Summary
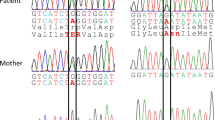
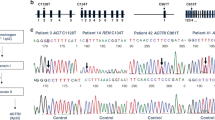
Arginase is an enzyme involved in the urea cycle and its deficiency results in argininemia, an autosomal recessive disorder accompanied by hyperammonemia. We isolated two overlapping cDNA clones and determined the entire nucleotide sequence (Haraguchiet al. 1987.Proc. Natl. Acad. Sci. USA 84: 412–415). Sequence analysis of eight cDNA clones revealed nucleotide variations at five positions in the protein coding region, and all these variations were accompanied by amino acid substitutions (Met to Lys at amino acid residue 1, Gln to Glu at residue 86, Ser to Phe at residue 109, Leu to Gln at residue 206, and Val to Ala at residue 233). These results suggest that human liver arginase is polymorphic in amino acid sequence, although some of the variations may be due to a transcription error or to cloning artifacts. Two restriction fragment length polymorphisms were identified at the arginase gene locus, using restriction endonucleasePvuII andHincII. These sequence polymorphisms and restriction fragment length polymorphisms may serve as linkage markers for arginase deficiency.
Similar content being viewed by others
Article PDF
References
Bell, G.I., Karam, J.H., and Rutter, W.I. 1981. Polymorphic DNA region adjacent to the 5′ end of the human insulin gene.Proc. Natl. Acad. Sci. USA 78: 5759–5763.
Beruter, J., Colombo, J.P., and Bachmann, C. 1978. Purification and properties of arginase from human liver and erythrocytes.Biochem. J. 175: 449–454.
Brusdeilins, M., Kuhner, R., and Schumacher, K. 1985. Purification, affinity to anti-human arginase immunoglobulin-Sepharose 4B and subunit molecular weights of mammalian arginases.Biochim. Biophys. Acta 840: 79–90.
Glass, R.D. and Knox, W.E. 1973. Arginase isozymes of rat mammary gland, liver, and other tissues.J. Biol. Chem. 248: 5785–5789.
Haraguchi, Y., Takiguchi, M., Amaya, K., Kawamoto, S., Matsuda, I., and Mori, M. 1987. Molecular cloning and nucleotide sequence of cDNA for human liver arginase.Proc. Natl. Acad. Sci. USA 84: 412–415.
Hattori, M. and Sakaki, Y. 1986. Dideoxy sequencing method using denatured plasmid templates.Anal. Biochem. 152: 232–238.
Herzfeld, A. and Raper, S.M. 1976. The heterogeneity of arginases in rat tissues.Biochem. J. 153: 469–478.
Hirsch-Kolb, H. and Greenberg, D.M. 1968. Molecular characteristics of rat liver arginase.J. Biol. Chem. 243: 6123–6129.
Kawamoto, S., Amaya, Y., Oda, T., Kuzumi, T., Saheki, T., Kimura, S., and Mori, M. 1986. Cloning and expression inEscherichia coli of cDNA for arginase of rat liver.Biochem. Biophys. Res. Commun. 136: 955–961.
Kawamoto, S., Amaya, Y., Murakami, K., Tokunaga, F., Iwanaga, S., Kobayashi, K., Saheki, T., Kimura, S., and Mori, M. 1987. Complete nucleotide sequence of cDNA and deduced amino acid sequence of rat liver arginase.J. Biol. Chem. 262: 6280–6283.
Kidd, J.R., Dizikes, G.J., Grody, W.W., Cederbaum, S.D., and Kidd, K.K. 1986. APvuII RFLP for the human liver arginase (ARG) gene.Nucleic Acids Res. 14: 2544.
Kyte, J. and Doolittle, R.F. 1982. A simple method for displaying the hydropathic character of a protein.J. Mol. Biol. 157: 105–132.
Maniatis, T., Fritsch, E.F., and Sambrook, J. 1982.Molecular Cloning, Cold Spring Harbor Laboratory, New York.
Pennickx, M., Simon, J.P., and Wiame, J.M. 1974. Interaction between arginase andl-ornithine carbamoyltransferase inSaccharomyces cerevisiae.Eur. J. Biochem. 49: 429–442.
Sanger, F., Nicklen, S., and Coulson, A.R. 1977. DNA sequencing with chain-terminating inhibitors.Proc. Natl. Acad. Sci. USA 74: 5463–5467.
Schimke, R.T. 1962. Adaptive characteristics of urea cycle enzymes in the rat.J. Biol. Chem. 237: 459–468.
Skrzypek-Osiecka, Robin, Y., and Porembska, Z. 1983. Purification of rat kidney arginase A1 and A4 and their subcellular distribution.Acta Biochim. Pol. 30: 83–92.
Sparkes, R.S., Dizikes, G.J., Klisak, I., Grody, W.W., Mohandas, T., Heinzmann, C., Zollman, S., Lusis, A.J., and Cederbaum, S.D. 1986. The gene for human liver arginase (ARG1) is assigned to chromosome band 6q23.Am. J. Hum. Genet. 39: 186–193.
Spector, E.B., Rice, S.C.H., and Cederbaum, S.D. 1983. Immunologic studies of arginase in tissues of normal human adult and arginase-deficient patients.Pediatr. Res. 17: 941–944.
Sumrada, R.A. and Cooper, T.G. 1984. Nucleotide sequence of theSaccharomyces cerevisiae arginase gene (CAR1) and its transcription under various physiological conditions.J. Bacteriol. 160: 1078–1087.
Tarrab, R., Rodriguez, J., Huitron, C., Palacios, R., and Soberon, G. 1974. Molecular forms of rat-liver arginase. Isolation and characterization.Eur. J. Biochem. 49: 457–468.
Vielle-Breitburd, F. and Orth, G. 1972. Rabbit liverl-arginase.J. Biol. Chem. 247: 1227–1235.
Walser, M. 1983. Urea cycle disorders and other hereditary hyperammonemic syndromes. InThe Metabolic Basis of Inherited Disease, Stanbury, J.B., Wyngaarden, J.B., Fredrickson, D.S., Goldstein, J.L., and Brown, M.S. eds. McGraw Hill, New York, pp. 402–438.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Haraguchi, Y., Takiguchi, M., Matsuda, I. et al. Sequence heterogeneity of human liver arginase cDNAs and restriction fragment length polymorphism of the gene locus. Jap J Human Genet 33, 305–313 (1988). https://doi.org/10.1007/BF02032860
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF02032860