Abstract
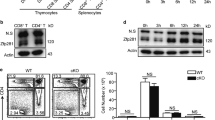
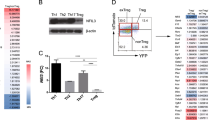
On activation, umbilical cord blood (UCB) CD4+ T cells demonstrate reduced expression of tumor necrosis factor-α (TNF-α) and interferon-γ (IFN-γ), whereas maintaining equivalent interleukin-2 (IL-2) levels, as compared with adult peripheral blood (PB) CD4+ T cells. Nuclear factor of activated T cells (NFAT1) protein, a transcription factor known to regulate the expression of IL-2, TNF-α and IFN-γ, is reduced in resting and activated UCB CD4+ T cells. In contrast, expression of Broad-complex-Tramtrack-Bric-a-Brac and Cap‘n’collar homology 1 bZip transcription factor 2 (BACH2) was shown by gene array analyses to be increased in UCB CD4+ T cells and was validated by qRT-PCR. Using chromatin immunoprecipitation, BACH2 was shown binding to the human IL-2 proximal promoter. Knockdown experiments of BACH2 by transient transfection of UCB CD4+ T cells with BACH2 siRNA resulted in significant reductions in stimulated IL-2 production. Decreased IL-2 gene transcription in UCB CD4+ T cells transfected with BACH2 siRNA was confirmed by a human IL-2 luciferase assay. In summary, BACH2 maintains IL-2 expression in UCB CD4+ T cells at levels equivalent to adult PB CD4+ T cells despite reduced NFAT1 protein expression. Thus, BACH2 expression is necessary to maintain IL-2 production when NFAT1 protein is reduced, potentially impacting UCB graft CD4+ T-cell allogeneic responses.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Chalmers I, Janossy G, Contreras M, Navarrete C . Intracellular cytokine profile of cord and adult blood lymphocytes. Blood 1998; 92: 11–18.
Risdon G, Gaddy J, Stehman FB, Broxmeyer HE . Proliferative and cytotoxic responses of human cord blood T lymphocytes following allogeneic stimulation. Cell Immunol 1994; 154: 14–24.
Kadereit S, Kozik MM, Junge GR, Miller RE, Slivka LF, Bos LS et al. Cyclosporin A effects during primary and secondary activation of human umbilical cord blood T lymphocytes. Exp Hematol 2001; 29: 903–909.
Risdon G, Gaddy J, Broxmeyer HE . Allogeneic responses of human umbilical cord blood. Blood Cells 1994; 20: 566–570; discussion 571–562.
Porcu P, Gaddy J, Broxmeyer HE . Alloantigen-induced unresponsiveness in cord blood T lymphocytes is associated with defective activation of Ras. Proc Natl Acad Sci USA 1998; 95: 4538–4543.
Kaminski BA, Kadereit S, Miller RE, Leahy P, Stein KR, Topa DA et al. Reduced expression of NFAT-associated genes in UCB versus adult CD4+ T lymphocytes during primary stimulation. Blood 2003; 102: 4608–4617.
Canto E, Rodriguez-Sanchez JL, Vidal S . Naive CD4+ cells from cord blood can generate competent Th effector cells. Transplantation 2005; 80: 850–858.
Andersson J, Stefanova I, Stephens GL, Shevach EM . CD4+CD25+ regulatory T cells are activated in vivo by recognition of self. Int Immunol 2007; 19: 557–566.
Krampera M, Tavecchia L, Benedetti F, Nadali G, Pizzolo G . Intracellular cytokine profile of cord blood T-, and NK-cells and monocytes. Haematologica 2000; 85: 675–679.
Bachmann MF, Oxenius A . Interleukin 2: from immunostimulation to immunoregulation and back again. EMBO Rep 2007; 8: 1142–1148.
Powell JD, Ragheb JA, Kitagawa-Sakakida S, Schwartz RH . Molecular regulation of interleukin-2 expression by CD28 co-stimulation and anergy. Immunol Rev 1998; 165: 287–300.
Wolf M, Schimpl A, Hunig T . Control of T-cell hyperactivation in IL-2-deficient mice by CD4(+)CD25(−) and CD4(+)CD25(+) T cells: evidence for two distinct regulatory mechanisms. Eur J Immunol 2001; 31: 1637–1645.
Bosque A, Marzo I, Naval J, Anel A . Apoptosis by IL-2 deprivation in human CD8+ T-cell blasts predominates over death receptor ligation, requires Bim expression and is associated with Mcl-1 loss. Mol Immunol 2007; 44: 1446–1453.
