Abstract
Objective:
To determine whether race/ethnicity and sex independently increase risk of respiratory distress syndrome (RDS) in late preterm and term infants.
Study Design:
Using a cohort design, we studied the risk of RDS associated with race/ethnicity and sex in infants with gestational age (GA) 34 to 42 weeks born between 1 January 2000 and 31 December 2009 (n=286 454) within 12 hospitals in the Northern California Kaiser Permanente Medical Care Program.
Result:
Male sex (adjusted odds ratio (aOR) 1.68; 95% confidence interval 1.45 to 1.93) and White race/ethnicity (vs Asians (aOR 0.57; 95% confidence interval 0.47 to 0.70), Blacks (aOR 0.66; 95% confidence interval 0.50 to 0.87), and Hispanics (aOR 0.76; 95% confidence interval 0.64 to 0.90)) independently increase risk for RDS regardless of GA. A GA <39 weeks, operative delivery, maternal diabetes, and chorioamnionitis also increased RDS risk in this cohort.
Conclusion:
Male sex and White race/ethnicity independently increase risk for RDS in late preterm and term infants. Timing of elective delivery should acknowledge these risks.
Similar content being viewed by others
Introduction
Risk of neonatal respiratory distress syndrome (RDS) is inversely associated with gestational age (GA) owing to developmental delay in production of the pulmonary surfactant. In extremely premature (birth weight <1000 g, <27 weeks GA) and very premature (birth weight <1500 g, <32 weeks GA) infants, male sex, maternal diabetes, White race, chorioamnionitis and operative delivery also increase risk and severity of RDS.1, 2, 3, 4, 5, 6 However, the associations of sex and race/ethnicity with RDS risk have not been characterized in late preterm and term infants. When compared with term infants, late preterm infants (340/7 weeks and 366/7 weeks GA) have higher rates of RDS and other non-respiratory morbidities that often require technological and pharmacological interventions and prolonged hospital stays.7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17 Although preterm and late preterm birth rates have declined in the last 5 years, a substantial fraction of infants (12.3% in the United States) is still delivered before 37 weeks gestation, most of whom (70%) are late preterm infants.8, 18 Term infants have increased risk of RDS through 39 weeks of gestation.2 To determine whether race/ethnicity and sex independently increase risk of RDS in late preterm and term infants, we used a standardized diagnosis of RDS in a multihospital-based cohort of racially and ethnically diverse, late preterm and term infants from the Northern California Kaiser Permanente Medical Care Program (NC-KPMCP). We found that male sex and White race/ethnicity independently increase risk for RDS in late preterm and term infants.
Methods
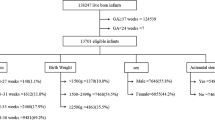
Using a clinical information system employed at all NC-KPMCP facilities that links maternal and neonatal records and clinical records to multiple information systems (for example, laboratory and hospitalization data),19, 20, 21, 22 we identified all live-born infants delivered at 12 NC-KPMCP facilities between 1 January 2000, and 31 December 2009 with a GA of 34 to 42 weeks (n=290 355). We excluded patients with unknown sex determination (n=14), congenital heart disease (n=764) and other major congenital defects (n=1574). To avoid confusion with other conditions that may present with respiratory distress at birth, we excluded infants with a diagnosis of meconium aspiration syndrome (n=1950) or culture-positive sepsis in the 1st four days of life (n=195). Some infants met multiple exclusion criteria. The final analysis cohort consisted of 286 454 infants.
