Abstract
Retinol-binding protein RBP4 is the specific carrier for retinol in the blood. We previously produced a Rbp4-deficient (Rbp4−/−) mouse that showed electroretinogram (ERG) abnormalities, accompanied by histological and electron-microscopic changes such as fewer synapses in the inner plexiform layer in the central retina. To address whether human RBP4 gene expression can rescue the phenotypes observed in Rbp4−/− mice, we produced a humanized (Rbp4hRBP4orf/ hRBP4orf) mouse with a human RBP4 open reading frame in the mouse Rbp4 locus using a Cre-mutant lox recombination system. In Rbp4hRBP4orf/hRBP4orf mice, the tissue-specific expression pattern of hRBP4orf was roughly the same as that of mouse Rbp4. ERG and morphological abnormalities observed in Rbp4−/− mice were rescued in Rbp4hRBP4orf/hRBP4orf mice as early as 7 weeks of age. The temporal expression pattern of hRBP4orf in the liver of Rbp4hRBP4orf/hRBP4orf mice was similar to that of mouse Rbp4 in Rbp4+/+mice. In contrast, hRBP4orf expression levels in eyes were significantly lower at 6 and 12 weeks of age compared with mouse Rbp4 but were restored to the control levels at 24 weeks. The serum hRBP4 levels in Rbp4hRBP4orf/hRBP4orf mice were approximately 30% of those in Rbp4+/+ at all ages examined. In accordance with this finding, the plasma retinol levels remained low in Rbp4hRBP4orf/hRBP4orf mice. Retinol accumulation in the liver occurred in control and Rbp4hRBP4orf/hRBP4orf mice but was higher in Rbp4hRBP4orf/hRBP4orf mice at 30 weeks of age. Mouse transthyretin expression was not altered in Rbp4−/− or Rbp4hRBP4orf/hRBP4orf mice. Taken together, 30% of the serum RBP4 level was sufficient to correct the abnormal phenotypes observed in Rbp4−/− mice.
Similar content being viewed by others
Main
The retinol-binding protein 4 (hRBP4 for human and mRbp4 for mouse) gene consists of six exons and five introns.1, 2 RBP4 is a single polypeptide chain with a molecular weight of approximately 21 kDa and has one binding site for retinol. RBP4 is synthesized primarily in the liver1, 3 and in other sites, including the kidney,3 the peritubular and Sertoli cells of the testis,4, 5 the retinal pigment epithelium,6, 7 and the choroid plexus of the brain8 (for a review, see Soprano and Blaner8). Vitamin A, absorbed from the intestine in the form of retinyl esters, is taken up by the liver; thus, the liver serves as the primary storage depot for vitamin A.9 In hepatocytes RBP4 associates with retinol, moves to the Golgi apparatus and is secreted into blood.8 In other words, retinol is mobilized from liver stores by binding to RBP4. Thus, retinol is deposited in the absence of RBP4. Vitamin A, in the form of retinal, is necessary for both low-light (scotopic vision) and color vision in the retina of the eye by binding with the protein opsin to form rhodopsin.10
In human patients with no detectable plasma RBP4 due to RBP4 gene mutations, various phenotypes, such as an iris coloboma, atrophy or focal loss of the retinal pigment epithelium (RPE) and the choroid, and reduced scotopic and photopic electro-retinograms (ERGs), have been reported.11, 12 Quadro et al13 produced Rbp4−/− mice in a mixed genetic background (129xC57BL/6J) and found that these mice had impaired visual function, as evidenced by abnormal ERG. The mice had decreased sensitivity to light at only the b-wave amplitude, with progressive improvement such that this sensitivity approached that of wild-type mice by 24 weeks of age. Quadro et al13, 14 also generated a transgenic mouse strain (hRBP4: mRbp4−/−) that expressed hRBP4 under the control of the muscle creatine kinase promoter in the Rbp4−/− background. Although no hRBP4 was detected in the retinal pigment epithelium of hRBP4:mRbp4−/− mice, a normal ERG profile and normal retinol levels were observed in the eyes. To examine whether similar phenotypes were observed in the C57BL/6 background, we previously produced Rbp4-deficient (Rbp4−/−) mice.15 These Rbp4−/− mice had decreased a- and b-wave amplitudes on scotopic ERGs. In accordance with these abnormalities, we found structural changes in these mice, such as the loss of the choroid and photoreceptor layers in the peripheral retina. In the central retina, the distance between the inner limiting membrane and the outer plexiform layer was much shorter with fewer ganglion cells and fewer synapses in the inner plexiform layer. Thus, Rbp4−/− mice had similar phenotypes as in human patients.
The RBP4–retinol complex binds to the transthyretin (TTR) tetramer in hepatocytes, and this complex is then secreted into the serum.16, 17, 18, 19 This binding is important for both TTR and RBP4 because it stabilizes the TTR tetramer and prevents the renal filtration of RBP4.9 As the dissociation of the TTR tetramer is a critical step in amyloid deposition, the binding affinity between TTR and RBP4 is an important factor towards the dissociation of the TTR tetramer. Therefore, it is essential to have both human TTR (hTTR) and human RBP4 (hRBP4) in the same mouse model to mimic the human environment.
To establish double-humanized mice at both the Ttr and Rbp4 loci as the final goal, we previously produced a Ttr-deficient mouse and a humanized mouse (TtrhTTRorf/hTTRorf) carrying the hTTR open reading frame (hTTRorf) in the mouse Ttr (mTtr) locus using a Cre-mutant lox recombination system.20 Using the same method, Rbp4-deficient mice have been produced.15 Here, a human RBP4 open reading frame (hRBP4orf) was inserted into the mouse Rbp4 (mRbp4) locus using these embryonic stem (ES) clones. Subsequently, humanized mice at the Rbp4 locus (Rbp4hRBP4orf/hRBP4orf) were established. We examined whether phenotypes observed in Rbp4−/− mice could be rescued by hRBP4 expression in Rbp4hRBP4orf/hRBP4orf mice. Here, we show that the retinal function and morphological defects observed in Rbp4−/− mice are rescued in Rbp4hRBP4orf/hRBP4orf, although the serum hRBP4 is approximately 30% of that of the endogenous mRBP4. This information will be valuable for estimating the expression level required to maintain function and to restore phenotypes in human genetic diseases.
