Abstract
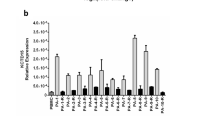
The main reason for the unfavorable clinical outcome of BCR–ABL1-positive acute lymphoblastic leukemia (ALL) is genetic instability. However, how normal B-cell precursors acquire the genetic changes that lead to transformation has not yet been completely defined. We investigated the expression of the activation-induced cytidine deaminase (AID) and its role in clinical outcome in 61 adult BCR–ABL1-positive ALL patients. AID expression was detected in 36 patients (59%); it correlated with the BCR–ABL1 transcript levels and disappeared after treatment with tyrosine kinase inhibitors. Different AID splice variants were identified: full-length isoform; AIDΔE4a, with a 30-bp deletion of exon 4; AIDΔE4, with the exon 4 deletion; AIDins3, with the retention of intron 3; AIDΔE3-E4 isoform without deaminase activity. AID-FL predominantly showed cytoplasmic localization, as did the AID-ΔE4a and AID-ΔE3E4 variants, whereas the C-terminal-truncated AID-ΔE4 showed a slightly increased nuclear localization pattern. AID expression correlated with a higher number of copy number alterations identified in genome-wide analysis using a single-nucleotide polymorphism array. However, the expression of AID at diagnosis was not associated with a worse prognosis. In conclusion, BCR–ABL1-positive ALL cells aberrantly express different isoforms of AID that may act as mutators outside the immunoglobulin (Ig) gene loci in promoting genetic instability.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Accession codes
References
Radich JP . Philadelphia chromosome-positive acute lymphocytic leukemia. Hematol Oncol Clin North Am 2001; 15: 21–36.
Faderl S, Jeha S, Kantarjian HM . The biology and therapy of adult acute lymphoblastic leukemia. Cancer 2003; 98: 1337–1354.
Mullighan CG, Miller CB, Radtke I, Phillips LA, Dalton J, Ma J et al. BCR-ABL1 lymphoblastic leukaemia is characterized by the deletion of Ikaros. Nature 2008; 453: 110–114.
Itoh K, Friel J, Kluge N, Kina T, Kondo-Takaori A, Kawamata S et al. A novel hematopoietic multilineage clone, Myl-D-7, is stromal cell-dependent and supported by an alternative mechanism(s) independent of stem cell factor/c-kit interaction. Blood 1996; 87: 3218–3228.
Paulsson K, Cazier JB, Macdougall F, Stevens J, Stasevich I, Vrcelj N et al. Microdeletions are a general feature of adult and adolescent acute lymphoblastic leukemia: Unexpected similarities with pediatric disease. Proc Natl Acad Sci USA 2008; 105: 6708–6713.
Feldhahn N, Henke N, Melchior K, Duy C, Soh BN, Klein F et al. Activation-induced cytidine deaminase acts as a mutator in BCR-ABL1-transformed acute lymphoblastic leukemia cells. J Exp Med 2007; 204: 1157–1166.
Muramatsu M, Sankaranand VS, Anant S, Sugai M, Kinoshita K, Davidson NO et al. Specific expression of activation-induced cytidine deaminase (AID), a novel member of the RNA-editing deaminase family in germinal center B cells. J Biol Chem 1999; 274: 18470–18476.
Honjo T, Kinoshita K, Muramatsu M . Molecular mechanism of class switch recombination: linkage with somatic hypermutation. Annu Rev Immunol 2002; 20: 165–196.
Muramatsu M, Kinoshita K, Fagarasan S, Yamada S, Shinkai Y, Honjo T . Class switch recombination and hypermutation require activation-induced cytidine deaminase (AID), a potential RNA editing enzyme. Cell 2000; 102: 553–563.
Revy P, Muto T, Levy Y, Geissmann F, Plebani A, Sanal O et al. Activation-induced cytidine deaminase (AID) deficiency causes the autosomal recessive form of the Hyper-IgM syndrome (HIGM2). Cell 2000; 102: 565–575.
Peled JU, Kuang FL, Iglesias-Ussel MD, Roa S, Kalis SL, Goodman MF et al. The biochemistry of somatic hypermutation. Annu Rev Immunol 2008; 26: 481–511.
