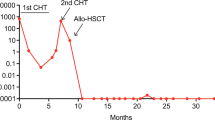
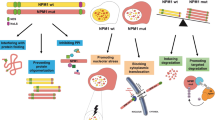
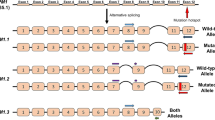
Abstract
Nucleophosmin (NPM1) is a highly conserved nucleo-cytoplasmic shuttling protein that shows a restricted nucleolar localization. Mutations of NPM1 gene leading to aberrant cytoplasmic dislocation of nucleophosmin (NPMc+) occurs in about one third of acute myeloid leukaemia (AML) patients that exhibit distinctive biological and clinical features. We discuss the latest advances in the molecular basis of nucleophosmin traffic under physiological conditions, describe the molecular abnormalities underlying altered transport of nucleophosmin in NPM1-mutated AML and present evidences supporting the view that cytoplasmic nucleophosmin is a critical event for leukaemogenesis. We then outline how a highly specific immunohistochemical assay can be exploited to diagnose NPM1-mutated AML and myeloid sarcoma in paraffin-embedded samples by looking at aberrant nucleophosmin accumulation in cytoplasm of leukaemic cells. This procedure is also suitable for detection of haemopoietic multilineage involvement in bone marrow trephines. Moreover, use of immunohistochemistry as surrogate for molecular analysis can serve as first-line screening in AML and should facilitate implementation of the 2008 World Health Organization classification of myeloid neoplasms that now incorporates AML with mutated NPM1 (synonym: NPMc+ AML) as a new provisional entity. Finally, we discuss the future therapeutic perspectives aimed at reversing the altered nucleophosmin transport in AML with mutated NPM1.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Accession codes
References
Frehlick LJ, Eirin-Lopez JM, Ausio J . New insights into the nucleophosmin/nucleoplasmin family of nuclear chaperones. Bioessays 2007; 29: 49–59.
Chan WY, Liu QR, Borjigin J, Busch H, Rennert OM, Tease LA et al. Characterization of the cDNA encoding human nucleophosmin and studies of its role in normal and abnormal growth. Biochemistry 1989; 28: 1033–1039.
Grisendi S, Bernardi R, Rossi M, Cheng K, Khandker L, Manova K et al. Role of nucleophosmin in embryonic development and tumorigenesis. Nature 2005; 437: 147–153.
Falini B, Nicoletti I, Bolli N, Martelli MP, Liso A, Gorello P et al. Translocations and mutations involving the nucleophosmin (NPM1) gene in lymphomas and leukemias. Haematologica 2007; 92: 519–532.
Wang D, Umekawa H, Olson MO . Expression and subcellular locations of two forms of nucleolar protein B23 in rat tissues and cells. Cell Mol Biol Res 1993; 39: 33–42.
Spector DL, Ochs RL, Busch H . Silver staining, immunofluorescence, and immunoelectron microscopic localization of nucleolar phosphoproteins B23 and C23. Chromosoma 1984; 90: 139–148.
Cordell JL, Pulford KA, Bigerna B, Roncador G, Banham A, Colombo E et al. Detection of normal and chimeric nucleophosmin in human cells. Blood 1999; 93: 632–642.
Dalenc F, Drouet J, Ader I, Delmas C, Rochaix P, Favre G et al. Increased expression of a COOH-truncated nucleophosmin resulting from alternative splicing is associated with cellular resistance to ionizing radiation in HeLa cells. Int J Cancer 2002; 100: 662–668.
Borer RA, Lehner CF, Eppenberger HM, Nigg EA . Major nucleolar proteins shuttle between nucleus and cytoplasm. Cell 1989; 56: 379–390.
Yun JP, Chew EC, Liew CT, Chan JY, Jin ML, Ding MX et al. Nucleophosmin/B23 is a proliferate shuttle protein associated with nuclear matrix. J Cell Biochem 2003; 90: 1140–1148.
Grisendi S, Mecucci C, Falini B, Pandolfi PP . Nucleophosmin and cancer. Nat Rev Cancer 2006; 6: 493–505.
Hingorani K, Szebeni A, Olson MO . Mapping the functional domains of nucleolar protein B23. J Biol Chem 2000; 275: 24451–24457.
