Abstract
Dietary risks for the German population owing to pesticide residues in foods were assessed based on food monitoring data, consumption surveys for children and adults and compound specific toxicological reference values or general thresholds of toxicological concern. A tiered probabilistic modelling was conducted to screen 700 pesticides for significant long- and short-term dietary exposures. Especially for the short-term dietary exposure, the probabilistic methodology used allows simultaneous consideration of the complete daily consumption, whereas most regulatory bodies still rely on single commodity approaches. After screening, refined exposure assessments were conducted for 19 compounds under consideration of conversion factors for toxicologically relevant metabolites, processing information, experimentally derived variability factors and the edible portion for each food item. In total, for 693 compounds the dietary exposure was unlikely to present a chronic or acute public health concern for the German population. In contrast, the refined assessments indicate that the short-term dietary exposure for chlorpyrifos and the cumulative short-term dietary exposure for dimethoate and omethoate may present a public health concern. For copper, owing to exposure assessment limitations, as well as for dimethylvinphos, halfenprox and tricyclazole, which exceeded the thresholds of toxicological concern, the dietary risk assessment remained inconclusive.
Similar content being viewed by others
Introduction
For pesticides, national and international monitoring programmes are in place to measure their occurrence in foods. These programmes are primarily focused on finding exceedances of established maximum residue levels (MRLs) but also on providing representative data to assess health risks for consumers.
With the German food monitoring, a concept was started in 2009 based on a representative food market basket covering at least 80% of the average daily consumption for German children over a 6-year period.1, 2 With the data generated, probabilistic modelling of the long- and short-term dietary exposure was conducted to assess chronic and acute risks for the German population and to identify possible areas of concern. Especially the probabilistic short-term dietary exposure estimation considering the complete daily consumption goes beyond the deterministic single commodity based International Estimated Short-Term Intake (IESTI) methodology (ref. 3: p.15, ref. 4: p.29, ref. 5: p.127) used on European level for MRL setting and the authorisation of plant protection products.6
This work represents the first pesticide dietary risk assessment for German consumers using population based models and German residue monitoring data. With the new focus of German residue monitoring in 2009, shifting from identifying MRL exceedances toward better suitability for consumer dietary risk assessments, more comprehensive conclusions on possible health concerns become possible.
Materials and methods
Probabilistic Methodology
The probabilistic modelling was performed with the Monte-Carlo Risk Assessment software (MCRA) Version 8.1, which was developed by the Dutch National Institute for Public Health and the Environment (RIVM).7 MCRA was designed primarily for the field of pesticide residues and addresses the “Guidance on the use of probabilistic modelling for pesticides” issued by the European Food Safety Authority (EFSA).8
According to this guidance (p.8), a “pessimistic” and an “optimistic” run shall be calculated to estimate the upper (1st tier) and lower (2nd tier) bound of the exposure, respectively. For 1st tier runs residues below the limit of quantification (LOQ) were substituted by the LOQ value, whereas 2nd tier runs consider all residues below the LOQ value to be present at ”zero”. Each run was generally based on 1,000,000 iterations, which means that one random individual was selected and each consumed food reported was correlated with an also randomly selected residue concentration (empiric sampling) per iteration. The sum of residues via all foods represents the total exposure for the selected individual.
The 1st and 2nd tier runs were used for a screening to identify substances with significant long- and short-term exposure levels. In general, the 99.9th percentile (P99.9) of the exposure distribution was used as basis for further considerations.9 In addition, the 90th, 95th, 99th and 99.99th percentile were reported to show the sharpness of the exposure distribution, to allow comparison between 1st and 2nd tier runs to consider the sensitivity of LOQs and detection frequencies and to identify possible exceedances of toxicololgical reference values at the very upper end of the exposure distribution.
