Abstract
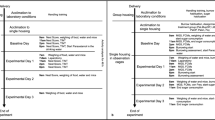
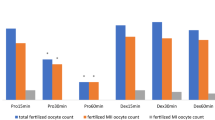
Embryo transfer is a surgical technique that is widely used in reproductive biotechnology. Despite the ethical obligation to relieve animals' post-operative pain, analgesia is not routinely provided after embryo transfer surgery because it has been suggested that analgesics may be detrimental to embryo survival. Studies suggest, however, that the potential for adverse effects varies depending on the type of analgesic used and the timing of its administration. The authors carried out a study to determine whether pre-operatively administered tramadol, a synthetic analogue of codeine, influenced birth rate, litter survival or the post-operative body weights of surrogate dams. Compared with controls that were not given any analgesic, surrogate dams given tramadol had similar birth rates and similar body weights at all time points. The tramadol-treated surrogate dams showed a statistically significant increase in the number of offspring that survived to weaning. The authors conclude that pre-operatively administered tramadol does not harm the success rate of embryo transfer surgery and even may improve litter survival.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
We are sorry, but there is no personal subscription option available for your country.
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Goulding, D.R. et al. The effects of perioperative analgesia on litter size in Crl:CD1(ICR) mice undergoing embryo transfer. J. Am. Assoc. Lab. Anim. Sci. 49, 423–426 (2010).
Parker, J.M., Austin, J., Wilkerson, J. & Carbone, L. Effects of multimodal analgesia on the success of mouse embryo transfer surgery. J. Am. Assoc. Lab. Anim. Sci. 50, 466–470 (2011).
American College of Laboratory Animal Medicine. Guidelines for the Assessment and Management of Pain in Rodents and Rabbits (Public Statement, ACLAM, 2007). (http://www.aclam.org/Content/files/files/Public/Active/position_pain-rodent-rabbit.pdf).
Lee-Parritz, D. Analgesia for rodent experimental surgery. Isr. J. Vet. Med. 62, 3–4 (2007).
Interagency Research Animal Committee. U.S. Government Principles for the Utilization and Care of Vertebrate Animals Used in Testing, Research, and Training (Office of Science and Technology Policy, Washington, DC, 1985).
Yentis, S., May, A. & Malhotra, S. Analgesia, Anaesthesia and Pregnancy: A Practical Guide 2nd edn. (Cambridge University Press, Cambridge, UK, 2007).
Kona-Boun, J.J., Silim, A. & Troncy, E. Immunologic aspects of veterinary anesthesia and analgesia. J. Am. Vet. Med. Assoc. 226, 355–363 (2005).
McGuire, L. et al. Pain and wound healing in surgical patients. Ann. Behav. Med. 31, 165–172 (2006).
Institute for Laboratory Animal Research. Guide for the Care and Use of Laboratory Animals 8th edn. (National Academies Press, Washington, DC, 2011).
Joshi, G.P. & Ogunnaike, B.O. Consequences of inadequate postoperative pain relief and chronic persistent postoperative pain. Anesthesiol. Clin. North America 23, 21–36 (2005).
Thaete, L.G., Levin, S.I. & Dudley, A.T. Impact of anesthetics and analgesics on fetal growth in the mouse. Lab. Anim. 47, 175–183 (2013).
Chiang, C.N. & Lee, C.C. (eds.) Prenatal Drug Exposure: Kinetics and Dynamics, NIDA Research Monograph Series #60 (U.S. Department of Health and Human Services, Rockville, MD, 1985).
Simmons, R.D., Miller, R.K. & Kellogg, C.K. Prenatal diazepam: distribution and metabolism in perinatal rats. Teratology 28, 181–188 (1983).
Littleford, J. Effects on the fetus and newborn of maternal analgesia and anesthesia: a review. Can. J. Anaesth. 51, 586–609 (2004).
Parboosingh, J. The effects of medication during pregnancy. Can. Fam. Physician 27, 1013–1015 (1981).
Dorsch, M.M., Otto, K. & Hedrich, H.J. Does preoperative administration of metamizol (Novalgin) affect postoperative body weight and duration of recovery from ketamine-xylazine anaesthesia in mice undergoing embryo transfer: a preliminary report. Lab. Anim. 38, 44–49 (2004).
Valle, M., Garrido, M.J., Pavón, J.M., Calvo, R. & Trocóniz, I.F. Pharmacokinetic-pharmacodynamic modeling of the antinociceptive effects of main active metabolites of tramadol, (+)-O-desmethyltramadol and (−)-O-desmethyltramadol, in rats. J. Pharmacol. Exp. Ther. 293, 646–653 (2000).
Hummel, T. et al. Assessment of analgesia in man: tramadol controlled release formula vs. tramadol standard formulation. Eur. J. Clin. Pharmacol. 51, 31–38 (1996).
Miranda, H.F. & Pinardi, G. Antinociception, tolerance, and physical dependence comparison between morphine and tramadol. Pharmacol. Biochem. Behav. 61, 357–360 (1998).
Gebhardt, G.F. & Schmidt, R.F. (eds.) Encyclopedia of Pain 2nd edn. (Springer, New York, 2007).