Materials and methods
Construction of Targeting/Replacement Vectors and the Isolation of Targeted/Replaced ES Cells Carrying hRBP4orf
The construction of targeting vector and the isolation of targeted ES cells were achieved using a similar method as described previously.20 Briefly, a 2.8 kb 5′ Rbp4 homologous region upstream of the ATG codon and a 6.7 kb 3′ Rbp4 homologous region downstream of the ATG codon were ligated with a p71neoP cassette that contained lox71-PGK-neoR-loxP-polyA-lox2272 to produce the Rbp4-71neoR-Rbp4 construct that comprised the 5′ Rbp4 homologous region-lox71-PGK-neoR-loxP-polyA-lox2272-3′ Rbp4 homologous region. Finally, a diphtheria toxin A (DT-A) fragment with an MC1 promoter was ligated to the 3′ end of 3′ Rbp4 homologous region to produce the targeting vector (Figure 1). The targeting vectors were introduced into TT2 ES cells21 derived from an F1 embryo that resulted from a mating between C57BL/6 and CBA mice (Charles River, Yokohama, Japan).
Creation of targeted and replaced alleles. Homologous recombination between the wild-type allele and the targeting vector yielded a targeted null allele carrying the PGK-neo gene flanked by lox71 and loxP. In the second step, targeted clones were electroporated with the replacement vector carrying the hRBP4orf flanked by loxKMR3 and loxP. Site-directed recombination between lox71/loxKMR3 and loxP/loxP resulted in the creation of the replaced allele.
For the construction of the replacement vector, we first made a cassette that contained ahRBP4 cDNA, and a puromycin resistance gene with a phosphoglycerokinase promoter (PGK-puro) flanked by two Flp recognition target (FRT) sequences. Then, the cassette flanked by loxKMR3 and loxP sites was inserted into pSP73 (Promega, Tokyo, Japan). The ES cell lines with the Rbp4 targeted null allele were coelectroporated (Bio-Rad Gene Pulser at 400 V, 125 μF) with 20 μg of the replacement vector plasmid and 20 μg of the pCAGGS-Cre22 to produce ES clones with the replaced allele.
Southern Blot Analyses for the Isolation of Targeted and Replaced ES Clones
The replaced ES clones were screened and confirmed by Southern blot analyses using genomic DNA obtained from ES cells according to the methods described previously.20 For the detection of homologous recombination in the 5′ region, DNA from ES cells was digested with XhoI/SalI or MunI and analyzed for the presence of the targeted allele using a neo and 5′ probes (Supplementary Figure S1). For the detection of homologous recombination in the 3′ region, DNA from ES cells was digested with AgeI/HpaI or SacII/HpaI and analyzed for the presence of the targeted allele using a neo and 3′ probe (Supplementary Figure S1). For the detection of the replaced allele, genomic DNA from ES cells was digested with BamHI or BglII and was used for Southern blot analysis with a puromycin-specific probe (Supplementary Figure S1).
Generation of RBP4orf Knock-in Mice
Chimeric mice were produced by the aggregation of ES cells with eight-cell embryos from ICR mice according to the methods described previously.20 Chimeric male mice were backcrossed to C57BL/6 females (Clea Japan, Tokyo, Japan) for seven generations including marker-assisted breeding using 123 polymorphic markers (111 markers on autosomes, eight markers on the X chromosome and four markers on the Y chromosome; Japan Mouse Clinic: http://ja.brc.riken.jp/lab/jmc/mouse_clinic/en/assistive/index.html) in the second and sixth generations. These mice had C57BL/6-homozygous alleles at all 123 polymorphic loci. The mice were fed a CE-2 diet (Clea Japan) which contained 1515 IU retinol/100 g (0.454 mg/100 g). To avoid possible sex-related differences, only males were used in all the analyses. The experimental protocols that involved animals were approved by the Kumamoto University Ethics Committee for Animal Experiments (F25–329, F27–122) and all the experiments were performed in accordance with the institute’s guidelines.
Northern Blot, Semiquantitative RT-PCR and Quantitative RT-PCR Analyses
Total RNA was isolated from the brain, whole eyes, heart, lung, small intestine, testis, skeletal muscle, liver, and kidney. Northern blot and semiquantitative reverse transcription-polymerase chain reaction (RT-PCR) analyses were performed according to the methods described by Zhao et al20 and Li et al,23 respectively. The nucleotide sequence identity between the mouse and human Rbp4-coding regions is ~87%. The primer set suitable for human and mouse RBP4-coding regions was as follows: sense (mhRBP4-F), 5′-GAGTCAAGGAGAACTTCGAC-3′; and antisense (mhRBP4-e4R), 5′-CAGTACTTCATCTTGAACTTGG-3′; the PCR product was expected to be 244 bp long. The primer set used for the detection of the Hprt mRNA was as follows: sense, 5′-CACAGGACTAGAACACCTGC-3′; antisense, 5′-GCTGGTGAAAAGGACCTCT-3′; the PCR product was expected to be 238 bp long. PCR products were separated on 1.5% agarose gels and stained with ethidium bromide. Quantitative RT-PCR (qRT-PCR) was performed with an Applied Biosystems 7500 Real-Time PCR system (Applied Biosystems, Foster City, CA, USA). The reactions were performed under the following conditions: 1 min at 95 °C; followed by 40 cycles of 15 s at 95 °C and 1 min at 60 °C. Quantified values were normalized against the input determined by the expression of the housekeeping gene Hprt.
Functional Analysis of the Retina
ERG was performed as previously described.24 The left eyes of Rbp4+/+, Rbp4−/− and Rbp4hRBP4orf/hRBP4orf mice at 7, 14, 24, and 40 weeks of age were used. The mice were dark-adapted overnight and anesthetized with a mixture of ketamine (80 mg/kg; Daiichi Sankyo Propharma, Tokyo, Japan) and xylazine (16 mg/kg; Bayer Medical, Tokyo, Japan) under dim red light. The pupils were dilated with Mydrin-P ophthalmic solution (1:5 dilution), and the corneas were anesthetized with 0.4% oxybuprocaine hydrochloride (Santen Pharmaceutical, Tokyo, Japan). Stimulus presentation was programmed and data acquisition was performed using data recording and analysis software (Scope v.3.7; ADInstruments Pty, New South Wales, Australia). Scotopic ERG responses to single flash presentations (10 ms duration) at three increasing intensities (2.0 log cd s/m2) were recorded. Triplicate responses were computer-averaged for each flash condition. The time intervals between flashes were 10, 30, and 60 s for intensities of 2.0 log cd s/m2, respectively. The amplitude and implicit time of the a-wave were measured from the prestimulus baseline to the a-wave trough. The amplitude and implicit time of the b-wave were measured from the a-wave trough to the peak of the b-wave or, if the a-wave was not measurable, from the prestimulus baseline.