When the highly conservative 1st tier run indicated a dietary exposure higher than 10% of the Acceptable Daily Intake value (ADI) or of the Acute Reference Dose (ARfD) at the P99.9, a 2nd tier run was calculated. The 10% criterion was selected to compensate for the lack of information on potentially relevant metabolites not covered by the residue definition for enforcement purposes. In current guidance documents more than 10% contribution of a metabolite to the total residue is considered as a major factor for its inclusion into a residue definition (ref. 5:Table 3. Ref.1, 10: Table 1).
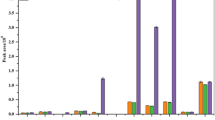
The 2nd tier run provides a more realistic estimate of the exposure. Although residues may have occasionally been present in foods at concentrations below the LOQ, which would cause an underestimation of the exposure when substituted by zero, the use of consumption data expressed as raw agricultural commodity (RAC) still provides an overall conservative exposure assessment since inedible parts are still included in the assumed portion sizes. Again, the 10% ADI/ARfD-threshold was used as screening criterion. In case of compounds with a very high rate of detected residues (e.g., chlorpyrifos or chlorpropham), the outcome of the 2nd tier run might be almost identical or even slightly higher than the 1st tier run. Upper percentiles of the exposure distribution are dominated by measured concentrations instead of LOQ values, making the impact of substituting the LOQ with “zero” insignificant. Especially for the short-term exposure, the variability between runs may be large and both the 1st and 2nd tier runs still represent a single point estimate of percentiles and depend on the random selection of data. Modelling uncertainty to describe this variability was only taken into account for in the refined assessments; however the range of expected variability is also covered by the 10% ADI/ARfD-threshold in the 1st and 2nd tier runs. For substances which exceeded the 10% ADI/ARfD-threshold in 2nd tier runs, additional information on conversion factors (CFs), food processing factors, experimentally derived variability factors and the percentage of the edible portion was used for a refined assessment (Table 1). The modelling uncertainty of the probabilistic runs was quantified by 100 additional resampling cycles of 10000 iterations each, estimating the 2.5th and 97.5th interval as lower and upper modelling range, respectively. For the interpretation, a large range around the results reflects high variability in the data.
The probabilistic modelling of long-term dietary exposures required adjustment of the consumption data collected for single days to address differing consumption frequencies in the long-term to avoid significant underestimation of the intake. The adjustment was primarily done parametically by using the MCRA built-in LogisticNormal-Normal (LNN) model suggested for a realistic “right-tail assessment”11 (p.20). This procedure requires a reasonable number of consumers for an acceptable correlation of data, which is not always reached especially for infrequent eaten foods. When correlations were not significant, the observed individual mean was used instead. Although all food surveys used were designed to generate representative consumption data for all seasons, the long-term consumption of some rarely eaten foods may be underestimated. However, owing to very low frequency of consumption, the average contribution to the overall long-term exposure is considered small.
In the probabilistic modelling of short-term exposures unit-to-unit variability was taken into account by applying a beta-distribution model based on default variability factors of 5 or 7 used in the European Union for commodities with unit-weights ≥25 g (ref. 6: Table:Acute_overview_children). These default factors are the highest currently used in regulatory systems. Within Codex Alimentarius, a general variability factor of 3 is used since 2003 (ref. 12: p.11). Experimentally derived variability factors, when available, were only used in refined assessments. For commodities with <25 g unit weight or for mixed/blended commodities (e.g., juices or cereal grains) a variability factor of 1 was considered.
