Abstract
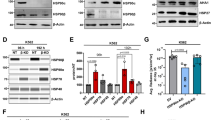
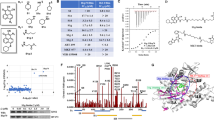
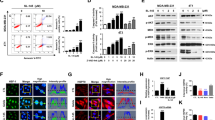
We as well as others have recently shown that Hsp90 is overexpressed in multiple myeloma (MM) and critically contributes to tumour cell survival. Pharmacologic blockade of Hsp90 has consistently been found to induce MM cell death. However, most data have been obtained with MM cell lines whereas knowledge about the molecular effects of pharmacologic Hsp90 blockade in primary tumour cells is limited. Furthermore, these investigations have so far focused on geldanamycin derivatives. We analysed the biochemical effects of a novel diarylisoxazole-based Hsp90 inhibitor (NVP-AUY922) on signalling pathways and cell death in a large set of primary MM tumour samples and in MM cell lines. Treated cells displayed the molecular signature and pharmacodynamic properties for abrogation of Hsp90 function, such as downregulation of multiple survival pathways and strong upregulation of Hsp70. NVP-AUY922 treatment efficiently induced MM cell apoptosis and revealed both sensitive and resistant subgroups. Sensitivity was not correlated with TP53 mutation or Hsp70 induction levels and stromal cells from the bone marrow microenvironment were unable to abrogate NVP-AUY922-induced apoptosis of MM cells. Thus, NVP-AUY922 may be a promising drug for treatment of MM and clinical studies are warranted.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Kuehl WM, Bergsagel PL . Early genetic events provide the basis for a clinical classification of multiple myeloma. Hematology 2005, 346–352. Available at http:\\asheducationbook.hematologylibrary.org/content/vol2005/issue1/.
Kyle RA, Rajkumar SV . Multiple myeloma. Blood 2008; 111: 2962–2972.
http://www.picard.ch/downloads/downloads.htm; accessed on 2 April 2008.
Whitesell L, Lindquist SL . HSP90 and the chaperoning of cancer. Nat Rev Cancer 2005; 5: 761–772.
Zhang H, Burrows F . Targeting multiple signal transduction pathways through inhibition of Hsp90. J Mol Med 2004; 82: 488–499.
Cullinan SB, Whitesell L . Heat shock protein 90: a unique chemotherapeutic target. Semin Oncol 2006; 33: 457–465.
Beere HM . The stress of dying: the role of heat shock proteins in the regulation of apoptosis. J Cell Sci 2004; 117: 2641–2651.
Sanderson S, Valenti M, Gowan S, Patterson L, Ahmad Z, Workman P et al. Benzoquinone ansamycin heat shock protein 90 inhibitors modulate multiple functions required for tumor angiogenesis. Mol Cancer Ther 2006; 5: 522–533.
Mitsiades CS, Mitsiades NS, McMullan CJ, Poulaki V, Kung AL, Davies FE et al. Antimyeloma activity of heat shock protein-90 inhibition. Blood 2006; 107: 1092–1100.
Chatterjee M, Jain S, Stühmer T, Andrulis M, Ungethüm U, Kuban R-J et al. STAT3 and MAPK signaling maintain overexpression of heat shock proteins 90α and β in multiple myeloma cells, which critically contribute to tumor cell survival. Blood 2007; 109: 720–728.
Sydor JR, Normant E, Pien CS, Porter JR, Ge J, Grenier L et al. Development of 17-allylamino-17-demethoxygeldanamycin hydroquinone hydrochloride (IPI-504), an anti-cancer agent directed against Hsp90. Proc Natl Acad Sci USA 2006; 103: 17408–17413.
Duus J, Bahar HI, Venkataraman G, Ozpuyan F, Izban KF, Al-Masri H et al. Analysis of expression of heat shock protein-90 (HSP90) and the effects of HSP90 inhibitor (17-AAG) in multiple myeloma. Leuk Lymphoma 2006; 47: 1369–1378.
Whitesell L, Mimnaugh EG, Costa BD, Myers CE, Neckers LM . Inhibition of heat shock protein HSP90-pp60v-src heteroprotein complex formation by benzoquinone ansamycins: essential role for stress proteins in oncogenic transformation. Proc Natl Acad Sci USA 1994; 91: 8324–8328.
Supko JG, Hickman RL, Grever MR, Malspeis L . Preclinical pharmacologic evaluation of geldanamycin as an antitumor agent. Cancer Chemother Pharmacol 1995; 36: 305–315.