Retinal Histology
Mice at 7 and 40 weeks of age were anesthetized with an intraperitoneal injection of pentobarbital sodium (90 mg/kg; Kyoritsu Seiyaku, Tokyo, Japan). The eyes were fixed with 2% paraformaldehyde (PFA) and 2% glutaraldehyde (GA) in 0.1 M cacodylate buffer pH 7.4 at 4 °C overnight, washed three times with 0.1 M cacodylate buffer for 30 min each, and postfixed with 2% osmium tetroxide (OsO4) in 0.1 M cacodylate buffer at 4 °C for 2 h. The samples were then dehydrated in a graded ethanol series: 50 and 70% for 30 min each at 4 °C, 90% for 30 min at room temperature, three changes of 100% for 30 min each at room temperature and 100% ethanol overnight at room temperature. The samples were infiltrated with propylene oxide (PO) twice for 30 min each and were placed into a 70:30 mixture of PO and Quetol-812 resin (Nisshin EM, Tokyo, Japan) for 1 h; the tubes were kept open overnight to evaporate the PO. The samples were transferred to fresh 100% resin and polymerized at 60 °C for 48 h. The polymerized resins were semi-thin-sectioned at 1.5 mm with glass knives using an ultramicrotome (Ultracut UCT; Leica, Vienna, Austria) and were stained with 0.5% Toluidine blue.
Measurement of Retinal Properties
The distance from the retinal surface to the end of the photoreceptor outer segments was measured as the retinal thickness. The thicknesses of the INL, the ONL, and the photoreceptor OS in the central retina were measured by light microscopy at the point where the ratio of the distance from the optic disc to the point to the distance from the optic disc to peripheral end of the retina was 0.2.24 The values recorded for each parameter for the nasal and temporal sides were averaged to obtain a single value for each eye. For consistency, only retinal sections with optic nerve stumps were used.
Transmission Electron Microscopy
Mice at 7 and 40 weeks of age were used for this study. The eyes were fixed as described above at 4 °C overnight, washed three times with 0.1 M cacodylate buffer for 30 min each, and postfixed with 2% osmium tetroxide in 0.1 M cacodylate buffer at 4 °C for 2 h. The samples were then dehydrated in a graded ethanol series: 50 and 70% for 30 min each at 4 °C, 90% for 30 min at room temperature, three changes of 100% for 30 min each at room temperature and 100% ethanol overnight at room temperature. The samples were infiltrated with propylene oxide (PO) twice for 30 min each and then placed into a 70:30 mixture of PO and Quetol-812 resin (Nisshin EM) for 1 h; the tube was kept open overnight to evaporate the PO. The samples were transferred to fresh 100% resin and polymerized at 60 °C for 48 h. Ultra-thin sections (70 nm) were cut with a diamond knife using an ultramicrotome (Ultracut UCT) and mounted on copper grids. They were stained with 2% uranyl acetate at room temperature for 15 min, washed with distilled water, and stained with lead staining solution (Sigma-Aldrich, Tokyo, Japan) at room temperature for 3 min. The grids were examined under a transmission electron microscope (JEM-1400Plus; JEOL, Tokyo, Japan) at an acceleration voltage of 80 kV. Digital images (2048 × 2048 pixels) were taken with a CCD camera (Veleta; Olympus Soft lmaging Solutions GmbH, Münster, Germany).
Determination of Plasma and Liver Retinol Concentrations
Plasma and liver retinol concentrations were measured when the mice were 8 and 30 weeks of age. The plasma retinol concentration was determined by SRL (Tokyo, Japan) according to the method described previously.25 The liver retinol was determined by Japan Food Research Company (Fukuoka, Japan) according to the method described previously.26 Briefly, the samples were saponified in a basic ethanol–water solution, neutralized, and diluted, which converted retinol esters to retinol. Retinol levels were quantified using high-performance liquid chromatography. As the protocol involves the saponification of retinyl esters, retinyl esters are converted to retinol. Thus, the amount of retinol is expressed as the total retinol.
Protein Extraction from Liver
The liver was homogenized in a RIPA lysis buffer system (sc24948, Santa Cruz, Biotechnology, Dallas, TX, USA). The homogenate was transferred to centrifuge tubes and the samples centrifuged at 10 000 g for 20 min at 4 °C. The protein concentrations were measured by using the BCA Protein Assay Kit (T9300A, Takara Bio, Shiga, Japan).
Serum and Liver RBP4 Levels
Commercial ELISA kits were used according to the manufacturers’ instructions to determine serum and liver concentrations of mRBP4 (Mouse Retinol-Binding protein 4 ELISA Kit SimpleStep, ab202404, Abcam, Tokyo, Japan) and hRBP4 (AG-45A-0035YEK-KI01, AdipoGen, San Diego, CA, USA).
Western Blot Analysis
Mouse serum (5 μl diluted 1:10 in 0.9% NaCl per lane) or liver extracts (60 μg of protein per lane) were applied to a 10–20% gradient polyacrylamide gel and transferred to an Immobilon polyvinylidene difluoride membrane (Millipore, Billerica, MA, USA). The following primary antibodies were used: rabbit anti-human RBP4 antibody (diluted 1:1000; ab73022, Abcam for Figure 2c and 11774-1-AP, Proteintech, Rosemont, IL, USA for Figure 10), rabbit anti-mouse TTR antibody (diluted 1:1000; Thermo Fisher Scientific K.K., Yokohama, Japan), anti-β actin antibody (diluted 1:10 000; Gene Tex, Tokyo Japan), and rabbit anti-bovine serum albumin antibody (diluted 1:1500; Merck Millipore, Tokyo, Japan). An anti-rabbit immunoglobulin G conjugated with horseradish peroxidase (Amersham Biosciences, Piscataway, NJ, USA) was used for detection. Recombinant mTTR protein (LSBio, Seattle, WA, USA) was used for the measurement of serum mTTR levels. Band intensities were quantified using ImageJ ver. 1.38 software (http://rsb.info.nih.gov/ij/docs/index.html).
