Abstract
Objective:
The objective of this study was to evaluate the occurrence of adverse effects during surfactant delivery, using a standardized protocol for administration and management of complications.
Study Design:
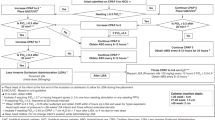
The protocol was developed, implemented and used for 6 months. Vital signs and ventilatory parameters were prospectively recorded during the procedure. Infants were classified into three groups, based on the occurrence and severity of complications: no, minor or major.
Result:
A total of 39 infants received surfactant and 19 presented some complication: 11 minor and 8 major. Six of the major complications were episodes of severe airway obstruction (SAO) and five occurred in extreme low birth weight (ELBW) infants that had more severe lung disease before surfactant delivery. Two cases of persistent pulmonary hypertension occurred in infants with birth weight>1000 g.
Conclusion:
This study identified a high rate of SAO and provides data to support changes in the protocol, which should include faster and more robust increases in positive inspiratory pressures in ELBW infants presenting with SAO.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Halliday HL . Surfactants: past, present and future. J Perinatol 2008; 28 (Suppl 1): S47–S56.
Hermeto F, Bottino MN, Vaillancourt K, Sant’Anna GM . Implementation of a respiratory therapist-driven protocol for neonatal ventilation: impact on the premature population. Pediatrics 2009; 123: e907–e916.
Kero PO, Makinen EO . Comparison between clinical and radiological classification of infants with the respiratory distress syndrome (RDS). Eur J Pediatr 1979; 130: 271–278.
Enhorning G, Shennan A, Possmayer F, Dunn M, Chen CP, Milligan J . Prevention of neonatal respiratory distress syndrome by tracheal instillation of surfactant: a randomized clinical trial. Pediatrics 1985; 76: 145–153.
Dunn MS, Shennan AT, Zayack D, Possmayer F . Bovine surfactant replacement therapy in neonates of less than 30 weeks’ gestation: a randomized controlled trial of prophylaxis versus treatment. Pediatrics 1991; 87: 377–386.
Notter RH . Exogenous lung surfactant: current and future. Lung Surfactants: basic science and clinical applications. In: Lenfant C (ed). Marcel Dekker, Inc.: New York, 2000 pp 319–344.
Proquitte H, Dushe T, Hammer H, Rudiger M, Schmalisch G, Wauer RR . Observational study to compare the clinical efficacy of the natural surfactants Alveofact and Curosurf in the treatment of respiratory distress syndrome in premature infants. Respir Med 2007; 101: 169–176.
Bloom BT, Clark RH . Comparison of Infasurf (calfactant) and Survanta (beractant) in the prevention and treatment of respiratory distress syndrome. Pediatrics 2005; 116: 392–399.
Hudak ML, Martin DJ, Egan EA, Matteson EJ, Cummings NJ, Jung AL et al. A multicenter randomized masked comparison trial of synthetic surfactant versus calf lung surfactant extract in the prevention of neonatal respiratory distress syndrome. Pediatrics 1997; 100: 39–50.
Peliowski A . A randomized, controlled, double blinded multicenter trial to compare synthetic surfactant (Exosurf) with a bovine surfactant (Bovine Lipid Extract Surfactant) for the treatment of respiratory distress syndrome of the newborn. 2002. Monograph..
Lam BC, Ng YK, Wong KY . Randomized trial comparing two natural surfactants (Survanta vs. bLES) for treatment of neonatal respiratory distress syndrome. Pediatr Pulmonol 2005; 39: 64–69.
Engle WA . Surfactant-replacement therapy for respiratory distress in the preterm and term neonate. Pediatrics 2008; 121: 419–432.
Fernandez-Ruanova MB, Alvarez FJ, Gastiasoro E, Arnaiz A, Robertson B, Curstedt T et al. Comparison of rapid bolus instillation with simplified slow administration of surfactant in lung lavaged rats. Pediatr Pulmonol 1998; 26: 129–134.
Hentschel R, Brune T, Franke N, Harms E, Jorch G . Sequential changes in compliance and resistance after bolus administration or slow infusion of surfactant in preterm infants. Intensive Care Med 2002; 28: 622–628.
Hentschel R, Jorch G . Acute side effects of surfactant treatment. J Perinat Med 2002; 30: 143–148.
Wheeler KI, Davis PG, Kamlin COF, Morley CJ . Assist control volume guarantee ventilation during surfactant administration. Arch Dis Child Fetal Neonatal Ed 2009; 94: F336–F338.
Metcalfe IL, Burgoyne R, Enhorning G . Surfactant supplementation in the preterm rabbit: effects of applied volume on compliance and survival. Pediatr Res 1982; 16: 834–839.
Goldsmith LS, Greenspan JS, Rubenstein SD, Wolfson MR, Shaffer TH . Immediate improvement in lung volume after exogenous surfactant: alveolar recruitment versus increased distention. J Pediatr 1991; 119: 424–428.
Dinger J, Topfer A, Schaller P, Schwarze R . Functional residual capacity and compliance of the respiratory system after surfactant treatment in premature infants with severe respiratory distress syndrome. Eur J Pediatr 2002; 161: 485–490.
Manczur T, Greenough A, Nicholson GP, Rafferty GF . Resistance of pediatric and neonatal endotracheal tubes: influence of flow rate, size, and shape. Crit Care Med 2000; 28: 1595–1598.
Krause M, Olsson T, Law AB, Parker RA, Lindstrom DP, Sundell HW et al. Effect of volume recruitment on response to surfactant treatment in rabbits with lung injury. Am J Respir Crit Care Med 1997; 156: 862–866.
Krause MF, Jakel C, Haberstroh J, Schulte-Monting J, Leititis JU, Orlowska-Volk M . Alveolar recruitment promotes homogeneous surfactant distribution in a piglet model of lung injury. Pediatr Res 2001; 50: 34–43.
Acknowledgements
Dr Amjad Tarawneh was supported by a scholarship from the Jordanian Government. Dr Marcela Bottino and Jennifer Kaczmarek were supported by scholarships from McMaster Children's Hospital, Hamilton, Ontario, Canada and Dr GM Sant' Anna received start-up funds from the McMaster Children's Hospital, Hamilton, Ontario, Canada for this research.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflicts of interests.
Rights and permissions
About this article
Cite this article
Tarawneh, A., Kaczmarek, J., Bottino, M. et al. Severe airway obstruction during surfactant administration using a standardized protocol: a prospective, observational study. J Perinatol 32, 270–275 (2012). https://doi.org/10.1038/jp.2011.89
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/jp.2011.89
Keywords
This article is cited by
-
Do Pain and Physiological Stress Occur During MIST?
Indian Pediatrics (2023)
-
Care of the critically ill neonate with hypoxemic respiratory failure and acute pulmonary hypertension: framework for practice based on consensus opinion of neonatal hemodynamics working group
Journal of Perinatology (2022)
-
Gas in scattering media absorption spectroscopy as a potential tool in neonatal respiratory care
Pediatric Research (2022)
-
“Optimal surfactant delivery protocol using the bovine lipid extract surfactant: a quality improvement study”
Journal of Perinatology (2021)
-
Volumetric capnography pre- and post-surfactant during initial resuscitation of premature infants
Pediatric Research (2021)