Consumption Data
For the German population three different consumption surveys were used for the probabilistic assessment covering different sub-populations. In the VELS-study13 from 2003 the daily consumption of children aged 6 months up to 4 years (n=816) was surveyed using a 3-day weighed/estimated food record repeated after 3–6 months (4–8 weeks for children aged <1 year). Older children aged 6–17 years were part of the EsKiMo-study14, 15 conducted by the Robert Koch-Institute and the University of Paderborn in 2006.16 In EsKiMo, the daily consumption for the sub-group of 6–11 years was recorded using a 3-day food protocol on 1234 individuals. For older children, a 4-week food frequency questionnaire was used in the EsKiMo-study, unsuitable for short-term exposure assessments on a daily basis. The third survey available was the German Nutrition Survey II (NVS II), conducted by the Max Rubner-Institute between November 2005 and December 2006.17, 18 The NVS II study part used for the probabilistic modelling measured the consumption behaviour of Germans aged 14–80 years by using two EPIC-SOFT19 assisted recall interviews per individual (2 × 24 h interviews for 13,926 individuals plus 1156 individuals with 1 × 24 h interview). The methodology selected in this work is based on the consumption of food items on a daily basis for each individual, making the additional information from food frequency interviews unsuitable for the modelling. In absence of consumption surveys covering all ages, daily based consumption data (food records or 24 h recalls) from all surveys were combined. Longitudinal differences between surveys were not assessed; however since each probabilistic iteration of the model estimates the total daily exposure on an individual level, the P99.9-criterion is considered sufficiently conservative to also identify vulnerable sub-populations without further stratification.
For the probabilistic modelling all consumption data (VELS, EsKiMo and NVS II) were converted into underlying RACs as defined in Annex I of Regulation (EU) No. 396/2005 and aggregated into 115 foods (Supplementary information, “SI”). This kind of conversion was already part of the VELS-study. For EsKiMo and NVS II, subsequent conversion of all foods was conducted using recipe data from the German Nutrient Data Base,20 amended by public literature on food processing and direct communication by manufacturers. These factors address the contribution of each RAC to the composition of complex foods (e.g., tomato in Pizza Napoli) as well as the yields for each processing step involved (e.g., juicing, baking, cooking, peeling). Consumption equivalents of the same particular RAC originating from different foods consumed within 1 day were aggregated into an overall RAC consumption equivalent like “wheat grain” or “tomato”. Exceptions were made for fruit juices, which were also referred back to their RAC but kept separate to avoid an overestimation due to their high daily consumption. This results into separate categories for e.g., “apple excluding juice” and “apple juice”. For refined assessments the percentage of edible portion was considered for a RAC, if applicable, as reported for the NVS II-model by the BfR.21
Residue Data
The German Food Monitoring provides representative data for pesticide residues in foods on the market.22, 23, 24, 25, 26, 27 In 2009 a national concept1, 2 laid out for a period of six years was implemented focusing on the dietary exposure for the German population. It was complemented by commodities scheduled in the EU Coordinated Control Program. Considering the degree of expected variability of residues in market samples, at least 188 samples have to be collected for commodities with high variability (e.g., most fresh fruit and vegetables) while mixed/blended commodities have to be monitored with at least 94 samples each. In addition, the importance of food commodities in the diet was considered to decide whether sampling has to be conducted once every three years (like apples or oranges) or once within six years (e.g., fresh herbs, Brussels sprouts). Measured residue concentrations or reported LOQ values were per se used for the modelling (empiric), however for pesticides with complex enforcement residue definitions including multiple analytes a total sum expressed as parent compound equivalents was calculated.
Toxicological Information
ADI and ARfD values were primarily drawn from the European Pesticide database of the European Commission,28 summarising agreed toxicological information for pesticides covered under the framework of Regulation (EC) No. 1107/2009.29 When no toxicological information from this database was available, ADI and ARfD values derived by other regulatory/scientific bodies (e.g., WHO) were taken into account. When no decision was made that an ARfD is unnecessary, but a specific ARfD has not been established yet for a particular compound, the ADI value was used as a conservative surrogate in the short-term dietary risk assessment. For compounds with no agreed ADI or ARfD values, the dietary exposure was expressed on a μg/kg bodyweight basis. Thresholds of toxicological concern (TTC),30 which represent empirically derived exposure levels under which toxicological effects are assumed to be of no concern, were considered for these substances. When an ARfD was not considered necessary due to low acute toxicity, no short-term dietary exposure was calculated.
The use of CF, addressing the difference between residue definitions for enforcement and risk assessment purposes, was not taken into account in screening tiers but accommodated for indirectly by applying the 10% criterion. For refined assessments, compound specific CFs were taken into account, if available.