Long SA, Buckner JH . Combination of rapamycin and IL-2 increases de novo induction of human CD4(+)CD25(+)FOXP3(+) T cells. J Autoimmun 2008; 30: 293–302.
Jain J, Loh C, Rao A . Transcriptional regulation of the IL-2 gene. Curr Opin Immunol 1995; 7: 333–342.
Jain J, Valge-Archer VE, Rao A . Analysis of the AP-1 sites in the IL-2 promoter. J Immunol 1992; 148: 1240–1250.
Chen L, Glover JN, Hogan PG, Rao A, Harrison SC . Structure of the DNA-binding domains from NFAT, Fos and Jun bound specifically to DNA. Nature 1998; 392: 42–48.
Chen L, Oakley MG, Glover JN, Jain J, Dervan PB, Hogan PG et al. Only one of the two DNA-bound orientations of AP-1 found in solution cooperates with NFATp. Curr Biol 1995; 5: 882–889.
Diebold RJ, Rajaram N, Leonard DA, Kerppola TK . Molecular basis of cooperative DNA bending and oriented heterodimer binding in the NFAT1–Fos–Jun–ARRE2 complex. Proc Natl Acad Sci USA 1998; 95: 7915–7920.
Kadereit S, Mohammad SF, Miller RE, Woods KD, Listrom CD, McKinnon K et al. Reduced NFAT1 protein expression in human umbilical cord blood T lymphocytes. Blood 1999; 94: 3101–3107.
Sterkers G, Henin Y, Kalil J, Bagot M, Levy JP . Influence of HLA class I- and class II-specific monoclonal antibodies on DR-restricted lymphoproliferative responses. I. Unseparated populations of effector cells. J Immunol 1983; 131: 2735–2740.
Frandji P, Tkaczyk C, Oskeritzian C, David B, Desaymard C, Mecheri S . Exogenous and endogenous antigens are differentially presented by mast cells to CD4+ T lymphocytes. Eur J Immunol 1996; 26: 2517–2528.
Pfaffl MW, Horgan GW, Dempfle L . Relative expression software tool (REST) for group-wise comparison and statistical analysis of relative expression results in real-time PCR. Nucl Acids Res 2002; 30: e36.
Mitchell CA, Jefferson AB, Bejeck BE, Brugge JS, Deuel TF, Majerus PW . Thrombin-stimulated immunoprecipitation of phosphatidylinositol 3-kinase from human platelets. Proc Natl Acad Sci USA 1990; 87: 9396–9400.
Schug J . Current Protocols in Bioinformatics. Wiley: Hoboken, 2003.
Oyake T, Itoh K, Motohashi H, Hayashi N, Hoshino H, Nishizawa M et al. Bach proteins belong to a novel family of BTB-basic leucine zipper transcription factors that interact with MafK and regulate transcription through the NF-E2 site. Mol Cell Biol 1996; 16: 6083–6095.
Bhaumik SR, Green MR . Interaction of Gal4p with components of transcription machinery in vivo. Methods Enzymol 2003; 370: 445–454.
Chuvpilo S, Avots A, Berberich-Siebelt F, Glockner J, Fischer C, Kerstan A et al. Multiple NF-ATc isoforms with individual transcriptional properties are synthesized in T lymphocytes. J Immunol 1999; 162: 7294–7301.
Muckenfuss H, Hamdorf M, Avots A, Sanzenbacher R, Tschulena U, Cichutek K et al. IL-2 induction by simian immunodeficiency virus involves MAP kinase signaling but is independent of calcineurin/NF-AT activity. Mol Immunol 2006; 43: 1172–1182.
Jain J, McCaffrey PG, Miner Z, Kerppola TK, Lambert JN, Verdine GL et al. The T-cell transcription factor NFATp is a substrate for calcineurin and interacts with Fos and Jun. Nature 1993; 365: 352–355.
D'Arena G, Musto P, Cascavilla N, Di Giorgio G, Fusilli S, Zendoli F et al. Flow cytometric characterization of human umbilical cord blood lymphocytes: immunophenotypic features. Haematologica 1998; 83: 197–203.
Garcia Vela JA, Delgado I, Bornstein R, Alvarez B, Auray MC, Martin I et al. Comparative intracellular cytokine production by in vitro stimulated T lymphocytes from human umbilical cord blood (HUCB) and adult peripheral blood (APB). Anal Cell Pathol 2000; 20: 93–98.