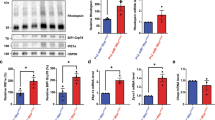
Tissue-specific and quantitative hRBP4 expression. (a) Tissue specificity of mRbp4 and hRBP4 mRNA expression. mRbp4 and hRBP4 were both expressed in various tissues, mainly in the liver, testis, and lung. (b) Northern blot analysis. The hRBP level in Rbp4hRBP4/hRBP4 mice was approximately double that in Rbp4+/hRBP4 mice. (c) Western blot analysis. The serum hRBP4 level in Rbp4hRBP4/hRBP4 mice was approximately double that in Rbp4+/hRBP4 mice. Before loading, sera from Rbp4+/+, Rbp4+/hRBP4, and Rbp4hRBP4/hRBP4 mice were diluted 10 times and human serum was diluted 50 times. B, brain; E, eye; H, heart; Lu, lung; I, intestine; T, testis; M, skeletal muscle; Li, liver; K, kidney; C, control; h, human; m, mouse.
Gel Filtration Chromatography for the Separation of RBP4–TTR Complexes
Plasma (0.1 ml) was loaded onto a Superdex 200 Increase 10/300 GL (GE Healthcare, Tokyo, Japan) column connected to an LC-8020 HPLC system (Toso, Tokyo, Japan). Protein complexes were separated by passing phosphate-buffered saline through at 0.5 ml/min for 50 min at room temperature. Eluted proteins were detected by their absorbance at 280 nm and collected in 0.5 ml fractions for western blot analysis.
Genotyping of the Crb1 Locus
DNA samples isolated from tail biopsies were amplified separately for wild-type allele and mutant rd8 alleles using the methods described previously.27, 28 The sizes of the amplified DNA of the wild-type and rd8 alleles are 220 and 244 bp, respectively.
Number of Mice Used for Each Experiment
The number of mice used for each experiment is shown in Supplementary Table 2.
Statistical Analysis
At least three independent experiments were performed. Unpaired Student’s t-tests were used for the statistical analysis. The results are expressed as the means±s.d. P<0.05 was considered to indicate a significant difference.
Results
Establishment of ES Clones and Mouse Strains with the Replaced hRBP4orf Allele
The production of a targeted null allele for the Rbp4 locus and Rbp4−/− mice by homologous recombination in ES cells was reported previously.15 Replacement vectors containing hRBP4orf and the Cre expression vector were electroporated into the targeted ES clones (Figure 1). Five of nine puromycin-resistant clones yielded 3.6 or 3.0 kb bands when digested with BamHI or BglII, respectively, in Southern blot analysis using the puromycin probe (Supplementary Figure S1). These clones were found to encode the replaced allele. Three ES clones (Nos. 2, 12, and 16) showed germline transmission (Supplementary Table S1). Homozygous humanized mice (Rbp4hRBP4orf/hRBPorf) were used in subsequent experiments. Both Rbp4−/− and Rbp4hRBP4orf/hRBPorf adults were viable and fertile under a diet of 1515 IU retinol/100 g food.
Tissue-Specific and Quantitative hRBP4 Expression
To examine the tissue-specific expression of mouse Rbp4 (mRbp4) and hRBP4, RNA was extracted from various tissues and analyzed by semiquantitative RT-PCR. hRBP4 was mainly expressed in the liver, testis, and lung, in which the mRbp4 gene was also expressed (Figure 2a). However, hRBP4 expression was lower in the brain and eyes and higher in the intestine and testis compared with the mRbp4 gene. Despite these differences, the tissue specificity of hRBP4 expression was roughly similar to that of mRbp4.
The expression levels of mRbp4- and hRBP4-encoding mRNA in Rbp4+/+, Rbp4+/hRBP4orf, and Rbp4hRBP4orf/hRBP4orf mice were analyzed by northern blot analyses. As expected, mouse and human mRNAs were detected in the livers of Rbp4+/hRBP4orf mice, but only human mRNA was detected in the livers of Rbp4hRBP4orf/hRBP4orf mice (Figure 2b). The human mRNA level in Rbp4hRBP4orf/hRBP4orf mice was approximately twice that in Rbp4+/hRBP4orf mice.
As reported previously,20 serum levels of hTTR in TtrhTTRorf/hTTRorf were lower than those in Ttr+/hTTRorf because heterotetramers between hTTR and mTTR are more stable than the hTTR homotetramer.29 To determine whether the serum hRBP level is affected in the presence of mouse RBP4, the serum levels of the hRBP4 protein were compared in Rbp4+/hRBP4orf and Rbp4hRBP4orf/hRBP4orf mice by western blot assay. In Rbp4hRBP4orf/hRBP4orf mice, the hRBP4 serum level was roughly twice that in Rbp4+/hRBP4orf mice (Figure 2c). These results show that hRBP4 was expressed in a dose-dependent manner in these mice.
Rescue of Retinal Function in Rbp4hRBP4orf/hRBP4orf Mice
A long-term ERG study was performed to analyze whether RBP4orf expression can rescue retinal function. Representative ERG waveforms are shown in Figure 3a.
Rescue of retinal function in Rbp4hRBP4/hRBP4 mice. (a) ERG waveform. ERG responses in Rbp4−/− mice recovered moderately at 40 weeks of age but were still lower than those in Rbp4+/+ and Rbp4hRBP4/hRBP mice. ERG responses in Rbp4hRBP4/hRBP4 mice were almost normal at all ages. (b) ERG responses among mouse strains. ERG responses in Rbp4hRBP4/hRBP4 mice were recovered to almost normal in all criteria. Data are presented as means±s.d.
As reported previously,15 the a- and b-wave amplitudes were significantly lower in Rbp4−/− mice throughout the 40-week observation period (Figure 3b). In contrast, the a- and b-wave amplitudes in Rbp4hRBP4orf/hRBP4orf mice were almost normal (Figure 3b). Although there was a tendency for low amplitudes in Rbp4hRBP4orf/hRBP4orf mice, differences in these amplitudes between Rbp4+/+ and Rbp4hRBP4orf/hRBP4orf mice were not statistically significant. The a- and b-wave implicit times were longer in Rbp4−/− mice at 7 weeks of age, but recovered to normal at 14 weeks of age (Figure 3b). This abnormality was not observed in Rbp4hRBP4orf/hRBP4orf mice (Figure 3b).