Wang W, Budhu A, Forgues M, Wang XW . Temporal and spatial control of nucleophosmin by the Ran-Crm1 complex in centrosome duplication. Nat Cell Biol 2005; 7: 823–830.
Yu Y, Maggi Jr LB, Brady SN, Apicelli AJ, Dai MS, Lu H et al. Nucleophosmin is essential for ribosomal protein L5 nuclear export. Mol Cell Biol 2006; 26: 3798–3809.
Bogerd HP, Fridell RA, Benson RE, Hua J, Cullen BR . Protein sequence requirements for function of the human T-cell leukemia virus type 1 Rex nuclear export signal delineated by a novel in vivo randomization-selection assay. Mol Cell Biol 1996; 16: 4207–4214.
Kau TR, Way JC, Silver PA . Nuclear transport and cancer: from mechanism to intervention. Nat Rev Cancer 2004; 4: 106–117.
Daelemans D, Costes SV, Lockett S, Pavlakis GN . Kinetic and molecular analysis of nuclear export factor CRM1 association with its cargo in vivo. Mol Cell Biol 2005; 25: 728–739.
Misteli T . Physiological importance of RNA and protein mobility in the cell nucleus. Histochem Cell Biol 2008; 129: 5–11.
Muro E, Hoang TQ, Jobart-Malfait A, Hernandez-Verdun D . In nucleoli, the steady state of nucleolar proteins is leptomycin B-sensitive. Biol Cell 2008; 100: 303–313.
Lam YW, Trinkle-Mulcahy L, Lamond AI . The nucleolus. J Cell Sci 2005; 118: 1335–1337.
Ahmad Y, Boisvert FM, Gregor P, Cobley A, Lamond AI . NOPdb: nucleolar proteome database-2008 update. Nucleic Acids Res 2009; 37 (Database issue): D181–D184.
Grummitt CG, Townsley FM, Johnson CM, Warren AJ, Bycroft M . Structural consequences of nucleophosmin mutations in acute myeloid leukemia. J Biol Chem 2008; 283: 23326–23332.
Wu B, Yee A, Huang YJ, Ramelot TA, Cort JR, Semesi A et al. The solution structure of ribosomal protein S17E from Methanobacterium thermoautotrophicum: a structural homolog of the FF domain. Protein Sci 2008; 17: 583–588.
Nishimura Y, Ohkubo T, Furuichi Y, Umekawa H . Tryptophans 286 and 288 in the C-terminal region of protein B23.1 are important for its nucleolar localization. Biosci Biotechnol Biochem 2002; 66: 2239–2242.
Falini B, Bolli N, Shan J, Martelli MP, Liso A, Pucciarini A et al. Both carboxy-terminus NES motif and mutated tryptophan(s) are crucial for aberrant nuclear export of nucleophosmin leukemic mutants in NPMc+ AML. Blood 2006; 107: 4514–4523.
Falini B, Mecucci C, Tiacci E, Alcalay M, Rosati R, Pasqualucci L et al. Cytoplasmic nucleophosmin in acute myelogenous leukemia with a normal karyotype. N Engl J Med 2005; 352: 254–266.
Bolli N, De Marco MF, Martelli MP, Bigerna B, Pucciarini A, Rossi R et al. A dose-dependent tug of war involving the NPM1 leukaemic mutant, nucleophosmin, and ARF. Leukemia 2009; 23: 501–509.
Chan PK, Chan FY . Nucleophosmin/B23 (NPM) oligomer is a major and stable entity in HeLa cells. Biochim Biophys Acta 1995; 1262: 37–42.
Yung BY, Chan PK . Identification and characterization of a hexameric form of nucleolar phosphoprotein B23. Biochim Biophys Acta 1987; 925: 74–82.
Wanzel M, Russ AC, Kleine-Kohlbrecher D, Colombo E, Pelicci PG, Eilers M . A ribosomal protein L23-nucleophosmin circuit coordinates Mizl function with cell growth. Nat Cell Biol 2008; 10: 1051–1061.
Wulff JE, Siegrist R, Myers AG . The natural product avrainvillamide binds to the oncoprotein nucleophosmin. J Am Chem Soc 2007; 129: 14444–14451.
Okuwaki M . The structure and functions of NPM1/nucleophosmin/B23, a multifunctional nucleolar acidic protein. J Biochem 2008; 143: 441–448.