Results and discussion
In the German food monitoring from 2009 to 2014, a total of 392 analytes were not found above the LOQ in any of the samples investigated (SI). No exposure assessment was conducted for these compounds and they are unlikely to present a public health concern for the German population.
Chronic Dietary Risk Assessment
For the chronic risk assessment, the 1st and 2nd screening tier indicated no exceedance of the 10% ADI value criterion (P99.9) for 183 and 113 compounds, respectively. In addition, 16 compounds without established toxicological reference values gave exposure levels below the lowest TTC of 0.0025 μg/kg bw/d for genotoxicity (P99.9) in the 2nd tier screening. Based on the results of both screening tiers, a chronic public health concern for the German population (SI) was not identified for residues of these substances.
A refined long-term dietary exposure assessment was conducted for 12 compounds (Table 2). For chlorpyrifos, diazinon, dimethoate and omethoate, dimethylvinphos, dithiocarbamates, hexachlorobenzene, imazalil, maleic hydrazide, pirimiphos-ethyl and prochloraz no exceedances of their respective ADI values or of the TTC for genotoxicity of 0.0025 μg/kg bw were identified at any selected percentile of the exposure distribution. These compounds are unlikely to present a chronic public health concern for the German population.
Chlorpropham gave a long-term dietary exposure of 40% of the ADI value (P99.9), which is also unlikely to present a chronic public health concern for the German population. However, the upper range of the modelling uncertainty for the extreme end of the exposure distribution (P99.99) exceeded the ADI with 170% utilisation, indicating that individuals may be exposed to chlorpropham residues above the ADI on single days. Although the probability for the long-term exposure to exceed the ADI on subsequent days is low and therefore insignificant for the German population, it is recommended to continue the analysis of chlorpropham at a high level to provide a complete picture of the exposure situation.
Both halfenprox and tricyclazole were found in one food commodity only (herbal infusions and rice grain, respectively) but the long-term dietary exposure for the German population (P99.9) exceeded the TTC for genotoxicity as both commodities represent frequently consumed foods. For halfenprox no further toxicological information concerning its genotoxicity are available while for tricyclazole at European level no toxicological reference values have been established so far owing to missing data on in vivo genotoxicity31 (p.8). Although the TTC for genotoxicity is already a very conservative approach in assessing the dietary risk, currently no conclusions on the dietary risk can be drawn for both compounds.
Copper is an element naturally occurring in many foods. The P99.9 of the exposure distribution represented 83% of the ADI value derived for copper in European plant protection legislation. Main contributors to the dietary exposure via foods were wheat (34.9%) and cacao (18.6%). However, the current assessment only considered residues in foods, whereas drinking water poses an additional source for copper. Average exposure data for copper from drinking water are not applicable as the major sources are copper plumbings in individual households themselves. The EFSA Scientific Committee on Food (SCF) derived an upper level (UL) for the daily copper intake of 5 mg/d for adults, with lower ULs for children while pregnant women were excluded from the UL32 (p.209). For an average adult of 65 kg bodyweight this UL would equal 0.077 mg/kg bw/d, which is approximately half of the ADI value derived for copper as pesticide. Owing to the missing information on drinking water, the current exposure assessment on copper is only indicative. Although the ADI is nearly completely utilised by the contribution from foods (83%) at P99.9, the UL derived by the SCF might be exceeded (~160%) for adults. In view of the lower or non-applicable ULs for the vulnerable sub-populations children and pregnant women, further investigation on the copper exposure in the German population is recommended, especially in combination with drinking water.
Acute Dietary Risk Assessment
For the short-term dietary exposure assessment the 1st tier screening identified 196 compounds (including compounds for which no ARfD is necessary) and the 2nd tier screening additional 84 compounds below the 10% ARfD criterion (P99.9). The short-term exposure of 14 compounds in the 2nd screening tier without established toxicological reference values was below the TTC for genotoxicity of 0.0025 μg/kg bw/d (P99.9). Based on the results of both screening tiers, an acute public health concern for the German population was not identified for residues of these substances (SI).