Ahmadzadeh M, Rosenberg SA . IL-2 administration increases CD4+ CD25(hi) Foxp3+ regulatory T cells in cancer patients. Blood 2006; 107: 2409–2414.
Malek TR, Bayer AL . Tolerance, not immunity, crucially depends on IL-2. Nat Rev 2004; 4: 665–674.
Muto A, Hoshino H, Madisen L, Yanai N, Obinata M, Karasuyama H et al. Identification of Bach2 as a B-cell-specific partner for small maf proteins that negatively regulate the immunoglobulin heavy chain gene 3′ enhancer. EMBO J 1998; 17: 5734–5743.
Sasaki S, Ito E, Toki T, Maekawa T, Kanezaki R, Umenai T et al. Cloning and expression of human B cell-specific transcription factor BACH2 mapped to chromosome 6q15. Oncogene 2000; 19: 3739–3749.
Vieira SA, Deininger MW, Sorour A, Sinclair P, Foroni L, Goldman JM et al. Transcription factor BACH2 is transcriptionally regulated by the BCR/ABL oncogene. Genes Chromosomes Cancer 2001; 32: 353–363.
Muto A, Tashiro S, Tsuchiya H, Kume A, Kanno M, Ito E et al. Activation of Maf/AP-1 repressor Bach2 by oxidative stress promotes apoptosis and its interaction with promyelocytic leukemia nuclear bodies. J Biol Chem 2002; 277: 20724–20733.
Hoshino H, Igarashi K . Expression of the oxidative stress-regulated transcription factor bach2 in differentiating neuronal cells. J Biochem (Tokyo) 2002; 132: 427–431.
Muto A, Tashiro S, Nakajima O, Hoshino H, Takahashi S, Sakoda E et al. The transcriptional programme of antibody class switching involves the repressor BACH2. Nature 2004; 429: 566–571.
Sakane-Ishikawa E, Nakatsuka S, Tomita Y, Fujita S, Nakamichi I, Takakuwa T et al. Prognostic significance of BACH2 expression in diffuse large B-cell lymphoma: a study of the Osaka Lymphoma Study Group. J Clin Oncol 2005; 23: 8012–8017.
Chen W, Zhang W, Zhu J . [Homozygous deletion and methylation of p16 and p15 gene in acute leukemia]. Zhonghua xue ye xue za zhi=Zhonghua xueyexue zazhi 1999; 20: 474–476.
Kadereit S, Junge GR, Kleen T, Kozik MM, Kaminski BA, Daum-Woods K et al. Deficient IFN-gamma expression in umbilical cord blood (UCB) T cells can be rescued by IFN-gamma-mediated increase in NFATc2 expression. J Clin Immunol 2003; 23: 485–497.
Kleen TO, Kadereit S, Fanning LR, Jaroscak J, Fu P, Meyerson HJ et al. Recipient-specific tolerance after HLA-mismatched umbilical cord blood stem cell transplantation. Transplantation 2005; 80: 1316–1322.
Han S, Williams S, Mustelin T . Cytoskeletal protein tyrosine phosphatase PTPH1 reduces T cell antigen receptor signaling. Eur J Immunol 2000; 30: 1318–1325.
Acknowledgements
We thank Dr A Avots of the Pathologisches Institut, Würzburg, Germany, for his generous donation of the human IL-2 promoter plasmid construct. We also thank Abraham J and Phyllis Katz Foundation, Fannie E Rippel Foundation and Dr Donald and Ruth Weber Goodman Philanthropic Fund for their generous financial contribution to our laboratory.
Funding: This work was supported by RO1-AI47289-01 (MJL), NIH/NCI 5T32 CA059366-13 Research Oncology Training Grant (MLL). This research was supported by the Gene Expression and Genotyping Facility of the Case Comprehensive Cancer Center 5P30CA043703 (ClinicalTrials.gov identifier: NCT00003335), Abraham J and Phyllis Katz Foundation, Fannie E Rippel Foundation and Dr Donald and Ruth Weber Goodman Philanthropic Fund (MJL).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lesniewski, M., Haviernik, P., Weitzel, R. et al. Regulation of IL-2 expression by transcription factor BACH2 in umbilical cord blood CD4+ T cells. Leukemia 22, 2201–2207 (2008). https://doi.org/10.1038/leu.2008.234
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/leu.2008.234
Keywords
This article is cited by
-
Foxp3 expression in induced T regulatory cells derived from human umbilical cord blood vs. adult peripheral blood
Bone Marrow Transplantation (2018)