Hennies, H.H., Friderichs, E. & Schneider, J. Receptor binding, analgesic and antitussive potency of tramadol and other selected opioids. Arzneimittelforschung 38, 877–880 (1988).
Raffa, R.B. & Fridericks, E. The basic science aspect of tramadol hydrochloride. Pain Rev. 3, 249–271 (1996).
Raffa, R.B. et al. Opioid and non-opioid components independently contribute to the mechanism of action of tramadol, an 'atypical' opioid analgesic. J. Pharmacol. Exp. Ther. 260, 275–285 (1992).
Bamigbade, T.A., Davidson, C., Langford, R.M. & Stamford, J.A. Actions of tramadol, its enantiomers and principle metabolite, O-desmethyltramadol on serotonin (5-HT) efflux and uptake in the rat dorsal raphe nucleus. Br. J. Anaesth. 79, 352–356 (1997).
Grond, S. & Sablotzki, A. Clinical pharmacology of tramadol. Clin. Pharmacokinet. 43, 879–923 (2004).
Symeon, E. et al. Evaluation of the analgesic effect of tramadol in two different strains of mice. Proceedings of the 35th Scientific Conference of the Hellenic Society for Biological Science (Nafplio, Greece, 23–25 May 2013).
Coetzee, J.F. & van Loggerenberg, H. Tramadol or morphine administered during operation: a study of immediate postoperative effects after abdominal hysterectomy. Br. J. Anaesth. 81, 737–741 (1998).
Houmes, R.J. et al. Efficacy and safety of tramadol versus morphine for moderate and severe postoperative pain with special regard to respiratory depression. Anesth. Analg. 74, 510–514 (1992).
Matthiesen, T., Wöhrmann, T., Coogan, T.P. & Uragg, H. The experimental toxicology of tramadol: an overview. Toxicol. Lett. 95, 63–71 (1998).
Hariharan, S., Moseley, H., Kumar, A. & Raju, S. The effect of preemptive analgesia in postoperative pain relief—a prospective double-blind randomized study. Pain Med. 10, 49–53 (2009).
Kissin, I. Preemptive analgesia at the crossroad. Anesth. Analg. 100, 754–756 (2005).
Baumans, V. Science-based assessment of animal welfare: laboratory animals. Rev. Sci. Tech. 24, 503–513 (2005).
Broom, D.M. Animal welfare: concepts and measurement. J. Anim. Sci. 69, 4167–4175 (1991).
Directive 2010/63/EU of the European Parliament and of the Council of 22 September 2010 on the protection of animals used for scientific purposes. Official Journal of the European Union 276, 33–79 (2010).
Nicklas, W. et al. Recommendations for the health monitoring of rodent and rabbit colonies in breeding and experimental units. Lab. Anim. 36, 20–42 (2002).
American Veterinary Medical Association. Guidelines for the Euthanasia of Animals: 2013 Edition (AVMA, Schaumberg, IL, 2013). (https://www.avma.org/kb/policies/documents/euthanasia.pdf).
Rätsep, M.T., Barrette, V.F., Winterborn, A., Adams, M.A. & Croy, B.A. Hemodynamic and behavioral differences after administration of meloxicam, buprenorphine, or tramadol as analgesics for telemeter implantation in mice. J. Am. Assoc. Lab. Anim. Sci. 52, 560–566 (2013).
Liles, J.H., Flecknell, P.A., Roughan, J. & Cruz-Madorran, I. Influence of oral buprenorphine, oral naltrexone or morphine on the effects of laparotomy in the rat. Lab. Anim. 32, 149–161 (1998).
Wiesenfeld-Hallin, Z. Sex differences in pain perception. Gend. Med. 2, 137–145 (2005).
McLaren, A. & Michie, D. Studies on the transfer of fertilized mouse eggs to uterine foster-mothers. I. Factors affecting the implantation and survival of native and transferred eggs. J. Exp. Biol. 33, 394–416 (1956).
Nagy, A., Gertsenstein, M., Vintersten, K. & Behringer, R. Manipulating the Mouse Embryo: A Laboratory Manual 3rd edn. (Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY, 2003).
Auerbach, A.B. et al. Strain-dependent differences in the efficiency of transgenic mouse production. Transgenic Res. 12, 59–69 (2003).
Bagis, H., Odaman Mercan, H. & Dinnyes, A. Exposure to warmer postoperative temperatures reduces hypothermia caused by anaesthesia and significantly increases the implantation rate of transferred embryos in the mouse. Lab. Anim. 38, 50–54 (2004).
Luo, C. et al. Superovulation strategies for 6 commonly used mouse strains. J. Am. Assoc. Lab. Anim. Sci. 50, 471–478 (2011).
Kamjoo, M., Brison, D.R. & Kimber, S.J. Apoptosis in the preimplantation mouse embryo: effect of strain difference and in vitro culture. Mol. Reprod. Dev. 61, 67–77 (2002).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Koutroli, E., Alexakos, P., Kakazanis, Z. et al. Effects of using the analgesic tramadol in mice undergoing embryo transfer surgery. Lab Anim 43, 167–172 (2014). https://doi.org/10.1038/laban.518
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/laban.518