Di Noia JM, Neuberger MS . Molecular mechanisms of antibody somatic hypermutation. Annu Rev Biochem 2007; 76: 1–22.
Pasqualucci L, Guglielmino R, Houldsworth J, Mohr J, Aoufouchi S, Polakiewicz R et al. Expression of the AID protein in normal and neoplastic B cells. Blood 2004; 104: 3318–3325.
Greeve J, Philipsen A, Krause K, Klapper W, Heidorn K, Castle BE et al. Expression of activation-induced cytidine deaminase in human B-cell non-Hodgkin lymphomas. Blood 2003; 101: 3574–3580.
Liu M, Duke JL, Richter DJ, Vinuesa CG, Goodnow CC, Kleinstein SH et al. Two levels of protection for the B cell genome during somatic hypermutation. Nature 2008; 451: 841–845.
Albesiano E, Messmer BT, Damle RN, Allen SL, Rai KR, Chiorazzi N . Activation-induced cytidine deaminase in chronic lymphocytic leukemia B cells: expression as multiple forms in a dynamic, variably sized fraction of the clone. Blood 2003; 102: 3333–3339.
Babbage G, Garand R, Robillard N, Zojer N, Stevenson FK, Sahota SS . Mantle cell lymphoma with t(11;14) and unmutated or mutated VH genes expresses AID and undergoes isotype switch events. Blood 2004; 103: 2795–2798.
Wu X, Darce JR, Chang SK, Nowakowski GS, Jelinek DF . Alternative splicing regulates activation-induced cytidine deaminase (AID): implications for suppression of AID mutagenic activity in normal and malignant B cells. Blood 2008; 112: 4675–4682.
Palandri F, Iacobucci I, Martinelli G, Amabile M, Poerio A, Testoni N et al. Long-term outcome of complete cytogenetic responders after imatinib 400 mg in late chronic phase, philadelphia-positive chronic myeloid leukemia: the GIMEMA Working Party on CML. J Clin Oncol 2008; 26: 106–111.
Esperou H, Boiron JM, Cayuela JM, Blanchet O, Kuentz M, Jouet JP et al. A potential graft-versus-leukemia effect after allogeneic hematopoietic stem cell transplantation for patients with Philadelphia chromosome-positive acute lymphoblastic leukemia: results from the French Bone Marrow Transplantation Society. Bone Marrow Transplant 2003; 31: 909–918.
Iacobucci I, Saglio G, Rosti G, Testoni N, Pane F, Amabile M et al. Achieving a major molecular response at the time of a complete cytogenetic response (CCgR) predicts a better duration of CCgR in imatinib-treated chronic myeloid leukemia patients. Clin Cancer Res 2006; 12: 3037–3042.
Iacobucci I, Lonetti A, Messa F, Cilloni D, Arruga F, Ottaviani E et al. Expression of spliced oncogenic Ikaros isoforms in Philadelphia-positive acute lymphoblastic leukemia patients treated with tyrosine kinase inhibitors: implications for a new mechanism of resistance. Blood 2008; 112: 3847–3855.
Kaplan E, Meier P . Non parametric estimation from incomplete observation. J Am Stat Assoc 1958; 53: 457–481.
Gooley TA, Leisenring W, Crowley J, Storer B . Estimation of failure probabilities in the presence of competing risks: new representations of old estimators. Stat Med 1999; 18: 695–706.
Simon R, Lee Y . Nonparametric confidence limits survival probabilities and median survival time. Cancer Treat Res 1982; 66: 37–42.
Ito S, Nagaoka H, Shinkura R, Begum N, Muramatsu M, Nakata M et al. Activation-induced cytidine deaminase shuttles between nucleus and cytoplasm like apolipoprotein B mRNA editing catalytic polypeptide 1. Proc Natl Acad Sci USA 2004; 101: 1975–1980.
Wu X, Geraldes P, Platt JL, Cascalho M . The double-edged sword of activation-induced cytidine deaminase. J Immunol 2005; 174: 934–941.