Restoration of Retinal Histology in Rbp4hRBP4orf/hRBP4orf Mice
To analyze the morphological bases for normalized ERG, we performed histological analyses at 7 and 40 weeks of age. Retinal histology in each genotype at 7 weeks of age (Figure 4a–c) was essentially the same as that at 40 weeks of age (Figure 4d–f). As reported previously,15 various morphological defects were observed in the retinas from Rbp4−/− mice, such as a thinner retina (Figure 4b). However, the retinas from Rbp4hRBP4orf/hRBP4orf mice (Figure 4c) showed almost no differences compared with the retinas from Rbp4+/+ mice (Figure 4a).
Restoration of retinal histology in Rbp4hRBP4/hRBP4 mice. Representative toluidine blue-stained retinal sections of 7 and 40-week-old mice are shown. (a) Central retina from an Rbp4+/+ mouse. (b) Central retina from an Rbp4−/− mouse with a thinner retina. (c) Central retina from an Rbp4hRBP4/hRBP4 mouse. (d–f) Central retinas from mice at 7 weeks of age. (g–i) Central retinas from mice at 40 weeks of age. Histological abnormalities found in Rbp4−/− mice were almost restored in Rbp4hRBP4/hRBP4 mice. GCL, ganglion cell layer; IPL, inner plexiform layer; INL, inner nuclear layer; OPL, outer plexiform layer; ONL, outer nuclear layer; IS, inner segments; OS, outer segments; RPE, retinal pigment epithelium; Ch, choroid; Sc, sclera.
Measurement of Retinal Properties
For the precise evaluation of the Rbp4−/− and Rbp4hRBP4orf/hRBP4orf retinas, quantitative analyses of several retinal properties were conducted. The Rbp4−/− retina showed normal retinal thickness (Figure 5a), but reduced outer nuclear layer (ONL) thickness (Figure 5c). There were also tendencies toward reduced inner nuclear layer (INL) and outer segment (OS) thickness in Rbp4hRBP4orf/hRBP4orf (Figure 5b and d). However, these differences were not statistically different, likely because of the considerable variation in retinal properties of Rbp4−/− mice, as indicated by large s.d. values. In any case, the retinal properties in Rbp4hRBP4orf/hRBP4orf were restored to normal (Figure 5a–d).
Measurement of retinal properties. (a) Retinal thickness from GCL to OSL. (b) INL thickness. The INL thickness was significantly decreased in the Rbp4−/− mice. (c) ONL thickness. (d) OS thickness. There was a tendency towards reduced INL and OS thickness in Rbp4hRBP4/hRBP4 mice. Data are presented as means±s.d.
Transmission Electron Microscopy
To analyze the retinal morphology more in detail, we performed TEM at 7 and 40 weeks of age. As in the histological analyses, the TEM data were essentially the same at 7 and 40 weeks of age; thus the data at 40 weeks of age are shown in Figure 6. The retina was thinner in a Rbp4−/− mice (Figure 6b and e) than in Rbp4+/+ mice (Figure 6a and d). The distance between the inner limiting membrane and outer plexiform layer (OPL) in Rbp4−/− mice (Figure 6b) was much shorter than that in Rbp4+/+ (Figure 6a) or Rbp4hRBP4orf/hRBP4orf (Figure 6c) mice, with fewer retinal ganglion cells (Figure 6b). In the inner plexiform layer (IPL), bipolar axons, amacrine dendrites and ganglion cell dendrites were fewer in one Rbp4−/− mice (Figure 6h) than in Rbp4+/+ (Figure 6g) or Rbp4hRBP4orf/hRBP4orf mice (Figure 6i). The electron density in the OPL (a layer of neuronal synapses) was very low in Rbp4−/− mice (Figure 6k) compared with Rbp4+/+ (Figure 6j) or Rbp4hRBP4orf/hRBP4orf (Figure 6l) mice. The Golgi apparatus was swollen in photoreceptor cell of Rbp4−/− (Figure 6n), but not in Rbp4+/+ (Figure 6m) or Rbp4hRBP4orf/hRBP4orf (Figure 6o) mice. Although the number of mitochondria in photoreceptor cell was similar among three strains, there were very few cristae in Rbp4−/− mice (Figure 6q) compared with Rbp4+/+ (Figure 6p) or Rbp4hRBP4orf/hRBP4orf (Figure 6r) mice. Overall, the TEM abnormalities observed in Rbp4−/− were not found in Rbp4hRBP4orf/hRBP4orf mice.
TEM. (a–c) GCL to INL. The distance between the inner limiting membrane and the OPL was much shorter in Rbp4−/− mice (b). (d–f) ONL to Sc. An Rbp4−/− mouse showed losses of the ONL and the IS/OS (e). (g–i) IPL. Fewer synapses were found in the IPL (g). (j–l) INL and OPL. The electron density in the OPL (surrounded by a white line) was very low (j). (m–o) Golgi apparatus in a photoreceptor cell. The Golgi apparatus (arrow) was swollen in Rbp4−/− mice (n). (p–r) Mitochondria in a photoreceptor cell. There were very few cristae in Rbp4−/− mice (q). Abnormalities found in an Rbp4−/− mouse were almost completely restored in an Rbp4hRBP4/hRBP4 mouse. GCL, ganglion cell layer; IPL, inner plexiform layer; INL, inner nuclear layer; OPL, outer plexiform layer; ONL, outer nuclear layer; IS, inner segments; OS, outer segments; RPE, retinal pigment epithelium; Ch, choroid; Sc, sclera.
Temporal Expression of Rbp4 and hRBP4
As the retinal phenotypes were already rescued at 7 weeks of age, we analyzed the temporal expression of mRbp4 and hRBP4. RNAs extracted from the liver and eyes was used in qRT-PCR analyses. In the liver, the mRbp4 levels at 6 and 12 weeks of age were 76 and 102% of the level (100%) at 24 weeks (Figure 7a, left), and the hRBP4 levels at 6 and 12 weeks were 61 and 95% of the level at 24 weeks (Figure 7a, right). For mRbp4 and hRBP4, there were significant differences in expression between 6 and 12 weeks and between 6 and 24 weeks, but not between 12 and 24 weeks. These data suggest that mRbp4 and hRBP4 have similar temporal expression patterns and that the levels of mRbp4 and human RBP4 reached a maximum at approximately 12 weeks of age.
Temporal expression of mRbp4 and hRBP4. The expression level at 24 weeks of age was set as 1.0 in all the experiments. (a) Expression in liver. (b) Expression in eyes. (c) Serum levels of mRbp4 and hRBP4. The mRbp4 and hRBP4 expression profiles in the liver and serum were similar, while hRBP4 expression levels in the eyes were lower at 6 and 12 weeks of age. Data are presented as means±s.d.