Case definition was based on Vermont Oxford Network criteria for RDS (impairment in oxygenation and a chest radiograph consistent with RDS), or surfactant administration.23 GA was entered as completed weeks of gestation from the obstetrical medical record and was based on last menstrual period, and was confirmed or modified by fetal ultrasound. We classified infants as small for GA, appropriate for GA or large for GA based on birth weight and GA in comparison with the 10th and 90th percentiles of Fenton growth curves.24 We determined mode of delivery (vaginal versus cesarean section) from maternal The International Classification of Diseases-9th revision procedure codes and the infant's medical record. Data regarding indications for cesarean section delivery was not available to be collected for analysis. We assigned multiplicity of gestation (singleton versus higher-order) based on infants’ sharing the same maternal medical record number and maternal admission date to labor and delivery. Race/ethnicity of the infant was based on maternal self-report. We created a combined race/ethnicity designation by first classifying subjects’ ethnicity as Hispanic or non-Hispanic. We further subdivided non-Hispanic subjects according to race into White, Black, Asian and American Indian/Eskimo. Parents could designate race as other if they did not identify with any of these discrete designations. If race/ethnicity was not recorded, the infant's race/ethnicity was assigned as unknown. We assigned maternal diagnoses of diabetes (including gestational) (250 to 250.9; 648.0 to 638.04; 775), pre-eclampsia/eclampsia (642.4 to 642.63), chorioamnionitis (658.4 to 658.43; 762.7), and prolonged rupture of membranes (658.2 to 658.23) based on The International Classification of Diseases-9th revision diagnosis codes. We extracted the use of surfactant and mechanical ventilation from each infant's medical record.
We compared demographic characteristics of infants with and without RDS using χ2-tests and the Mann-Whitney test, when appropriate. We created a multivariate logistic regression model with RDS as the dependent variable, and sex, race/ethnicity, GA, size for GA, mode of delivery, multiplicity of gestation, pre-eclampsia/eclampsia, maternal diabetes, chorioamnionitis and prolonged rupture of membranes as predictor variables. We chose variables on the basis of prior studies of risk factors for RDS in lower GA populations.25, 26, 27, 28 Data from a subgroup of infants admitted to a level III Neonatal Intensive Care Unit (6.43% of the cohort) included antenatal glucocorticoid administration and permitted analysis of differential administration by race/ethnicity and sex. We calculated odds ratios for each predictor variable along with 95% confidence intervals and tested hypotheses using a two-sided α=0.05. To explore effects of interactions of race or GA with sex on the risk for RDS, we calculated race- and GA-specific odds ratios, adjusting for size for GA, mode of delivery, multiplicity of gestation pre-eclampsia/eclampsia and maternal diabetes. We conducted all analyses using STATA v.9.2 (StataCorp LP, College Station, TX, USA). The NC-KPMCP Institutional Review Board approved this project.
Results and discussion
The overall incidence of RDS in this cohort was 0.31% (n=895), with incidence in late preterm infants of 3.22% and in term infants of 0.12%. Infants with RDS were more likely to be male, White, of lower GA and delivered by cesarean section. Our data analysis showed RDS infants to be either SGA or LGA. Among infants with RDS, 44.8% required mechanical ventilation and 38.9% received surfactant (Table 1).
To characterize the relationships among GA, sex, and race/ethnicity and the absolute risk of RDS, we plotted the crude incidence of RDS by GA for each race and sex category (Figure 1). At 34 weeks of GA, White males had the highest risk of RDS (13.0%), whereas Hispanic females had the lowest risk of RDS (4.3%). In each race/ethnicity and sex category, risk for RDS decreased with increasing GA until 37 weeks of gestation. Regardless of race/ethnicity, females had a lower incidence of RDS than males. In Whites, the absolute risk difference between males and females persisted until approximately 38 weeks of GA. However, the difference decreased with advancing GA. In the other race/ethnicity categories, we observed no consistent differences between males and females after 34 weeks GA, except for an unexplained large difference between Asian males and females at 35 weeks (0.7% in females and 3.5% in males).
In the multivariate logistic regression model, GA was the strongest predictor of RDS (Table 2). Compared with infants born at 40 weeks GA, those born at 34 weeks GA had an adjusted odds ratio (aOR) for RDS of 114.12 (95% confidence interval 84.71 to 153.74). Even infants born at 38 weeks gestation had a greater risk of RDS (aOR 1.67; 95% confidence interval 1.16 to 2.41). After adjusting for other covariables, males still had a greater risk of RDS than females (aOR 1.68; 95% confidence interval 1.45 to 1.93). Consistent with previously reported racial/ethnic risk differences for RDS among infants born at <32 weeks gestation, we found Blacks had a lower risk of RDS than Whites.29, 30, 31, 32 As our cohort had a large proportion of Hispanics (25%) and Asians (20%), we were able to evaluate their risk of RDS compared with non-Hispanic Whites, and found both Asians and Hispanics to be at lower risk for RDS (Table 2). Additional significant independent predictors of RDS in the multivariate regression were Caesarean delivery, maternal diabetes and chorioamnionitis. Multiple gestation, maternal pre-eclampsia and prolonged rupture of membranes were significant protective factors.
