming Chen, MM Research Center of Beijing, Beijing, China
Tony Child, Leeds General Hospital, Leeds, United Kingdom
James Chim, Department of Medicine, Queen Mary Hospital, Hong Kong
Wee-Joo Chng, National University Health System, Singapore
Ray Comenzo, Tufts Medical School, Boston, Massachusetts, USA
John Crowley, Cancer Research and Biostatistics, Seattle, Washington, USA
William Dalton, H Lee Moffitt, Tampa, Florida, USA
Faith Davies, Royal Marsden Hospital, London, England
Cármino de Souza, Univeridade de Campinas, Caminas, Brazil
Michel Delforge, University Hospital Gasthuisberg, Leuven, Belgium
Meletios Dimopoulos, University of Athens School of Medicine, Athens, Greece
Angela Dispenzieri, Mayo Clinic, Rochester, Minnesota, USA
Brian GM Durie, Cedars-Sinai Outpatient Cancer Center, Los Angeles, California, USA
Johannes Drach, University of Vienna, Vienna, Austria
Hermann Einsele, Universitätsklinik Würzburg, Würzburg, Germany
Theirry Facon, Centre Hospitalier Regional Universitaire de Lille, Lille, France
Dorotea Fantl, Socieded Argentinade Hematolgia, Buenos Aires, Argentina
Jean-Paul Fermand, Hopitaux de Paris, Paris, France
Rafael Fonseca, Mayo Clinic Arizona, Scottsdale, Arizona, USA
Gösta Gahrton, Karolinska Institute for Medicine, Huddinge, Sweden
Ramon Garcia-Sanz, University Hospital of Salamanca, Salamanca, Spain
Christina Gasparetto, Duke University Medical Center, Durham, North Carolina, USA
Morie Gertz, Mayo Clinic, Rochester, Minnesota, USA
John Gibson, Royal Prince Alfred Hospital, Sydney, Australia
Sergio Giralt, MD Anderson Cancer Center, Houston, Texas, USA
Hartmut Goldschmidt, University Hospital Heidelberg, Heidelberg, Germany
Philip Greipp, Mayo Clinic, Rochester, Minnesota, USA
Roman Hajek, Brno University, Brno, Czech Republic
Izhar Hardan, Tel Aviv University, Tel Aviv, Israel
Jean-Luc Harousseau, Institute de Biologie, Nantes, France
Hiroyuki Hata, Kumamoto University Hospital, Kumamoto, Japan
Yutaka Hattori, Keio University School of Medicine, Tokyo, Japan
Tom Heffner, Emory University, Atlanta, Georgia, USA
Joy Ho, Royal Prince Alfred Hospital, Sydney, Australia
Vania Hungria, Clinica San Germano, Sao Paolo, Brazil
Shinsuke Ida, Nagoya City University Medical School, Nagoya, Japan
Peter Jacobs, Constantiaberg Medi-Clinic, Plumstead, South Africa
Sundar Jagannath, St Vincent's Comprehensive Cancer Center, New York, New York, USA
Hou Jian, Shanghai Chang Zheng Hospital, Shanghai, China
Douglas Joshua, Royal Prince Alfred Hospital, Sydney, Australia
Artur Jurczyszyn, The Myeloma Treatment Foundation, Poland
Michio Kawano, Yamaguchi University, Ube, Japan
Nicolaus Kröger, University Hospital Hamburg, Hamburg, Germany
Shaji Kumar, Department of Hematology, Mayo Clinic, Minnesota, USA
Robert A Kyle, Department of Laboratory Med. and Pathology, Mayo Clinic, Minnesota, USA
Juan Lahuerta, Grupo Espanol di Mieloma, Hospital Universitario, Madrid, Spain
Ola Landgren, National Cancer Institute, Bethesda, Maryland, USA
Jacob Laubach, Dana-Farber Cancer Institute, Boston, Massachusetts, USA
Jae Hoon Lee, Gachon University Gil Hospital, Incheon, Korea
Xavier LeLeu, Hospital Huriez, CHRU Lille, France
Suzanne Lentzsch, University of Pittsburgh, Pittsburgh, Pennsylvania, USA
Henk Lokhorst, University Medical CenterUtrecht, Utrecht, The Netherlands
Sagar Lonial, Emory University Medical School, Atlanta, Georgia, USA
Heinz Ludwig, Wilhelminenspital Der Stat Wien, Vienna, Austria
Angelo Maiolino, Rua fonte da Saudade, Rio de Janeiro, Brazil
Maria Mateos, University of Salamanca, Salamanca, Spain
Jayesh Mehta, Northwestern University, Chicago, Illinois, USA
Ulf-Henrik Mellqvist, Sahlgrenska University Hospital, Gothenburg, Sweden
GiamPaolo Merlini, University of Pavia, Pavia, Italy
Joseph Mikhael, Mayo Clinic Arizona, Scottsdale, Arizona, USA
Angelina Rodriquez Morales, Bonco Metro Politano de Sangre, Caracas, Venezuela
Philippe Moreau, University Hospital, Nantes, France