Okuwaki M, Tsujimoto M, Nagata K . The RNA binding activity of a ribosome biogenesis factor, nucleophosmin/B23, is modulated by phosphorylation with a cell cycle-dependent kinase and by association with its subtype. Mol Biol Cell 2002; 13: 2016–2030.
Tarapore P, Shinmura K, Suzuki H, Tokuyama Y, Kim SH, Mayeda A et al. Thr199 phosphorylation targets nucleophosmin to nuclear speckles and represses pre-mRNA processing. FEBS Lett 2006; 580: 399–409.
Negi SS, Olson MO . Effects of interphase and mitotic phosphorylation on the mobility and location of nucleolar protein B23. J Cell Sci 2006; 119: 3676–3685.
Endo A, Matsumoto M, Inada T, Yamamoto A, Nakayama KI, Kitamura N et al. Nucleolar structure and function are regulated by the deubiquitylating enzyme USP36. J Cell Sci 2009; 122: 678–686.
Liu X, Liu Z, Jang SW, Ma Z, Shinmura K, Kang S et al. Sumoylation of nucleophosmin/B23 regulates its subcellular localization, mediating cell proliferation and survival. Proc Natl Acad Sci USA 2007; 104: 9679–9684.
Cheutin T, O'Donohue MF, Beorchia A, Vandelaer M, Kaplan H, Defever B et al. Three-dimensional organization of active rRNA genes within the nucleolus. J Cell Sci 2002; 115: 3297–3307.
Sirri V, Urcuqui-Inchima S, Roussel P, Hernandez-Verdun D . Nucleolus: the fascinating nuclear body. Histochem Cell Biol 2008; 129: 13–31.
Maggi Jr LB, Kuchenruether M, Dadey DY, Schwope RM, Grisendi S, Townsend RR et al. Nucleophosmin serves as a rate-limiting nuclear export chaperone for the mammalian ribosome. Mol Cell Biol 2008; 28: 7050–7065.
Bolli N, Nicoletti I, De Marco MF, Bigerna B, Pucciarini A, Mannucci R et al. Born to be exported: COOH-terminal nuclear export signals of different strength ensure cytoplasmic accumulation of nucleophosmin leukemic mutants. Cancer Res 2007; 67: 6230–6237.
Nozawa Y, Van Belzen N, Van der Made AC, Dinjens WN, Bosman FT . Expression of nucleophosmin/B23 in normal and neoplastic colorectal mucosa. J Pathol 1996; 178: 48–52.
Subong EN, Shue MJ, Epstein JI, Briggman JV, Chan PK, Partin AW . Monoclonal antibody to prostate cancer nuclear matrix protein (PRO:4-216) recognizes nucleophosmin/B23. Prostate 1999; 39: 298–304.
Liso A, Bogliolo A, Freschi V, Martelli MP, Pileri SA, Santodirocco M et al. In human genome, generation of a nuclear export signal through duplication appears unique to nucleophosmin (NPM1) mutations and is restricted to AML. Leukemia 2008; 22: 1285–1289.
Falini B, Martelli MP, Bolli N, Bonasso R, Ghia E, Pallotta MT et al. Immunohistochemistry predicts nucleophosmin (NPM) mutations in acute myeloid leukemia. Blood 2006; 108: 1999–2005.
Falini B, Mason DY . Proteins encoded by genes involved in chromosomal alterations in lymphoma and leukemia: clinical value of their detection by immunocytochemistry. Blood 2002; 99: 409–426.
Delsol G, Falini B, Muller-Hermelink HK, Campo E, Jaffe ES, Gascoyne RD et al. Anaplastic large cell lymphoma (ALCL), ALK-positive. In: Swerdlow S et al. (eds). WHO Classification of Tumours of Haematopoietic and Lymphoid Tissues. IARC: Lyon, 2008, pp 312–316.
Morris SW, Kirstein MN, Valentine MB, Dittmer KG, Shapiro DN, Saltman DL et al. Fusion of a kinase gene, ALK, to a nucleolar protein gene, NPM, in non-Hodgkin's lymphoma. Science 1994; 263: 1281–1284.
Falini B, Pulford K, Pucciarini A, Carbone A, De Wolf-Peeters C, Cordell J et al. Lymphomas expressing ALK fusion protein(s) other than NPM-ALK. Blood 1999; 94: 3509–3515.