We also developed GA- and race/ethnicity-specific regression models (Figure 2). For GAs <39 weeks, males had a higher risk of RDS compared with females (point estimates for the aOR of 1.2 to 2.2). After 39 weeks, the point estimates still showed males at increased risk but were not statistically significant. For race/ethnicity, males were at a statistically increased risk for RDS compared with females in Whites, Asians and Hispanics.
We evaluated a potentially confounding association between maternal antenatal corticosteroid administration and either race/ethnicity or sex by comparing a subgroup of male and female infants admitted to a level III neonatal intensive care unit for whom data on maternal antenatal corticosteroid administration were available (n=1015). In this subgroup, 5.35% of males and 5.72% of females were born to mothers who received antenatal corticosteroids (χ2 P value=0.28). In addition, using pairwise comparisons, we found that the only significant difference occurred between Asians (4.62%) and Blacks (6.25%) (χ2 P value=0.009) who had similar rates of RDS. Differential administration of antenatal corticosteroids by sex or race/ethnicity does not appear to have confounded the overall analysis.
Our findings confirm and extend a recent, large population-based European cohort study of RDS risk factors,2 and are consistent with prior studies that found increased risk of RDS by week through 39 weeks of gestation.16, 33, 34, 35, 36 These findings illustrate the importance of each week of gestation for RDS risk reduction through 39 weeks gestation. We also found that the increased risk of RDS associated with male sex and White race/ethnicity observed in preterm infants persists in late preterm and term infants. Differences in hormonal regulation of lung development provide candidate mechanisms to account for an increased risk of RDS associated with male sex.37, 38, 39, 40, 41, 42 The racial disparity in risk of RDS between Whites and Blacks previously observed in infants <32 weeks GA also persists in late preterm and term infants.29, 30 The composition of our cohort permitted demonstration of increased RDS risk in White infants versus both Asian and Hispanic infants. The mechanisms that account for these differences require further investigation and replication in other cohorts. These findings contrast with those of studies that have not shown lower rates of RDS among Asian infants, and may reflect admixture in the California Asian population.9 Hispanics in Northern California are also an ethnically heterogeneous group associated with admixture with European, African, and Indigenous American populations.43 On the basis of the low frequency of Medi-Cal insurance in our cohort (<5% compared with 47% in the general population in California),44 race/ethnicity likely had a minimal influence on access to care.
Although the size of our cohort provides adequate statistical power to observe the difference in RDS risk in male versus female infants and between infants of different races/ethnicities, our study has several limitations. First, although maternal and neonatal data were collected prospectively, our analysis was retrospective and therefore, we did not have complete data for the entire cohort on antenatal steroids. However, subgroup analysis suggested that differential antenatal corticosteroid administration did not account for sex and race/ethnicity differences in RDS risk. Second, our case definition may include infants with transient tachypnea of the newborn. To reduce misclassification, we repeated our multivariate analysis using a more restrictive definition of RDS (only infants who received surfactant). Males remained at a greater risk of RDS than females (aOR 1.75; 95% confidence interval 1.39 to 2.20). Asians were still at a lower risk of RDS than Whites (aOR 0.54; 95% confidence interval 0.39 to 0.74). Although not statistically significant, there was trend for Hispanics (aOR 0.83; 95% confidence interval 0.63 to 1.08) and Blacks (aOR 0.70; 95% confidence interval 0.46 to 1.06) to have a decreased risk of RDS compared with Whites. The reduced number of cases with the more restrictive definition of RDS diminished our ability to find statistical differences. Third, although it is unlikely that changes in frequency of use of inhaled nitric oxide that occurred during the 9-year interval of cohort accumulation impacted RDS risk,45 other unrecognized practice changes may have confounded our analysis. Despite these limitations, our study suggests that decisions concerning timing of delivery should acknowledge RDS risk associated with male sex and White race/ethnicity in late preterm and term infants. For example, because cesarean delivery on maternal request prompts 4 to 18% of the ∼1 000 000 cesarean sections annually in the United States,46, 47 counseling for women to avoid elective delivery before 39 completed weeks of gestation should also include acknowledgement of RDS risk associated with male sex and White race/ethnicity.48
In conclusion, our study shows that male sex and White race/ethnicity are independent risk factors for RDS in late preterm and term infants. Future studies to elucidate mechanisms that account for this differential risk38, 39, 49 will inform obstetrical counseling and decision-making.
