Abstract
Objective:
In 2001, the US Food and Drug Administration approved recombinant tissue plasminogen activator (alteplase, Cathflo Activase) to reestablish patency of central catheters occluded, presumably, by a fibrin clot. We conducted a multicenter quality improvement study to determine the value of this procedure in our Neonatal Intensive Care Unit (NICUs), including analyses of efficacy, safety and costs.
Study Design:
We conducted a retrospective quality analysis of neonates in level III NICUs, who received alteplase for the purpose of reestablishing patency of occluded central catheters.
Results:
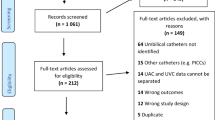
Alteplase was administered to 169 neonates, each given one to four doses, totaling 205 episodes of administration. The most common type of catheter where alteplase was used was percutaneously inserted central catheter (PICC) lines (78% of uses), 8% were umbilical venous catheters (UVCs), 6% arterial lines, 5% chest tubes and 3% other catheters. Postnatal age at first dose ranged from 0 to 132 days (median, 12); dosed patients were 22 to 41 weeks gestation at birth (median, 31). Fifty-eight percentage of administrations restored catheter function. Success was more likely at younger postnatal age (10±2 days old in successful vs 14±1 days in unsuccessful treatments; P=0.023). Seventy-two percentage of the re-canalized catheters remained functional until they were no longer needed (2 to 30 days later). Nine percentage of episodes were treated with a second dose 1 to 17 days later for re-occlusion and 50% of those were successful. Bleeding consequences were identified in only one case, where three separate lines were treated (chest tube, PICC and UVC) within a 6-h period. Costs to the health system of doses, minus savings to the system by not needing to replace lines, averaged a net of $34 per dose.
Conclusions:
The apparent safety and favorable value analysis prompted us to develop a consistent approach to alteplase usage in the Intermountain Healthcare NICUs, using the data in this report to standardize the guidelines across our health system.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Hébert M, Lesept F, Vivien D, Macrez R . The story of an exceptional serine protease, tissue-type plasminogen activator (tPA). Rev Neurol (Paris) 2016; 172 (3): 186–197.
Ansah DA, Patel KN, Montegna L, Nicholson GT, Ehrlich AC, Petit CJ . Tissue plasminogen activator use in children: bleeding complications and thrombus resolution. J Pediatr 2016; 171: 67–72.e2.
Smith LH . Alteplase for the management of occluded central venous access devices: safety considerations. Clin J Oncol Nurs 2008; 12 (1): 155–157.
Zivin JA . Acute stroke therapy with tissue plasminogen activator (tPA) since it was approved by the U.S. Food and Drug Administration (FDA). Ann Neurol 2009; 66 (1): 6–10.
Raad II, Hanna HA . Intravascular catheter-related infections: new horizons and recent advances. Arch Intern Med 2002; 162 (8): 871–878.
Blaney M, Shen V, Kerner JA, Jacobs BR, Gray S, Armfield J et al. Alteplase for the treatment of central venous catheter occlusion in children: results of a prospective, open-label, single-arm study (The Cathflo Activase Pediatric Study). J Vasc Interv Radiol 2006; 17: 1745–1751.
Dunn AL, Abshire TC . Recombinant tissue plasminogen activator may reduce frequency of central venous access device infection in hemophilia patients undergoing immune tolerance therapy. Pediatr Blood Cancer 2008; 50 (3): 627–629.
Anderson D, Pesaturo K, Casavant J, Ramsey E . Alteplase for the treatment of catheter occlusion in pediatric patients. Ann Pharm 2013; 47: 405–410.
Haas N, Haas S . Central venous catheter techniques in infants and children. Curr Opin Anaesthesiol 2003; 16: 291–303.
Jacobs B, Haygood M, Hingl J . Recombinant tissue plasminogen activator in the treatment of central venous catheter occlusion in children. J Pediatr 2001; 139: 593–596.
Lee E . Alteplase use for prevention of central line occlusion in a preterm infant. Ann Pharm 2002; 36: 272–274.
Soylu H, Brandao L, Lee K . Efficacy of local instillation of recombinant tissue plasminogen activator for restorting occluded central venous catheters in neonates. J Pediatr 2009; 156 (2): 197–201.
Sharma RP, Ree CJ, Ree A . Efficacy and safety of a single 2 mg dose or 4 mg double dose of alteplase for 50 occluded chest ports using a unique instillation technique. Int J Angiol 2008; 17 (3): 125–128.
Gilarde JA, Chung AM, Vidal R, Falkos S . Efficacy and economic evaluation of a volume-based cathflo activase protocol versus a fixed-dose alteplase protocol for catheter occlusions in pediatric patients. J Pediatr Pharmacol Ther 2006; 11 (4): 237–244.
Ernst FR, Chen E, Lipkin C, Tayama D, Amin AN . Comparison of hospital length of stay, costs, and readmissions of alteplase versus catheter replacement among patients with occluded central venous catheters. J Hosp Med 2014; 9 (8): 490–496.
Thakarar K, Collins M, Kwong L, Sulis C, Korn C, Bhadelia N . The role of tissue plasminogen activator use and systemic hypercoagulability in central line-associated bloodstream infections. Am J Infect Control 2014; 42: 417–420.
CathfloTM Activase® (Alteplase) [packasge insert]. Genentech, Inc.: South San Francisco, CA, USA, 2001.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Supplementary information
Rights and permissions
About this article
Cite this article
Scott, D., Ling, C., MacQueen, B. et al. Recombinant tissue plasminogen activator to restore catheter patency: efficacy and safety analysis from a multihospital NICU system. J Perinatol 37, 291–295 (2017). https://doi.org/10.1038/jp.2016.203
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/jp.2016.203
This article is cited by
-
Alteplase
Reactions Weekly (2017)