Yoneda-Kato N, Look AT, Kirstein MN, Valentine MB, Raimondi SC, Cohen KJ et al. The t(3;5)(q25.1;q34) of myelodysplastic syndrome and acute myeloid leukemia produces a novel fusion gene, NPM-MLF1. Oncogene 1996; 12: 265–275.
Falini B, Bigerna B, Pucciarini A, Tiacci E, Mecucci C, Morris SW et al. Aberrant subcellular expression of nucleophosmin and NPM-MLF1 fusion protein in acute myeloid leukaemia carrying t(3;5): a comparison with NPMc+ AML. Leukemia 2006; 20: 368–371.
Redner RL, Rush EA, Faas S, Rudert WA, Corey SJ . The t(5;17) variant of acute promyelocytic leukemia expresses a nucleophosmin-retinoic acid receptor fusion. Blood 1996; 87: 882–886.
Hummel JL, Wells RA, Dube ID, Licht JD, Kamel-Reid S . Deregulation of NPM and PLZF in a variant t(5;17) case of acute promyelocytic leukemia. Oncogene 1999; 18: 633–641.
Grimwade D, Biondi A, Mozziconacci MJ, Hagemeijer A, Berger R, Neat M et al. Characterization of acute promyelocytic leukemia cases lacking the classic t(15;17): results of the European Working Party. Groupe Francais de Cytogenetique Hematologique, Groupe de Francais d'Hematologie Cellulaire, UK Cancer Cytogenetics Group and BIOMED 1 European Community-Concerted Action ‘Molecular Cytogenetic Diagnosis in Haematological Malignancies’. Blood 2000; 96: 1297–1308.
Sportoletti P, Grisendi S, Majid SM, Cheng K, Clohessy JG, Viale A et al. Npm1 is a haploinsufficient suppressor of myeloid and lymphoid malignancies in the mouse. Blood 2008; 111: 3859–3862.
Ley TJ, Mardis ER, Ding L, Fulton B, McLellan MD, Chen K et al. DNA sequencing of a cytogenetically normal acute myeloid leukaemia genome. Nature 2008; 456: 66–72.
Nakagawa M, Kameoka Y, Suzuki R . Nucleophosmin in acute myelogenous leukemia (letter). N Engl J Med 2005; 352: 1819–1820.
Falini B, Nicoletti I, Martelli MF, Mecucci C . Acute myeloid leukemia carrying cytoplasmic/mutated nucleophosmin (NPMc+ AML): biologic and clinical features. Blood 2007; 109: 874–885.
Jeong EG, Lee SH, Yoo NJ . Absence of nucleophosmin 1 (NPM1) gene mutations in common solid cancers. APMIS 2007; 115: 341–346.
Hsu CY, Yung BY . Down-regulation of nucleophosmin/B23 during retinoic acid-induced differentiation of human promyelocytic leukemia HL-60 cells. Oncogene 1998; 16: 915–923.
Alcalay M, Tiacci E, Bergomas R, Bigerna B, Venturini E, Minardi SP et al. Acute myeloid leukemia bearing cytoplasmic nucleophosmin (NPMc+ AML) shows a distinct gene expression profile characterized by up-regulation of genes involved in stem-cell maintenance. Blood 2005; 106: 899–902.
Garzon R, Garofalo M, Martelli MP, Briesewitz R, Wang L, Fernandez-Cymering C et al. Distinctive microRNA signature of acute myeloid leukemia bearing cytoplasmic mutated nucleophosmin. Proc Natl Acad Sci USA 2008; 105: 3945–3950.
Chou WC, Tang JL, Lin LI, Yao M, Tsay W, Chen CY et al. Nucleophosmin mutations in de novo acute myeloid leukemia: the age-dependent incidences and the stability during disease evolution. Cancer Res 2006; 66: 3310–3316.
Palmisano M, Grafone T, Ottaviani E, Testoni N, Baccarani M, Martinelli G . NPM1 mutations are more stable than FLT3 mutations during the course of disease in patients with acute myeloid leukemia. Haematologica 2007; 92: 1268–1269.
Falini B, Martelli MP, Mecucci C, Liso A, Bolli N, Bigerna B et al. Cytoplasmic mutated nucleophosmin is stable in primary leukemic cells and in a xenotransplant model of NPMc+ acute myeloid leukemia in SCID mice. Haematologica 2008; 93: 775–779.