A refined short-term dietary exposure assessment was conducted for 14 compounds (Table 3). For mirex and pirimiphos-ethyl, no exceedance of their toxicological reference values was identified. Residues of mirex and pirimiphos-ethyl are unlikely to present an acute public health concern for the German population.
Chlorpropham, cyhalothrin (excluding lambda-cyhalothrin), deltamethrin, ethephon, hexachlorobenezene and imazalil resulted in short-term dietary exposures below their respective ARfDs at the P99.9. Based on this percentile it is unlikely that residues of these substances present an acute public health concern for the German population. However, taking into account the ranges of modelling uncertainty, the extreme upper end (P99.99) of the exposure distribution exceeded the respective ARfDs or the TDI for hexachlorobenzene. The probability for such an exceedance is low and therefore insignificant for the German population but cannot be fully excluded for all combinations of large portions and high residues. It is recommended that the analysis of these compounds in monitoring is maintained at a high level to assess the future exposure situation. Hexachlorobenzene is a persistent organic pollutant according to the Stockholm Protocol.33 Its use is globally banned, but owing to the long environmental half-life times accumulation especially in fish, fatty tissues and milk is inevitable. It is recommended to reduce its occurrence as far as technically possible.
For chlorpyrifos, the refined short-term dietary exposure indicated a nearly complete utilisation of the ARfD with 99% (P99.9) with an exceedance of 104% of the ARfD taking into account the upper range of modelling uncertainty. Also, the 1st and 2nd tier runs for chlorpyrifos indicated no significant influence of <LOQ values, showing utilisations of the ARfD of 90% and 114%, respectively. The higher ARfD utilisation in the 2nd tier run compared with the 1st tier run is based on the high variability of the short-term exposure modelling without upper/lower bound estimation of the modelling range for screening. The refined short-term exposure for dimethoate and omethoate, which are closely rated compounds and were assessed for their cumulative dietary risks34 (p.18), gave a lower average utilisation of the ARfD with 21%. However, the upper range of the modelling uncertainty at the P99.9 represented 280% of the ARfD, indicating a high variability in the results. This variability may have been increased by single samples exceeded the MRLs and also the ARfD based on the IESTI-concept. Random selection of residue concentrations measured in such samples during probabilistic modelling would results in total exposure levels highly above levels based on MRL-compliant samples. For both compounds highest food contributors to the short-term exposure were broadly spread as these substances are frequently found in monitoring samples. For chlorpyrifos, apples excluding juice (42.2%), pears (30.6%), bananas (9.9%) and peaches (7.6%) contributed mostly to the total exposure while for dimethoate and omethoate potatoes (26.1%), cherries excluding juice (18.3%), spinach (12.6%) and barley (10.2%) were the main contributors. Taking into account the range of modelling uncertainty, residues of chlorpyrifos and cumulative exposure to dimethoate and omethoate in foods may present an acute public health concern for the German population. A general reduction strategy for residues of these compounds in foods is recommended.
The refined assessment for chlorobromuron indicated a short-term dietary exposure below the TTC for genotoxicity of 0.0025 μg/kg bw (P99.9). Based on the TTC approach it is unlikely that residues of chlorobromuron present an acute public health concern for the German population.
For dimethylvinpos, halfenprox and tricyclazole the short-term dietary exposure (P99.9) was above the TTC for genotoxicity of 0.0025 μg/kg bw used as a conservative surrogate in absence of specific toxicological data. Each compound was found in one food commodity only (wine, herbal infusions and rice grain, respectively); however these commodities represent frequently consumed foods. For diemethylvinphos and halfenprox no further toxicological information concerning its genotoxicity are available while for tricyclazole at European level no toxicological reference values have been established so far owing to missing data on the in vivo genotoxicity31 (p.8). Although the TTC for genotoxicity is already a very conservative approach in assessing the dietary risk, currently no conclusions on the dietary risk can be drawn for these compounds.