References
Dani C, Reali MF, Bertini G, Wiechmann L, Spagnolo A, Tangucci M et al. Risk factors for the development of respiratory distress syndrome and transient tachypnoea in newborn infants. Italian Group of Neonatal Pneumology. Eur Respir J: Official J Eur Soc Clin Respir Physiol 1999; 14: 155–159.
Gouyon JB, Vintejoux A, Sagot P, Burguet A, Quantin C, Ferdynus C . Neonatal outcome associated with singleton birth at 34-41 weeks of gestation. Int J Epidemiol 2010; 39: 769–776.
Ingemarsson I . Gender aspects of preterm birth. Bjog 2003; 110 (Suppl 20): 34–38.
Luerti M, Parazzini F, Agarossi A, Bianchi C, Rocchetti M, Bevilacqua G . Risk factors for respiratory distress syndrome in the newborn: a multicenter Italian survey. Acta Obstetricia et Gynecologica Scandinavica 1993; 72: 359–364.
Papageorgiou AN, Colle E, Farri-Kostopoulos E, Gelfand MM . Incidence of respiratory distress syndrome following antenatal betamethasone: role of sex, type of delivery, and prolonged rupture of membranes. Pediatrics 1981; 67: 614–617.
Perelman R, Palta M, Kirby R, Farrell PM . Discordance between male and female deaths due to the respiratory distress syndrome. Pediatrics 1986; 78: 238–244.
Jain L, Dudell GG . Respiratory transition in infants delivered by cesarean section. Semin Perinatol 2006; 30: 296–304.
Khashu M, Narayanan M, Bhargava S, Osiovich H . Perinatal outcomes associated with preterm birth at 33 to 36 weeks’ gestation: a population-based cohort study. Pediatrics 2009; 123: 109–113.
Ma X, Huang C, Lou S, Lv Q, Su W, Tan J et al. The clinical outcomes of late preterm infants: a multi-center survey of Zhejiang, China. J Perinat Med 2009; 37: 695–699.
McIntire DD, Leveno KJ . Neonatal mortality and morbidity rates in late preterm births compared with births at term. Obstet Gynecol 2008; 111: 35–41.
Melamed N, Klinger G, Tenenbaum-Gavish K, Herscovici T, Linder N, Hod M et al. Short-term neonatal outcome in low-risk, spontaneous, singleton, late preterm deliveries. Obstet Gynecol 2009; 114: 253–260.
Ramachandrappa A, Jain L . Health issues of the late preterm infant. Pediatr Clin North Am 2009; 56: 565–577, Table of Contents.
Roth-Kleiner M, Wagner BP, Bachmann D, Pfenninger J . Respiratory distress syndrome in near-term babies after caesarean section. Swiss Med Wkly 2003; 133: 283–288.
Vachharajani AJ, Dawson JG . Short-term outcomes of late preterms: an institutional experience. Clin Pediatr (Phila) 2009; 48: 383–388.
Ventolini G, Neiger R, Mathews L, Adragna N, Belcastro M . Incidence of respiratory disorders in neonates born between 34 and 36 weeks of gestation following exposure to antenatal corticosteroids between 24 and 34 weeks of gestation. Am J Perinatol 2008; 25: 79–83.
Wang ML, Dorer DJ, Fleming MP, Catlin EA . Clinical outcomes of near-term infants. Pediatrics 2004; 114: 372–376.