In the eyes, the mRbp4 levels at 6 and 12 weeks were 42 and 79% compared with that at 24 weeks (Figure 7b, left). There was a significant difference in expression between 6 and 12 and 6 and 24 weeks, but not between 12 and 24 weeks. In contrast, the hRBP4 levels at 6 and 12 weeks were 12 and 45% compared with that at 24 weeks (Figure 7b, right), with significant differences between 6 and 12 weeks, 12 and 24 weeks, and 6 and 24 weeks. These results suggested that mRbp4 reached a maximum level at 12 weeks, whereas hRBP4 expression was very low at 6 weeks and increased gradually up to 24 weeks.
At 6, 12, and 24 weeks of age, the serum mRBP4 levels in Rbp4+/+ mice were 21.5±2.03, 24.7±2.37, and 25.0±2.94 μg/ml (Figure 7c, left) and those of hRBP4 in Rbp4hRBP4/hRBP4 mice were 5.9±0.55, 7.4±1.50, and 7.8±0.53 μg/ml (Figure 7c, right). Thus, the hRBP4 levels in Rbp4hRBP4/hRBP4 mice at 6, 12, and 24 weeks were 27%, 30%, and 31%, respectively, compared with those of mRBP4 in Rbp4+/+ mice. For mRBP4 and hRBP4, there were significant differences in expression between 6 and 12 and 6 and 24 weeks, but not between 12 and 24 weeks. These results are consistent with the RNA expression levels in the liver, with mRbp4 and hRBP4 expression both reaching maxima at 12 weeks.
Retinol Levels in the Plasma and Liver
Previously, we showed that plasma retinol levels were below the detection threshold (<5 IU/dl), whereas liver retinol levels were increased in Rbp4−/− mice.15 To examine the retinol status in Rbp4hRBP4orf/hRBP4orf mice, retinol levels in the plasma and liver from Rbp4+/+, Rbp4−/−, and Rbp4hRBP4orf/hRBP4orf mice were determined. The plasma levels of retinol in these mice were 41.3±10.1, <5, and 16.3±4.2 IU/dl, respectively, at 8 weeks of age, and 97±4.4, <5, and 24.7±5.1 IU/dl, respectively, at 30 weeks (Figure 8, left). Thus, in Rbp4hRBP4orf/hRBP4orf mice, the plasma retinol levels were 39% and 25% of those in Rbp4+/+ mice at 8 and 30 weeks, respectively.
The liver retinol levels in Rbp4+/+, Rbp4−/−, and Rbp4hRBP4orf/hRBP4orf mice were 59.5±6.5, 60.6±0.6, and 55.9±4.7 mg/100 g, respectively, at 8 weeks and 153.3±3.5, 185.7±2.5, and 169.7±5.0 mg/100 g, respectively, at 30 weeks (Figure 8, right). Thus, at 8 weeks, the liver retinol levels were similar in the three strains, which indicated that the low plasma retinol level did not affect hepatic retinol stores. The liver retinol level at 30 weeks was highest in Rbp4−/−, followed by Rbp4hRBP4orf/hRBP4orf and Rbp4+/+ mice.
Expression of TTR and RBP4 in Liver and Serum
To examine whether mTTR expression in liver or serum was altered in Rbp4+/+, Rbp4−/− and Rbp4hRBP4orf/hRBP4orf mice, northern blot and western blot analyses were performed. As shown in Figure 9a, the mRNA and protein levels of mTTR in liver were similar among these strains. We also measured the mRBP4 or hRBP4 level in liver of Rbp4+/+ or Rbp4hRBP4orf/hRBP4orf mice, respectively. The observed concentrations were 32.9±1.2 mg/g protein and 10.6±0.38 mg/g protein, respectively. These data are consistent with those for serum RBP4 concentrations. Namely, human RBP4 levels in Rbp4hRBP4orf/hRBP4orf mice were one-third of the RBP4 levels in Rbp4+/+ mice. In addition, the serum levels of mTTR were also found to be similar among these strains as demonstrated by western blot analyses (Figure 9b). At 12 weeks, the serum levels in Rbp4+/+, Rbp4−/−, and Rbp4hRBP4orf/hRBP4orf mice were 306±10.2, 306±5.8, and 305±4.8 μg/ml, respectively. There were no significant differences observed for these levels among mouse strains (Figure 9b). Taken together, these results suggest that mTTR expression was not affected in Rbp4−/− and Rbp4hRBP4orf/hRBP4orf mice.
Gel Filtration Chromatography
Rbp4+/+ and Rbp4hRBP4orf/hRBP4orf plasma samples were subjected to gel filtration chromatography, and column fractions were analyzed by western blotting. The majority of immunoreactive RBP4 and TTR eluted together in fractions 28 to 31 in both the strains. A small amount of immunoreactive RBP4 eluted after this primary peak in subsequent fractions 32 to 39 (Figure 10). These secondary fractions represent non-TTR-bound RBP4 because no associated TTR immunoreactivity was observed. The percentage of the TTR-RBP4 bound form in RbphRBP4orf/hRBP4orf mice (90.1±1.8) was higher than in Rbp4+/+ mice (82.0±2.2; Figure 10). Although these results suggest that the affinity of hRBP4 for mouse TTR was slightly higher than mRBP4 for mTTR, this could be due to the lower hRBP4 concentration than the mRBP4 concentration.
Gel filtration chromatography. Each fraction was analyzed by western blotting using anti-RBP4 and TTR antibodies. The bound form of RBP4 and TTR was found in fractions 28–31, whereas the free form of RBP4 was found in fractions 32–39. The percentage of the TTR-RBP4 bound form in Rbp4hRBP4/hRBP4 mice was higher than in Rbp4+/+ mice. **P<0.01.
Rd8 Mutation
As reported by Mattapallil et al,27 the rd8 mutation in the Crb1 gene was detected in the C57BL/6N substrain, but not in the C57BL/6J substrain. This mutation results in ocular spots and retinal lesions such as retinal folding, dysplasia of the nuclear layers, retinal degeneration, and vacuolation of the retinal pigment epithelium.27, 28 To rule out the possibility that Rbp4hRBP4/hRBP4 mice carried the rd8 mutation, we analyzed this mutation in our mice. We confirmed that none of the mice used in our morphological and functional analyses of the retina had the rd8 mutation (Supplementary Figure S2).