Bolli N, Galimberti S, Martelli MP, Tabarrini A, Roti G, Mecucci C et al. Cytoplasmic nucleophosmin in myeloid sarcoma occurring 20 years after diagnosis of acute myeloid leukaemia. Lancet Oncol 2006; 7: 350–352.
Meloni G, Mancini M, Gianfelici V, Martelli MP, Foa R, Falini B . Late relapse of acute myeloid leukemia with mutated NPM1 after eight years: evidence of NPM1 mutation stability. Haematologica 2009; 94: 298–300.
Quentmeier H, Martelli MP, Dirks WG, Bolli N, Liso A, Macleod RA et al. Cell line OCI/AML3 bears exon-12 NPM gene mutation-A and cytoplasmic expression of nucleophosmin. Leukemia 2005; 19: 1760–1767.
Papadaki C, Dufour A, Seibl M, Schneider S, Bohlander SK, Zellmeier E et al. Monitoring minimal residual disease in acute myeloid leukaemia with NPM1 mutations by quantitative PCR: clonal evolution is a limiting factor. Br J Haematol 2009; 144: 517–523.
Kronke J, Schlenk RF, Jensen KO, Eiwen K, Habdank M, Spath D et al. Monitoring of minimal residual disease in NPM1 mutated acute myeloid leukemia (AML): results of the AML Study Group (AMLSG). Blood (ASH Annual Meeting Abstracts) 2008; 112: 699.
Kern W, Haferlach T, Schnittger S, Ludwig WD, Hiddemann W, Schoch C . Karyotype instability between diagnosis and relapse in 117 patients with acute myeloid leukemia: implications for resistance against therapy. Leukemia 2002; 16: 2084–2091.
Mariano AR, Colombo E, Luzi L, Martinelli P, Volorio S, Bernard L et al. Cytoplasmic localization of NPM in myeloid leukemias is dictated by gain-of-function mutations that create a functional nuclear export signal. Oncogene 2006; 25: 4376–4380.
Albiero E, Madeo D, Bolli N, Giaretta I, Bona ED, Martelli MF et al. Identification and functional characterization of a cytoplasmic nucleophosmin leukaemic mutant generated by a novel exon-11 NPM1 mutation. Leukemia 2007; 21: 1099–1103.
Pitiot AS, Santamaria I, Garcia-Suarez O, Centeno I, Astudillo A, Rayon C et al. A new type of NPM1 gene mutation in AML leading to a C-terminal truncated protein. Leukemia 2007; 21: 1564–1566.
Falini B, Albiero E, Bolli N, De Marco MF, Madeo D, Martelli M et al. Aberrant cytoplasmic expression of C-terminal-truncated NPM leukaemic mutant is dictated by tryptophans loss and a new NES motif. Leukemia 2007; 21: 2052–2054.
la Cour T, Gupta R, Rapacki K, Skriver K, Poulsen FM, Brunak S . NESbase version 1.0: a database of nuclear export signals. Nucleic Acids Res 2003; 31: 393–396.
Henderson BR, Eleftheriou A . A comparison of the activity, sequence specificity, and CRM1-dependence of different nuclear export signals. Exp Cell Res 2000; 256: 213–224.
den Besten W, Kuo ML, Williams RT, Sherr CJ . Myeloid leukemia-associated nucleophosmin mutants perturb p53-dependent and independent activities of the Arf tumor suppressor protein. Cell Cycle 2005; 4: 1593–1598.
Colombo E, Martinelli P, Zamponi R, Shing DC, Bonetti P, Luzi L et al. Delocalization and destabilization of the Arf tumor suppressor by the leukemia-associated NPM mutant. Cancer Res 2006; 66: 3044–3050.
Bischof D, Pulford K, Mason DY, Morris SW . Role of the nucleophosmin (NPM) portion of the non-Hodgkin's lymphoma-associated NPM-anaplastic lymphoma kinase fusion protein in oncogenesis. Mol Cell Biol 1997; 17: 2312–2325.
Pasqualucci L, Liso A, Martelli MP, Bolli N, Pacini R, Tabarrini A et al. Mutated nucleophosmin detects clonal multilineage involvement in acute myeloid leukemia: impact on WHO classification. Blood 2006; 108: 4146–4155.