Yoder BA, Gordon MC, Barth Jr WH . Late-preterm birth: does the changing obstetric paradigm alter the epidemiology of respiratory complications? Obstet Gynecol 2008; 111: 814–822.
Mathews TJ, Minino AM, Osterman MJ, Strobino DM, Guyer B . Annual summary of vital statistics: 2008. Pediatrics 2011; 127: 146–157.
Escobar GJ, Fischer A, Kremers R, Usatin MS, Macedo AM, Gardner MN . Rapid retrieval of neonatal outcomes data: the Kaiser Permanente Neonatal Minimum Data Set. Qual Manag Health Care 1997; 5: 19–33.
Escobar GJ, Greene JD, Hulac P, Kincannon E, Bischoff K, Gardner MN et al. Rehospitalisation after birth hospitalisation: patterns among infants of all gestations. Arch Dis Child 2005; 90: 125–131.
Escobar GJ, Li DK, Armstrong MA, Gardner MN, Folck BF, Verdi JE et al. Neonatal sepsis workups in infants >/=2000 grams at birth: a population-based study. Pediatrics 2000; 106: 256–263.
Newman TB, Liljestrand P, Jeremy RJ, Ferriero DM, Wu YW, Hudes ES et al. Outcomes among newborns with total serum bilirubin levels of 25 mg per deciliter or more. N Engl J Med 2006; 354: 1889–1900.
Vermont Oxford Network. Vermont Oxford Network Database, Manual of Operations - Release 14.0. Burlington, 2009. http://www.vtoxford.org/tools/2010ManualofOperationswithindex14.pdf.
Fenton TR . A new growth chart for preterm babies: Babson and Benda's chart updated with recent data and a new format. BMC Pediatr 2003; 3: 13.
Lee J, Oh KJ, Park CW, Park JS, Jun JK, Yoon BH . The presence of funisitis is associated with a decreased risk for the development of neonatal respiratory distress syndrome. Placenta 2011; 32: 235–240.
Sato M, Nishimaki S, Yokota S, Seki K, Horiguchi H, An H et al. Severity of chorioamnionitis and neonatal outcome. J Obstet Gynaecol Res 2011; 37 (10): 1313–1319.
Le Ray C, Boithias C, Castaigne-Meary V, l’Helias LF, Vial M, Frydman R . Caesarean before labour between 34 and 37 weeks: what are the risk factors of severe neonatal respiratory distress? Eur J Obstet Gynecol Reprod Biol 2006; 127: 56–60.
Sims EJ, Vermillion ST, Soper DE . Preterm premature rupture of the membranes is associated with a reduction in neonatal respiratory distress syndrome. Am J Obstet Gynecol 2002; 187: 268–272.
Balchin I, Whittaker JC, Lamont RF, Steer PJ . Timing of planned cesarean delivery by racial group. Obstet Gynecol 2008; 111: 659–666.
Farrell PM, Wood RE . Epidemiology of hyaline membrane disease in the United States: analysis of national mortality statistics. Pediatrics 1976; 58: 167–176.
Kavvadia V, Greenough A, Dimitriou G, Hooper R . Influence of ethnic origin on respiratory distress syndrome in very premature infants. Arch Dis Child Fetal Neonatal Ed 1998; 78: F25–F28.
Richardson DK, Torday JS . Racial differences in predictive value of the lecithin/sphingomyelin ratio. Am J Obstet Gynecol 1994; 170: 1273–1278.
Raju TN, Higgins RD, Stark AR, Leveno KJ . Optimizing care and outcome for late-preterm (near-term) infants: a summary of the workshop sponsored by the National Institute of Child Health and Human Development. Pediatrics 2006; 118: 1207–1214.
Tita AT, Landon MB, Spong CY, Lai Y, Leveno KJ, Varner MW et al. Timing of elective repeat cesarean delivery at term and neonatal outcomes. N Engl J Med 2009; 360: 111–120.
Balchin I, Steer PJ . Race, prematurity and immaturity. Early Hum Dev 2007; 83: 749–754.
Reddy UM, Bettegowda VR, Dias T, Yamada-Kushnir T, Ko CW, Willinger M . Term pregnancy: a period of heterogeneous risk for infant mortality. Obstet Gynecol 2011; 117: 1279–1287.