Discussion
In this study, we established the mouse strain, Rbp4hRBP4orf/hRBP4orf mouse strain with a normal human RBP4orf at the mouse Rbp4 locus. As the serum hRBP4 levels in Rbp4hRBP4orf/hRBP4orf mice were approximately 30% of those of mRBP4 in Rbp4+/+ mice, we used this mouse strain to examine whether this level of hRBP4 expression can rescue phenotypes observed in Rbp4−/− mice. This result will provide valuable information regarding gene therapy for single-gene disorders such as inborn errors of metabolism. Indeed, the retinal impairment and morphological abnormalities observed in Rbp4−/− mice were almost completely rescued in Rbp4hRBP4orf/hRBP4orf mice.
The hRBP4orf with a PGK-puro cassette was inserted into the mouse endogenous Rbp4 gene locus. The hRBP4orf in the replaced allele was expressed in similar tissues, including the liver, testis, and lung, as those for the mRbp4 gene with different expression levels in some tissues. Temporal expression patterns of mRbp4 and hRBP4 were similar in the liver. However, in the eyes, hRBP expression was low at 6 and 12 weeks of age but increased substantially at 24 weeks. The reasons for the moderately altered expression patterns are unclear, but the integration of hRBP4 with a PGK-puro cassette may affect the spatial, temporal, and quantitative expression of hRBP4 in the replaced allele.
The plasma retinol level in Rbp4RBP4orf/RBP4orf mice was approximately one-third or one-fourth of that in Rbp4+/+ mice at 8 or 30 weeks of age, respectively. Interestingly, the plasma retinol level increased with age both in Rbp4+/+ and Rbp4RBP4orf/RBP4orf mice, although the increase rate in Rbp4RBP4orf/RBP4orf mice was much smaller than that in Rbp4+/+ mice. The liver retinol concentrations at 8 weeks of age were similar in both Rbp4+/+ mice and Rbp4RBP4orf/RBP4orf mice. In contrast, the liver retinol concentrations in Rbp4RBP4orf/RBP4orf mice were higher than those in Rbp4+/+ mice, suggesting that low hRBP4 expression resulted in high liver retinol concentrations. In any case, these findings are consistent with the fact that the plasma RBP4 concentrations in Rbp4RBP4orf/RBP4orf mice were lower than those in Rbp4+/+ mice.
The Rbp4−/− mice showed a severe impairment of retinal function and morphological abnormalities. However, our Rbp4hRBP4orf/hRBP4orf mice had almost normal ERG responses and retinal morphologies. Retinal impairment could be caused by either the loss of Rbp4 expression in the eyes or retinol deficiency in the blood. In the eyes, the level of human RBP4 at 6 weeks was 12% compared with that at 24 weeks. Nevertheless, ERG and histology were almost normal in Rbp4hRBP4orf/hRBP4orf mice at 7 weeks of age. This finding suggests that Rbp4 expression in the eyes does not have an important role in visual function. Quadro et al30 generated a transgenic mouse strain (hRBP4: mRbp4−/−) that expressed hRBP4 under the control of the muscle creatine kinase promoter on an Rbp4−/− background. Although no hRBP4 was detected in the retinal pigment epithelium of hRBP4: mRbp4−/− mice, a normal ERG profile was observed in the eyes. As plasma retinol levels were normal, circulating RBP4 efficiently delivered retinol to the eye. Thus, the ocular phenotypes in our Rbp4−/− mice may be rescued by expression of hRBP4 in the liver, which resulted in retinol delivery to the eyes. In other words, circulating retinol may restore morphological abnormalities and result in the rescue of retinal function.
Similar rescue experiments have been performed using knockout mouse models.31, 32, 33 Mpv17 mice, a model of glomerulosclerosis, that expressed the human MVP17 gene showed normal glomerular histology.32 In human p25 (C-terminal truncation product of p35) transgenic mice with a Cdk activator protein p35-null background, brain anatomy (ie, loss of the laminar structure within the cerebral cortex) was partially rescued.31 Furthermore, a 130-kilobase (kb) yeast artificial chromosome containing the human SCL (stem cell leukemia) locus completely rescued the embryonic lethal phenotype of Scl−/− mice.33 However, in these cases, the expression levels of transgenes vs mouse endogenous genes were not clearly demonstrated. There is little data on how much expression is required to rescue phenotypes in human recessively inherited diseases. Our results suggest that the 30% expression of hRBP4 in the liver is sufficient to rescue phenotypes caused by the Rbp4 mutation in our mouse model, similar to human patients.
References
D'Onofrio C, Colantuoni V, Cortese R . Structure and cell-specific expression of a cloned human retinol binding protein gene: the 5'-flanking region contains hepatoma specific transcriptional signals. EMBO J 1985; 4: 1981–1989.
Laurent BC, Nilsson MH, Bavik CO et al, Characterization of the rat retinol-binding protein gene and its comparison to the three-dimensional structure of the protein. J Biol Chem 1985; 260: 11476–11480.
Soprano DR, Soprano KJ, Goodman DS . Retinol-binding protein messenger RNA levels in the liver and in extrahepatic tissues of the rat. J Lipid Res 1986; 27: 166–171.
Davis JT, Ong DE . Synthesis and secretion of retinol-binding protein by cultured rat Sertoli cells. Biol Reprod 1992; 47: 528–533.
Davis JT, Ong DE . Retinol processing by the peritubular cell from rat testis. Biol Reprod 1995; 52: 356–364.
Jaworowski A, Fang Z, Khong TF et al, Protein synthesis and secretion by cultured retinal pigment epithelia. Biochim Biophys Acta 1995; 1245: 121–129.
Ong DE, Davis JT, O'Day WT et al, Synthesis and secretion of retinol-binding protein and transthyretin by cultured retinal pigment epithelium. Biochemistry 1994; 33: 1835–1842.
Soprano DR, Blaner WS . Plasma retinol-binding protein. In: Sporn MB, Roberts AB, Goodman DS (eds). The Retinoids: Biology, Chemistry, and Medicine, 2nd edn. Raven Press: New York, NY, USA, 1994, pp 257–281.
Goodman DS . Plasma Retinol-Binding Protein. Academic Press: New York, NY, USA, 1984.
Wolf G . The discovery of the visual function of vitamin A. J Nutr 2001; 131: 1647–1650.