Conclusions
The risk for the German population, which might arise from the acute and chronic dietary exposure of pesticide residues, was assessed by a two-step screening approach followed by refinement, if necessary. The probabilistic model used provides full consideration of all foods eaten within a day for the estimation of the total exposure. However, short-term exposure results are difficult to compare with the IESTI-methodology. Single samples containing residue concentrations, which would exceed the ARfD based on the IESTI, may become insignificant only affecting the high percentiles of the exposure distribution above the P99.9. Also, the model is sensible to foods with a high percentage of detected residues in combination with a low number of total samples. Owing to the random selection of data, residue concentrations above the LOQ are overrepresented, also resulting in an overestimation for the total exposure. As final result, for 693 of 700 compounds a chronic or acute public health concern was unlikely for the German population. On the other hand, the refined assessments indicated that the short-term exposures for chlorpyrifos and for dimethoate and omethoate, assessed for their cumulative dietary risks, may present an acute public health concern for the German population. A general reduction of chlorpyrifos, dimethoate and omethoate residues in foods is recommended.
For copper the current assessment is only indicative, because drinking water as a major source of exposure was not yet taken into account. The ADI value derived for copper used as pesticide was nearly completely utilised by the exposure via foods, whereas the UL derived by the SCF might even be exceeded. Further investigations of the exposure of copper in the German population, especially under consideration of drinking water, are recommended before final conclusions on the dietary consumer risk can be drawn. However, the results already indicate that the long-term dietary exposure for copper might exceed tolerable levels and further reduction might be inevitable.
Dimethylvinphos, halfenprox and tricyclazole exceeded the TTC for genotoxicity either in the long- or short-term dietary exposure. As information to exclude a genotoxic potential for these compounds is not available, the dietary risk assessment remains inconclusive.
By applying available probabilistic methodologies, an important aspect is added to the dietary consumer risk assessment by consideration of the total daily consumption, especially for the short-term dietary exposure. Using the advantageous general features of probabilisitic assessments like the distribution of exposures amongst multiple individuals and the variability in food consumption, a more comprehensive assessment of the dietary exposure can be achieved. Another beneficial aspect is the application of the TTC concept, improving the interpretation of results for compounds where adequate toxicological reference values are not available. Further investigations on chronic and acute cumulative risks arising from compounds with similar targets of toxicity are required in the future to improve the assessment of dietary risks for consumers exposed to pesticide residues.
References
Sieke C, Lindtner O, Banasiak U . Pflanzenschutzmittelrückstände, Nationales Monitoring, Abschätzung der Verbraucherexposition: Teil 1. Deutsche Lebensmittel-Rundschau 2008; 104, (2008) 6 271–279.
Sieke C, Lindtner O, Banasiak U . Pflanzenschutzmittelrückstände, Nationales Monitoring, Abschätzung der Verbraucherexposition: Teil 2. Deutsche Lebensmittel-Rundschau 2008; 104, (2008) 7 336–342.
Global Environment Monitoring System – Food Contamination Monitoring and Assessment Programme (GEMS/Food). Guidelines for predicting dietary intake of pesticide residues (revised). 1997;WHO/FSF/FOS/97.7:41.
World Health Organization (WHO). Food consumption and exposure assessment of chemicals - Report of a FAO/WHO Consultation, Geneva, Switzerland 10-14 February 1997. 1997(WHO/FSF/FOS/97.5).
Food and Agriculture Organization of the United Nations (FAO) Submission and evaluation of pesticide residue data for the estimation of maximum residue levels in food and feed (3rd edn). Food and Agriculture Organization of the United Nations (FAO): Rome, Italy, 2016.
European Food Safety Authority (EFSA). Pestice Residue Intake Model ("PRIMo") Rev.2 2007. Available at http://www.efsa.europa.eu/sites/default/files/assets/calculationacutechronic_2.xls.Accessed on 1 March 2016.
Dutch National Institute for Public Health and the Environment (RIVM). MCRA 8.1 Reference Manual: WUR/Biometris, FERA and RIVM; 2015. Available at https://mcra.rivm.nl. Accessed on 1 March 2016.