Gallagher SJ, Kefford RF, Rizos H . The ARF tumour suppressor. Int J Biochem Cell Biol 2006; 38: 1637–1641.
Bertwistle D, Sugimoto M, Sherr CJ . Physical and functional interactions of the Arf tumor suppressor protein with nucleophosmin/B23. Mol Cell Biol 2004; 24: 985–996.
Sherr CJ . Divorcing ARF and p53: an unsettled case. Nat Rev Cancer 2006; 6: 663–673.
Kuo ML, den Besten W, Bertwistle D, Roussel MF, Sherr CJ . N-terminal polyubiquitination and degradation of the Arf tumor suppressor. Genes Dev 2004; 18: 1862–1874.
Colombo E, Bonetti P, Lazzerini Denchi E, Martinelli P, Zamponi R, Marine JC et al. Nucleophosmin is required for DNA integrity and p19Arf protein stability. Mol Cell Biol 2005; 25: 8874–8886.
Goldberg AL . Protein degradation and protection against misfolded or damaged proteins. Nature 2003; 426: 895–899.
Gjerset RA, Bandyopadhyay K . Regulation of p14ARF through subnuclear compartmentalization. Cell Cycle 2006; 5: 686–690.
Gurumurthy M, Tan CH, Ng R, Zeiger L, Lau J, Lee J et al. Nucleophosmin interacts with HEXIM1 and regulates RNA polymerase II transcription. J Mol Biol 2008; 378: 302–317.
Grandage VL, Gale RE, Linch DC, Khwaja A . PI3-kinase/Akt is constitutively active in primary acute myeloid leukaemia cells and regulates survival and chemoresistance via NF-kappaB, Mapkinase and p53 pathways. Leukemia 2005; 19: 586–594.
Cilloni D, Messa F, Rosso V, Arruga F, Defilippi I, Carturan S et al. Increase sensitivity to chemotherapeutical agents and cytoplasmatic interaction between NPM leukemic mutant and NF-kappaB in AML carrying NPM1 mutations. Leukemia 2008; 22: 1234–1240.
Bonetti P, Davoli T, Sironi C, Amati B, Pelicci PG, Colombo E . Nucleophosmin and its AML-associated mutant regulate c-Myc turnover through Fbw7 gamma. J Cell Biol 2008; 182: 19–26.
Popov N, Wanzel M, Madiredjo M, Zhang D, Beijersbergen R, Bernards R et al. The ubiquitin-specific protease USP28 is required for MYC stability. Nat Cell Biol 2007; 9: 765–774.
Di Fiore PP . Playing both sides: nucleophosmin between tumor suppression and oncogenesis. J Cell Biol 2008; 182: 7–9.
Cheng K, Grisendi S, Clohessy JG, Majid S, Bernardi R, Sportoletti P et al. The leukemia-associated cytoplasmic nucleophosmin mutant is an oncogene with paradoxical functions: Arf inactivation and induction of cellular senescence. Oncogene 2007; 26: 7391–7400.
Wertheim G, Bagg A . Nucleophosmin (NPM1) mutations in acute myeloid leukemia: an ongoing (cytoplasmic) tale of dueling mutations and duality of molecular genetic testing methodologies. J Mol Diagn 2008; 10: 198–202.
Martelli MP, Manes N, Liso A, Pettirossi V, Verducci Galletti B, Bigerna B et al. A western blot assay for detecting mutant nucleophosmin (NPM1) proteins in acute myeloid leukaemia. Leukemia 2008; 22: 2285–2288.
Falini B, Lenze D, Hasserjian R, Coupland S, Jaehne D, Soupir C et al. Cytoplasmic mutated nucleophosmin (NPM) defines the molecular status of a significant fraction of myeloid sarcomas. Leukemia 2007; 21: 1566–1570.
Arber DA, Brunning RD, Le Beau MM, Falini B, Vardiman JW, Porwit A, et al., Bloomfield CD Acute myeloid leukaemia with recurrent genetic abnormalities. In: Swerdlow S et al. (eds). WHO Classification of Tumours of Haematopoietic and Lymphoid Tissues. IARC: Lyon, 2008, pp 110–123.
