Abstract
Objective:
Serum amyloid A (SAA) is an acute phase inflammatory marker that is closely associated with ischemic injuries. Its expression in neonatal hypoxic ischemic encephalopathy (HIE) has not been studied. To test the hypothesis that SAA is increased in neonatal HIE and its concentration correlates with the severity of encephalopathy.
Study Design:
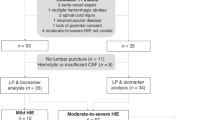
We conducted a prospective case-control study on 54 full-term neonates; 27 cases with evidence of perinatal compromise and 27 healthy controls. Blood samples were collected from cases and controls at postnatal day 1 and day 7, and SAA was measured by ELISA.
Results:
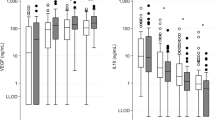
SAA concentrations (μg ml−1) were significantly increased in cases when compared with controls at day 1 and at day 7 (P<0.001). SAA concentrations at day 1 were greater in cases who died when compared with those who survived, and correlated significantly with the severity of HIE (146.9±56.4 vs 79.8±24.7 vs 58.1±21.5) in severe, moderate and mild HIE, respectively (P=0.001).
Conclusions:
The expression of SAA is increased in response to hypoxia ischemia of the neonate. The increased concentration correlates with the severity of encephalopathy and is associated with mortality. This is the first study of SAA in neonatal HIE.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Taniguchi H, Andreasson K . The hypoxic ischemic encephalopathy model of perinatal ischemia. J Vis Exp 2008; 19: 3791–3955.
Volpe J . Neurology of the Newborn. 5th edn. Saunders: Philadelphia, PA, 2008; 245–372.
Buonocore G, Zani S, Perrone S, Caciotti B, Bracci R . Intraerythrocyte nonprotein-bound iron and plasma malondialdehyde in the hypoxic newborn. Free Radic Biol Med 1998; 25: 770–776.
Belet N, Belet U, Incesu L, Uysal S, Ozinal S, Keskin T et al. Hypoxic-ischemic encephalopathy: correlation of serial MRI and outcome. Pediatr Neurol 2004; 31: 267–274.
Cunnane G, Whitehead AS . Amyloid precursors and amyloidosis in rheumatoid arthritis. Baillieres Best Pract Res Clin Rheumatol 1999; 13: 615–628.
Mozes G, Friedman N, Shainkin-Kestenbaum R . Serum amyloid A: an extremely sensitive marker for the intensity of tissue damage in trauma patients and indicator of acute response in various diseases. J Trauma 1989; 29: 71–74.
Lannergård A, Larsson A, Friman G, Ewald U . Human serum amyloid A (SAA) and high sensitive C-reactive protein (hsCRP) in preterm newborn infants with nosocomial infections. Acta Paediatr 2008; 97: 1061–1065.
Cunnane G . Effects of anti tumour necrosis factor alpha on synovium in patients with spondylarthropathy: comment on the article by Baeten et al. Arthritis Rheum 2001; 44: 2937–2939.
Song C, Shen Y, Yamen E, Hsu K, Yan W, Witting PK et al. Serum amyloid A may potentiate prothrombotic and proinflammatory events in acute coronary syndromes. Atherosclerosis 2009; 202: 596–604.
Kosuge M, Ebina T, Ishikawa T, Hibi K, Tsukahara K, Okuda J et al. Serum amyloid A is a better predictor of clinical outcomes than C-reactive protein in non-ST-segment elevation acute coronary syndromes. Circ J 2007; 71: 186–190.
Hua S, Song C, Geczy CL, Freedman SB, Witting PK . A role of acute phase serum amyloid A and high density lipoprotein in oxidative stress, endothelial dysfunction and atherosclerosis. Redox Rep 2009; 14: 187–196.
Aly H, El Beshlawi A, Badrawi N, Mohsen L, Mansour E . Thrombopoeitin level is increased in the serum of asphyxiated neonates. J Perinatol 2005; 25: 320–324.
Sarnat HB, Sarnat MS . Neonatal encephalopathy following fetal distress. Arch Neurol 1976; 33: 696–705.
Bayramli G, Ulutas B . Acute phase protein response in dogs with experimentally induced gastric mucosal injury. Vet Clin Pathol 2008; 37: 312–316.
Verklan MT . The chilling details: hypoxic ischemic encephalopathy. J Perinat Neonatal Nurs 2009; 23: 56–68.
Lomberg SR, Nielson LR, Heegaard PM . Acute phase proteins in cattle after exposure to complex stress. Vet Res Commun 2008; 32: 578–582.
Jabor A, Holub Z, Franekova J . Serum amyloid A as an effective marker for the assessment of surgical trauma and risk of post operative complications. Ceska Gynekol 2006; 71: 131–136.
Dani C, Cecchi A, Bertini G . Role of oxidative stress as physiopathologic factor in the preterm infant. Minerva Pediatr 2004; 56: 381–394.
Alsemgeest SP, Jonker FH, Taverne MA, Kalsbeek HC . Serum amyloid A (SAA) and haptoglobin (Hp) plasma concentrations in newborn calves. Theriogenology 1995; 43: 381–387.
Kristensen K, Wide-Swensson D, Lindstrom V, Schmidt C, Grubb A, Stevens H . Serum amyloid a protein and C- reactive protein in normal pregnancy and preeclampsia. Gynecol Obstet Invest 2009; 67: 275–280.
Scher M . Perinatal asphyxia: timing and mechanism of injury in neonatal encephalopathy. Cur Neurol Neurosci 2001; 1: 175–184.
Bekedam DJ, Engelsbel S, Mol BW, Buitendijk SE, Vander Pal- de Bruin KM . Male predominance in fetal distress during labor. J Obstet Gynecol 2002; 187: 1605–1607.
Lieberman E, Lang JM, Cohen AP, Frigoletto Jr FD, Acker D, Ras R . The association of fetal sex with the rate of cesarean section. Am J Obstet Gynecol 1997; 176: 667–671.
Dawes NW, Dawes GS, Moulden M, Redman GW . Fetal heart rate patterns in term labor vary with sex, gestational age, epidural analgesia and fetal weight. Am J Obstet Gynecol 1999; 180: 181–187.
Edwards A, Megens A, Peek M, Wallace EM . Sexual origins of placental dysfunction. Lancet 2000; 355: 203–204.
Clarke C, Mittwoch U . Changes in the male to female ratio at different stages of life. Br J Obstet Gynaecol 1995; 102: 677–679.
Thorand B, Baumert J, Doring A, Herder C, Kolb H . Sex differences in the relation of body composition to inflammatory markers. Atherosclerosis 2006; 184: 216–224.
Maury CP . Comparative study of serum amyloid A protein and C Reactive protein in disease. Clin Sci (Lond) 1985; 68: 233–238.
Acknowledgements
We thank Tahmina Ahmad at The George Washington University Hospital for editorial assistance and preparation of the figures.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Aly, H., Hamed, Z., Mohsen, L. et al. Serum amyloid A protein and hypoxic ischemic encephalopathy in the newborn. J Perinatol 31, 263–268 (2011). https://doi.org/10.1038/jp.2010.130
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/jp.2010.130
Keywords
This article is cited by
-
Serum amyloid A is increased in children with abusive head trauma: a gel-based proteomic analysis
Pediatric Research (2014)
-
Neonatal levels of acute phase proteins and later risk of non-affective psychosis
Translational Psychiatry (2013)