Cukras C, Gaasterland T, Lee P et al, Exome analysis identified a novel mutation in the RBP4 gene in a consanguineous pedigree with retinal dystrophy and developmental abnormalities. PLoS One 2012; 7: e50205.
Seeliger MW, Biesalski HK, Wissinger B et al, Phenotype in retinol deficiency due to a hereditary defect in retinol binding protein synthesis. Invest Ophthalmol Vis Sci 1999; 40: 3–11.
Quadro L, Blaner WS, Salchow DJ et al, Impaired retinal function and vitamin A availability in mice lacking retinol-binding protein. EMBO J 1999; 18: 4633–4644.
Quadro L, Blaner WS, Hamberger L et al, Muscle expression of human retinol-binding protein (RBP). Suppression of the visual defect of RBP knockout mice. J Biol Chem 2002; 277: 30191–30197.
Shen J, Shi D, Suzuki T et al, Severe ocular phenotypes in Rbp4-deficient mice in the C57BL/6 genetic background. Lab Invest 2016; 96: 680–691.
Berni R, Malpeli G, Folli C et al, The Ile-84—>Ser amino acid substitution in transthyretin interferes with the interaction with plasma retinol-binding protein. J Biol Chem 1994; 269: 23395–23398.
Episkopou V, Maeda S, Nishiguchi S et al, Disruption of the transthyretin gene results in mice with depressed levels of plasma retinol and thyroid hormone. Proc Natl Acad Sci USA 1993; 90: 2375–2379.
Melhus H, Nilsson T, Peterson PA et al, Retinol-binding protein and transthyretin expressed in HeLa cells form a complex in the endoplasmic reticulum in both the absence and the presence of retinol. Exp Cell Res 1991; 197: 119–124.
Wei S, Episkopou V, Piantedosi R et al, Studies on the metabolism of retinol and retinol-binding protein in transthyretin-deficient mice produced by homologous recombination. J Biol Chem 1995; 270: 866–870.
Zhao G, Li Z, Araki K et al, Inconsistency between hepatic expression and serum concentration of transthyretin in mice humanized at the transthyretin locus. Genes Cells 2008; 13: 1257–1268.
Yagi T, Tokunaga T, Furuta Y et al, A novel ES cell line, TT2, with high germline-differentiating potency. Anal Biochem 1993; 214: 70–76.
Araki K, Imaizumi T, Okuyama K et al, Efficiency of recombination by Cre transient expression in embryonic stem cells: comparison of various promoters. J Biochem 1997; 122: 977–982.
Li Z, Zhao G, Shen J et al, Enhanced expression of human cDNA by phosphoglycerate kinase promoter-puromycin cassette in the mouse transthyretin locus. Transgenic Res 2011; 20: 191–200.
Sato H, Suzuki T, Ikeda K et al, A monogenic dominant mutation in Rom1 generated by N-ethyl-N-nitrosourea mutagenesis causes retinal degeneration in mice. Mol Vis 2010; 16: 378–391.
Notsumoto S, Kobayashi K, Hirauchi K et al, Determination of retinol in serum by high-performance liquid chromatograpgy with fluorometric detection using a column-switching system. Clin Chem 1993; 22: 190–194.
Thompson LB, Schimpf KJ, Stiner LA et al, Determination of vitamin A (retinol) in infant and medical nutritional formulas with AOAC method 992.06 using a modified extraction procedure: single-laboratory validation. J AOAC Int 2010; 93: 1523–1529.
Mattapallil MJ, Wawrousek EF, Chan CC et al, The Rd8 mutation of the Crb1 gene is present in vendor lines of C57BL/6N mice and embryonic stem cells, and confounds ocular induced mutant phenotypes. Invest Ophthalmol Vis Sci 2012; 53: 2921–2927.
Mehalow AK, Kameya S, Smith RS et al, CRB1 is essential for external limiting membrane integrity and photoreceptor morphogenesis in the mammalian retina. Hum Mol Genet 2003; 12: 2179–2189.
Reixach N, Foss TR, Santelli E et al, Human-murine transthyretin heterotetramers are kinetically stable and non-amyloidogenic: a lesson in the generation of transgenic models of diseases involving oligomeric proteins. J Biol Chem 2007; 283: 2098–2107.
Quadro L, Hamberger L, Colantuoni V et al, Understanding the physiological role of retinol-binding protein in vitamin A metabolism using transgenic and knockout mouse models. Mol Aspects Med 2003; 24: 421–430.
Patzke H, Maddineni U, Ayala R et al, Partial rescue of the p35−/− brain phenotype by low expression of a neuronal-specific enolase p25 transgene. J Neurosci 2003; 23: 2769–2778.
Schenkel J, Zwacka RM, Rutenberg C et al, Functional rescue of the glomerulosclerosis phenotype in Mpv17 mice by transgenesis with the human Mpv17 homologue. Kidney Int 1995; 48: 80–84.
Sinclair AM, Bench AJ, Bloor AJ et al, Rescue of the lethal scl(−/−) phenotype by the human SCL locus. Blood 2002; 99: 3931–3938.
Acknowledgements
We thank Kyoko Ikeda and Shundan Jin for their assistance with eye morphological and functional analyses. This work was supported by a Grant-in-Aid for Scientific Research (C) (Grant number 24590404; Japan Society for the Promotion of Science to ZL), and Grant-in-Aid for Scientific Research (S) (Grant number 21220010; Japan Society for the Promotion of Science), Scientific Support Programs for Cancer Research Grant-in-Aid for Scientific Research on Innovative Areas (Grant number 221S0001; Ministry of Education, Culture, Sports, Science and Technology), CREST (JST), and a research grant from TransGenic (to KY).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Laboratory Investigation website
Retinol-binding protein RBP4 is the specific carrier for retinol in the blood. The authors produced a humanized Rbp4hRBP4/ hRBP4 mouse in which the serum hRBP4 level is approximately 30% of that in Rbp4+/+ mice. The electroretinogram and morphological abnormalities observed in Rbp4-/- animals are not detected in the humanized mice.
Supplementary information
Rights and permissions
About this article
Cite this article
Liu, L., Suzuki, T., Shen, J. et al. Rescue of retinal morphology and function in a humanized mouse at the mouse retinol-binding protein locus. Lab Invest 97, 395–408 (2017). https://doi.org/10.1038/labinvest.2016.156
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/labinvest.2016.156
This article is cited by
-
Amyloid deposition in a mouse model humanized at the transthyretin and retinol-binding protein 4 loci
Laboratory Investigation (2018)