Oelschlaegel U, Koch S, Schaich M, Kroschinsky F, Parmentier S, Ehninger G et al. A rapid flow cytometric method for the detection of NPM1 mutated patients with acute myeloid leukemia (AML). Blood (ASH Annual Meeting Abstracts) 2008; 112: 1490.
Gorello P, Cazzaniga G, Alberti F, Dell'Oro MG, Gottardi E, Specchia G et al. Quantitative assessment of minimal residual disease in acute myeloid leukemia carrying nucleophosmin (NPM1) gene mutations. Leukemia 2006; 20: 1103–1108.
Chou WC, Tang JL, Wu SJ, Tsay W, Yao M, Huang SY et al. Clinical implications of minimal residual disease monitoring by quantitative polymerase chain reaction in acute myeloid leukemia patients bearing nucleophosmin (NPM1) mutations. Leukemia 2007; 21: 998–1004.
Schnittger S, Schoch C, Kern W, Mecucci C, Tschulik C, Martelli MF et al. Nucleophosmin gene mutations are predictors of favorable prognosis in acute myelogenous leukemia with a normal karyotype. Blood 2005; 106: 3733–3739.
Dohner K, Schlenk RF, Habdank M, Scholl C, Rucker FG, Corbacioglu A et al. Mutant nucleophosmin (NPM1) predicts favorable prognosis in younger adults with acute myeloid leukemia and normal cytogenetics: interaction with other gene mutations. Blood 2005; 106: 3740–3746.
Verhaak RG, Goudswaard CS, van Putten W, Bijl MA, Sanders MA, Hugens W et al. Mutations in nucleophosmin (NPM1) in acute myeloid leukemia (AML): association with other gene abnormalities and previously established gene expression signatures and their favorable prognostic significance. Blood 2005; 106: 3747–3754.
Thiede C, Koch S, Creutzig E, Steudel C, Illmer T, Schaich M et al. Prevalence and prognostic impact of NPM1 mutations in 1485 adult patients with acute myeloid leukemia (AML). Blood 2006; 107: 4011–4020.
Gale RE, Green C, Allen C, Mead AJ, Burnett AK, Hills RK et al. The impact of FLT3 internal tandem duplication mutant level, number, size and interaction with NPM1 mutations in a large cohort of young adult patients with acute myeloid leukemia. Blood 2008; 111: 2776–2784.
Schlenk RF, Dohner K, Krauter J, Frohling S, Corbacioglu A, Bullinger L et al. Mutations and treatment outcome in cytogenetically normal acute myeloid leukemia. N Engl J Med 2008; 358: 1909–1918.
Haferlach C, Mecucci C, Schnittger S, Kohlmann A, Mancini M, Cuneo A et al. AML with mutated NPM1 carrying a normal or aberrant karyotype show overlapping biological, pathological, immunophenotypic, and prognostic features. Blood 2009 [E-pub ahead of print].
Liso A, Colau D, Benmaamar R, De Groot A, Martin W, Benedetti R et al. Nucleophosmin leukaemic mutants contain C-terminus peptides that bind HLA class I molecules. Leukemia 2008; 22: 424–426.
Schnittger S, Kern W, Weiss T, Tschulik C, Dicker F, Haferlach C et al. Minimal residual disease assessed by NPM1 mutation specific RQ-PCR is the most relevant prognostic parameter in NPM1-mutated AML and highly useful to guide therapy. Blood 2008; 112: 698.
Scholl S, Theuer C, Scheble V, Kunert C, Heller A, Mugge LO et al. Clinical impact of nucleophosmin mutations and Flt3 internal tandem duplications in patients older than 60 yr with acute myeloid leukaemia. Eur J Haematol 2008; 80: 208–215.
Schlenk RF, Dohner K, Kneba M, Gotze K, Hartmann F, Del Valle F et al. Gene mutations and response to treatment with all-trans retinoic acid in elderly patients with acute myeloid leukaemia. Results from the AMLSG Trial AML HD98B. Haematologica 2009; 94: 54–60.
Kudo N, Matsumori N, Taoka H, Fujiwara D, Schreiner EP, Wolff B et al. Leptomycin B inactivates CRM1/exportin 1 by covalent modification at a cysteine residue in the central conserved region. Proc Natl Acad Sci USA 1999; 96: 9112–9117.
Dong X, Biswas A, Suel KE, Jackson LK, Martinez R, Gu H et al. Structural basis for leucine-rich nuclear export signal recognition by CRM1. Nature 2009; 458: 1136–1141.