Beyer C, Kuppers E, Karolczak M, Trotter A . Ontogenetic expression of estrogen and progesterone receptors in the mouse lung. Biol Neonate 2003; 84: 59–63.
Carey MA, Card JW, Voltz JW, Arbes Jr SJ, Germolec DR, Korach KS et al. It's all about sex: gender, lung development and lung disease. Trends Endocrinol Metab 2007; 18: 308–313.
Carey MA, Card JW, Voltz JW, Germolec DR, Korach KS, Zeldin DC . The impact of sex and sex hormones on lung physiology and disease: lessons from animal studies. Am J Physiol Lung Cell Mol Physiol 2007; 293: L272–L278.
Seaborn T, Simard M, Provost PR, Piedboeuf B, Tremblay Y . Sex hormone metabolism in lung development and maturation. Trends Endocrinol Metab 2010; 21: 729–738.
Trotter A, Ebsen M, Kiossis E, Meggle S, Kueppers E, Beyer C et al. Prenatal estrogen and progesterone deprivation impairs alveolar formation and fluid clearance in newborn piglets. Pediatr Res 2006; 60: 60–64.
Trotter A, Kipp M, Schrader RM, Beyer C . Combined application of 17beta-estradiol and progesterone enhance vascular endothelial growth factor and surfactant protein expression in cultured embryonic lung cells of mice. Int J Pediatr 2009; 2009: 170491.
Halder I, Yang BZ, Kranzler HR, Stein MB, Shriver MD, Gelernter J . Measurement of admixture proportions and description of admixture structure in different US populations. Hum Mut 2009; 30: 1299–1309.
California Department of Health Services. Birth Public Use File 2008.
Clark RH, Ursprung RL, Walker MW, Ellsbury DL, Spitzer AR . The changing pattern of inhaled nitric oxide use in the neonatal intensive care unit. J Perinatol 2010; 30: 800–804.
National Institutes of Health state-of-the-science conference statement: cesarean delivery on maternal request March 27-29, 2006. Obstet Gynecol 2006; 107: 1386–1397.
Lee YM, D’Alton ME . Cesarean delivery on maternal request: the impact on mother and newborn. Clin Perinatol 2008; 35: 505–518, x.
American College of Obstetricians and Gynecologists. ACOG Committee Opinion No. 394, December 2007. Cesarean delivery on maternal request. Obstet Gynecol 2007; 110: 1501.
Provost PR, Boucher E, Tremblay Y . Apolipoprotein A-I, A-II, C-II, and H expression in the developing lung and sex difference in surfactant lipids. J Endocrinol 2009; 200: 321–330.
Acknowledgements
This study was supported by awards from the National Institutes of Health (R01 HL065174 (FSC, AH), R01 HL082747 (FSC, AH), and K23 HD055963 (MWK)), the Saigh Foundation (FSC, AH) and the Doris Duke Charitable Foundation (BPC).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
This work is licensed under the Creative Commons Attribution-NonCommercial-No Derivative Works 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-nd/3.0/
About this article
Cite this article
Anadkat, J., Kuzniewicz, M., Chaudhari, B. et al. Increased risk for respiratory distress among white, male, late preterm and term infants. J Perinatol 32, 780–785 (2012). https://doi.org/10.1038/jp.2011.191
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/jp.2011.191
Keywords
This article is cited by
-
Predictors of successful extubation from volume-targeted ventilation in extremely preterm neonates
Journal of Perinatology (2024)
-
Fetal lung growth predicts the risk for early-life respiratory infections and childhood asthma
World Journal of Pediatrics (2024)
-
Global, Regional and National Trends in the Burden of Neonatal Respiratory Failure and essentials of its diagnosis and management from 1992 to 2022: a scoping review
European Journal of Pediatrics (2023)
-
Efficacy and safety of metformin compared to insulin in gestational diabetes: a systematic review and meta-analysis of Chinese randomized controlled trials
International Journal of Clinical Pharmacy (2022)
-
Race as a moderator of the association between ethnicity, preeclampsia and neonatal respiratory distress syndrome
World Journal of Pediatrics (2022)