Egorin MJ, Zuhowski EG, Rosen DM, Sentz DL, Covey JM, Eiseman JL et al. Plasma pharmacokinetics and tissue distribution of 17-(allylamino)-17-demethoxygeldanamycin (NSC 330507) in CD2F1 mice1. Cancer Chemother Pharmacol 2001; 47: 291–302.
Glaze ER, Lambert AL, Smith AC, Page JG, Johnson WD, McCormick DL et al. Preclinical toxicity of a geldanamycin analog, 17-(dimethylaminoethylamino)-17-demethoxygeldanamycin (17-DMAG), in rats and dogs: potential clinical relevance. Cancer Chemother Pharmacol 2005; 56: 637–647.
Neckers L, Ivy SP . Heat shock protein 90. Curr Opin Oncol 2003; 15: 419–424.
Brough PA, Aherne W, Barril X, Borgognoni J, Boxall K, Cansfield JE et al. 4,5-diarylisoxazole Hsp90 chaperone inhibitors: potential therapeutic agents for the treatment of cancer. J Med Chem 2008; 51: 196–218.
Janz M, Stühmer T, Vassilev LT, Bargou RC . Pharmacologic activation of p53-dependent and p53-independent apoptotic pathways in Hodgkin/Reed-Sternberg cells. Leukemia 2007; 21: 772–779.
Siegmund D, Wicovsky A, Schmitz I, Schulze-Osthoff K, Kreuz S, Leverkus M et al. Death receptor-induced signaling pathways are differentially regulated by gamma interferon upstream of caspase 8 processing. Mol Cell Biol 2005; 25: 6363–6379.
Chiosis G, Neckers L . Tumor selectivity of Hsp90 inhibitors: the explanation remains elusive. ACS Chem Biol 2006; 1: 279–284.
Sato N, Yamamoto T, Sekine Y, Yumioka T, Junicho A, Fuse H et al. Involvement of heat-shock protein 90 in the interleukin-6-mediated signaling pathway through STAT3. Biochem Biophys Res Commun 2003; 300: 847–852.
Shang L, Tomasi TB . The heat shock protein 90-CDC37 chaperone complex is required for signaling by types I and II interferons. J Biol Chem 2006; 281: 1876–1884.
Mitsiades CS, Mitsiades NS, Munshi NC, Richardson PG, Anderson KC . The role of the bone microenvironment in the pathophysiology and therapeutic management of multiple myeloma: interplay of growth factors, their receptors and stromal interactions. Eur J Cancer 2006; 42: 1564–1573.
Chauhan D, Hideshima T, Mitsiades C, Richardson P, Anderson KC . Proteasome inhibitor therapy in multiple myeloma. Mol Cancer Ther 2005; 4: 686–692.
Sharp S, Workman P . Inhibitors of the HSP90 molecular chaperone: Current status. Adv Cancer Res 2006; 95: 323–348.
Sharp SY, Boxall K, Rowlands M, Prodromou C, Roe SM, Maloney A et al. In vitro biological characterization of a novel, synthetic diaryl pyrazole resorcinol class of heat shock protein 90 inhibitors. Cancer Res 2007; 67: 2206–2216.
Acknowledgements
This work was supported in parts by grants from the Deutsche Forschungsgemeinschaft (SFB-TR17/C5), the Deutsche Krebshilfe (grant no. 107715) and the Deutsche José Carreras Leukämie-Stiftung e. V. (grant no. R06/17).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Stühmer, T., Zöllinger, A., Siegmund, D. et al. Signalling profile and antitumour activity of the novel Hsp90 inhibitor NVP-AUY922 in multiple myeloma. Leukemia 22, 1604–1612 (2008). https://doi.org/10.1038/leu.2008.111
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/leu.2008.111
Keywords
This article is cited by
-
Heat shock factor 1 is a promising therapeutic target against adult T-cell leukemia
Medical Oncology (2023)
-
Inhibitors of the protein disulfide isomerase family for the treatment of multiple myeloma
Leukemia (2019)
-
Phase I study of KW-2478, a novel Hsp90 inhibitor, in patients with B-cell malignancies
British Journal of Cancer (2016)
-
CD20-targeting in B-cell malignancies: novel prospects for antibodies and combination therapies
Investigational New Drugs (2016)
-
Synthesis, characterization, and pharmacological evaluation of novel azolo- and azinothiazinones containing 2,4-dihydroxyphenyl substituent as anticancer agents
Monatshefte für Chemie - Chemical Monthly (2015)