Newlands ES, Rustin GJ, Brampton MH . Phase I trial of elactocin. Br J Cancer 1996; 74: 648–649.
Mutka SC, Yang WQ, Dong SD, Ward SL, Craig DA, Timmermans PB et al. Identification of nuclear export inhibitors with potent anticancer activity in vivo. Cancer Res 2009; 69: 510–517.
Scaloni F, Gianni S, Federici L, Falini B, Brunori M . Folding mechanism of the C-terminal domain of nucleophosmin: residual structure in the denatured state and its pathophysiological significance. FASEB J 2009 [Epub ahead of print].
Perera Y, Farina HG, Gil J, Rodriguez A, Benavent F, Castellanos L et al. Anticancer peptide CIGB-300 binds to nucleophosmin/B23, impairs its CK2-mediated phosphorylation, and leads to apoptosis through its nucleolar disassembly activity. Mol Cancer Ther 2009; 8: 1189–1196.
Benson C, White J, De Bono J, O'Donnell A, Raynaud F, Cruickshank C et al. A phase I trial of the selective oral cyclin-dependent kinase inhibitor seliciclib (CYC202; R-Roscovitine), administered twice daily for 7 days every 21 days. Br J Cancer 2007; 96: 29–37.
Zhou Y, Du W, Koretsky T, Bagby GC, Pang Q . TAT-mediated intracellular delivery of NPM-derived peptide induces apoptosis in leukemic cells and suppresses leukemogenesis in mice. Blood 2008; 112: 2474–2483.
Walensky LD, Kung AL, Escher I, Malia TJ, Barbuto S, Wright RD et al. Activation of apoptosis in vivo by a hydrocarbon-stapled BH3 helix. Science 2004; 305: 1466–1470.
Bernal F, Tyler AF, Korsmeyer SJ, Walensky LD, Verdine GL . Reactivation of the p53 tumor suppressor pathway by a stapled p53 peptide. J Am Chem Soc 2007; 129: 2456–2457.
Gale R, Hills R, Green C, Patel Y, Gilkes A, Lazenby M et al. The impact of FLT3-ITD and NPM1 mutational status on the outcome of ATRA therapy in patients with non-APL AML: results of the UK MRC12 trial. Blood (ASH Annual Meeting Abstracts) 2008; 112: 554.
Hutter ML, Dohner K, Schmidt-Wolf IGH, Del Valle F, Heydrich B, Heit W et al. Role of etoposide in combination with all-trans retinoic acid in the treatment of elderly patients with acute myeloid leukemia and NPM1 mutation. Blood (ASH Annual Meeting Abstracts) 2008; 112: 559.
Martelli MP, Pettirossi V, Manes N, Liso A, Mezzasoma F, Cecchetti F et al. Selective silencing of the NPM1 mutant protein and apoptosis induction upon ATRA in vitro treatment of AML cells carrying NPM1 mutations. Blood (ASH Annual Meeting Abstracts) 2007; 110: 868.
Acknowledgements
This work was supported by grants from Associazione Italiana Ricerca Cancro (AIRC) and Fondazione Cassa di Risparmio di Perugia, project code 2007.0099.020. We thank Roberta Pacini and Manola Carini for performing the immunohistochemical studies and Dr Geraldine Boyd for helping editing the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Falini, B., Bolli, N., Liso, A. et al. Altered nucleophosmin transport in acute myeloid leukaemia with mutated NPM1: molecular basis and clinical implications. Leukemia 23, 1731–1743 (2009). https://doi.org/10.1038/leu.2009.124
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/leu.2009.124
Keywords
This article is cited by
-
Regulation of HOX gene expression in AML
Blood Cancer Journal (2024)
-
The acidic stretch and the C-terminal nuclear export signal motif of NPM1 mutant: are they druggable in AML?
Leukemia (2023)
-
Causal linkage of presence of mutant NPM1 to efficacy of novel therapeutic agents against AML cells with mutant NPM1
Leukemia (2023)
-
Current status and future perspectives in targeted therapy of NPM1-mutated AML
Leukemia (2022)
-
The absent/low expression of CD34 in NPM1-mutated AML is not related to cytoplasmic dislocation of NPM1 mutant protein
Leukemia (2022)