Gareth Morgan, Royal Marsden Hospital, London, England
Nikhil Munshi, Diane Farber Cancer Institute, Boston, Massachusetts, USA
Ruben Niesvizky, Weill Medical College of Cornell University, New York, New York, USA
Amara Nouel, Hospital Rutz y Paez, Bolivar, Venezuela
Yana Novis, Hospital SírioLibanês, Bela Vista, Brazil
Robert Orlowski, MD Anderson Cancer Center, Houston, Texas, USA
Antonio Palumbo, Cathedra Ematologia, Torino, Italy
Santiago Pavlovsky, Fundaleu, Buenos Aires, Argentina
Linda Pilarski, University of Alberta, Alberta, Canada
Raymond Powles, Leukemia & Myeloma, Wimbledon, England
S Vincent Rajkumar, Mayo Clinic, Rochester, Minnesota, USA
Donna Reece, Princess Margaret Hospital, Toronto, Canada
Tony Reiman, Cross Cancer Institute, Alberta, Canada
Paul G Richardson, Dana Farber Cancer Institute, Boston, Massachusetts, USA
David Roodman, University of Pittsburgh School of Medicine, Pittsburgh, Pennsylvania USA
Laura Rosinol, Hospital Clinic, Barcelona, Spain
Jesus San Miguel, University of Salamanca, Salamanca, Spain
Orhan Sezer, Universitätsklinik Würzburg, Würzburg, Germany
Jatin J Shah, MD Anderson Cancer Institute, Houston, Texas, USA
John Shaughnessy, MIRT UAMS, Little Rock, Arkansas, USA
Kazuyuki Shimizu, Nagoya City Midori General Hospital, Nagoya, Japan
Chaim Shustik, McGill University, Montreal, Canada
David Siegel, Hackensack, Cancer Center, Hackensack, New Jersey, USA
Seema Singhal, Northwestern University, Chicago, Illinois, USA
Pieter Sonneveld, Erasmus MC, Rotterdam, The Netherlands
Andrew Spencer, The Alfred Hospital, Melbourne, Australia
Edward Stadtmauer, University of Pennsylvania, Philadelphia, Pennsylvania, USA
Keith Stewart, Mayo Clinic Arizona, Scottsdale, Arizona, USA
Evangelos Terpos, University of Athens School of Medicine, Athens, Greece
Patrizia Tosi, Italian Cooperative Group, Istituto di Ematologia Seragnoli, Bologna, Italy
Guido Tricot, Huntsman Cancer Institute, Salt Lake City, Utah, USA
Ingemar Turesson, SKANE University Hospital, Malmo, Sweden
Karin Vanderkerken, Vrije University Brussels VUB, Brussels, Belgium
Brian Van Ness, University of Minnesota, Minneapolis, Minnesota, USA
Ivan Van Riet, Brussels Vrija University, Brussels, Belgium
Robert Vescio, Cedars-Sinai Cancer Center, Los Angeles, California, USA
David Vesole, Hackensack Cancer Center, Hackensack, New Jersey, USA
Anders Waage, University Hospital, Trondheim, Norway NSMG
Michael Wang, MD Anderson, Houston, Texas, USA
Donna Weber, MD Anderson, Houston, Texas, USA
Jan Westin, Sahlgrenska University Hospital, Gothenburg, Sweden
Keith Wheatley, University of Birmingham, Birmingham, United Kingdom
Dina B Yehuda, Department of Hematology, Hadassah University Hospital, Hadassah, Israel
Jeffrey Zonder, Karmanos Cancer Institute, Detroit, Michigan, USA.
Rights and permissions
About this article
Cite this article
Kyle, R., Durie, B., Rajkumar, S. et al. Monoclonal gammopathy of undetermined significance (MGUS) and smoldering (asymptomatic) multiple myeloma: IMWG consensus perspectives risk factors for progression and guidelines for monitoring and management. Leukemia 24, 1121–1127 (2010). https://doi.org/10.1038/leu.2010.60
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/leu.2010.60
Keywords
This article is cited by
-
Oral health, dental treatment, and medication related osteonecrosis of the jaw in multiple myeloma – a longitudinal cohort study
BMC Oral Health (2024)
-
A gene signature can predict risk of MGUS progressing to multiple myeloma
Journal of Hematology & Oncology (2023)
-
Artificial intelligence-enabled screening strategy for drug repurposing in monoclonal gammopathy of undetermined significance
Blood Cancer Journal (2023)
-
Single-cell RNA sequencing in orthopedic research
Bone Research (2023)
-
Disentangling age, gender, and racial/ethnic disparities in multiple myeloma burden: a modeling study
Nature Communications (2023)