Marr S, Morales H, Bottaro A, Cooper M, Flajnik M, Robert J . Localization and differential expression of activation-induced cytidine deaminase in the amphibian Xenopus upon antigen stimulation and during early development. J Immunol 2007; 179: 6783–6789.
Larijani M, Frieder D, Sonbuchner TM, Bransteitter R, Goodman MF, Bouhassira EE et al. Methylation protects cytidines from AID-mediated deamination. Mol Immunol 2005; 42: 599–604.
Vignetti M, Fazi P, Cimino G, Martinelli G, Di Raimondo F, Ferrara F et al. Imatinib plus steroids induces complete remissions and prolonged survival in elderly Philadelphia chromosome-positive patients with acute lymphoblastic leukemia without additional chemotherapy: results of the Gruppo Italiano Malattie Ematologiche dell′Adulto (GIMEMA) LAL0201-B protocol. Blood 2007; 109: 3676–3678.
Foà R, Vitale A, Meloni G, Guarini A, De Propris S, Elia L et al. Dasatinib as Front-Line Monotherapy for the Induction Treatment of Adult and Elderly Ph+ Acute Lymphoblastic Leukemia (ALL) Patients: Interim Analysis of the GIMEMA Prospective Study LAL1205. Blood 2007; 110: 10a.
Foà R, Vitale A, Guarini A, De Propris MS, Elia L, Cimino G et al. Dasatinib monotherapy as 1st line treatment of Ph acute lymphoblastic leukemia (ALL) patients: update of Gimema LAL1205 study. Haematologica 2008; 93 (s1): 367.
Wang CL, Harper RA, Wabl M . Genome-wide somatic hypermutation. Proc Natl Acad Sci USA 2004; 101: 7352–7356.
Franco S, Gostissa M, Zha S, Lombard DB, Murphy MM, Zarrin AA et al. H2AX prevents DNA breaks from progressing to chromosome breaks and translocations. Mol Cell 2006; 21: 201–214.
Chen KM, Harjes E, Gross PJ, Fahmy A, Lu Y, Shindo K et al. Structure of the DNA deaminase domain of the HIV-1 restriction factor APOBEC3G. Nature 2008; 452: 116–119.
Williams RT, Roussel MF, Sherr CJ . Arf gene loss enhances oncogenicity and limits imatinib response in mouse models of Bcr-Abl-induced acute lymphoblastic leukemia. Proc Natl Acad Sci USA 2006; 103: 6688–6693.
Acknowledgements
This work was supported by AIL, European LeukaemiaNet, AIRC, Fondazione Del Monte di Bologna e Ravenna, FIRB 2006, Ateneo 60% grants and Gimema Onlus Working Party ALL and CML. We specially thank Serena Formica (University of Bologna) and Annalisa Astolfi (University of Bologna) for help in performing SNP arrays, and Barbara Lama (University of Bologna) for collecting clinical data. We also acknowledge all the people who took care of the patients involved in the GIMEMA studies (see Appendix in Supplementary Information). Conception and design II, MB, GM; Provision of study materials or patients, CP, AV, FP; PPP; SP, GP, RF; Collection and assembly of data, AL; AF; SS; FA; SC; MM; FP, MV; Data analysis and interpretation II, DC, FM, AB, EO; Paper writing II; final approval of paper, GM.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on the Leukemia website (http://www.nature.com/leu)
Rights and permissions
About this article
Cite this article
Iacobucci, I., Lonetti, A., Messa, F. et al. Different isoforms of the B-cell mutator activation-induced cytidine deaminase are aberrantly expressed in BCR–ABL1-positive acute lymphoblastic leukemia patients. Leukemia 24, 66–73 (2010). https://doi.org/10.1038/leu.2009.197
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/leu.2009.197
Keywords
This article is cited by
-
Activation-induced deaminase and its splice variants associate with trisomy 12 in chronic lymphocytic leukemia
Annals of Hematology (2019)
-
Eye on the B-ALL: B-cell receptor repertoires reveal persistence of numerous B-lymphoblastic leukemia subclones from diagnosis to relapse
Leukemia (2016)
-
Variation in CDKN2A at 9p21.3 influences childhood acute lymphoblastic leukemia risk
Nature Genetics (2010)