EFSA Panel on Plant Protection Products and their Residues (PPR). Guidance on the Use of Probabilistic Methodology for Modelling Dietary Exposure to Pesticide Residues. EFSA Journal 2012.
U.S. Environmental Protection Agency - Office of Pesticide Programs. Choosing a percentile of acute dietary exposure as a threshold of regulatory concern. Washington, D.C. (USA): U.S. Environmental Protection Agency (EPA), 2000 16.03.2000. Report No.: Contract No.: 6046.
OECD. Test No. 501: Metabolism in Crops: OECD Publishing 2007.
van Klaveren JD, Goedhart PW, Wapperom D, vdV H . A European tool for usual intake distribution estimation in relation to data collection by EFSA - External Scientific Report. EFSA Supporting Publications. 2012;2012:EN-300:42.
Food and Agriculture Organization of the United Nations (FAO). Pesticide Residues in Food - Report of the 2003 Joint FAO/WHO Meeting. Rome: FAO, 2003 No. 176.
Heseker H, Oepping A, Vohmann C . Nutrition survey to determine the food intake of babies and infants for the estimation of the exposure with pesticide residues - VELS 2003.
Mensink GBM, Bauch A, Vohmann C, Stahl A, Six J, Kohler S et al. EsKiMo – Das Ernährungsmodul im Kinder- und Jugendgesundheitssurvey (KiGGS). Bundesgesundheitsbl 2007; 50: 902–908.
Mensink GBM, Heseker H, Richter A, Stahl A, Vohmann C . Forschungsbericht - Ernährungsstudie als KiGGS-Modul (EsKiMo) 2007;143.
Hölling H, Schlack R, Kamtsiuris P, Butschalowsky H, Schlaud M, Kurth BM . Die KiGGS-Studie. Bundesgesundheitsbl 2012; 55: 836–842.
Brombach C, Wagner U, Eisinger-Watzl M, Heyer A . Die Nationale Verzehrsstudie II - Ziel: Aktuelle und belastbare Primärdaten für die Ernährungsberichterstattung des Bundes generieren. Ernährungs-Umschau 2006; 53: 5.
Krems C, Bauch A, Götz A, Heuer T, Hild A, Möseneder J et al. Methoden der Nationalen Verzehrsstudie II. Ernährungs-Umschau 2006; 53: 6.
Voss S, URC, Slimani N, Kroke A, Riboli E, Wahrendorf J et al. EPIC-SOFT a European dietary assessment instrument for 24-h recalls. Eur J Nutr 1998; 1998/3: 227–233.
Max Rubner-Institut (MRI). German Nutrient Data Base. Available at http://www.bls.nvs2.de/index.php?id=39&L=1.Accessed on 1 March 2016.
Federal Institute for Risk Assessment (BfR). BfR develops a new dietary intake model for the German population aged 14 to 80 years in order to calculate the intake of pesticide residues in food [Opinion]. Updated on 19 October 2011. Available at http://www.bfr.bund.de/cm/349/bfr-model-for-pesticide-residue-intake-calculations-nvs2.zip.Accessed on 1 March 2016.
Bundesamt für Verbraucherschutz und Lebensmittelsicherheit (BVL). Berichte zur Lebensmittelsicherheit 2009: Springer Basel 2011.
Bundesamt für Verbraucherschutz und Lebensmittelsicherheit (BVL). Berichte zur Lebensmittelsicherheit 2010: Springer Basel 2012.
Bundesamt für Verbraucherschutz und Lebensmittelsicherheit (BVL). Berichte zur Lebensmittelsicherheit 2011: Springer Basel 2013.
Bundesamt für Verbraucherschutz und Lebensmittelsicherheit (BVL). Berichte zur Lebensmittelsicherheit 2012: Springer International Publishing 2014.
Bundesamt für Verbraucherschutz und Lebensmittelsicherheit (BVL). Berichte zur Lebensmittelsicherheit 2013: Birkhäuser Basel 2015.
Bundesamt für Verbraucherschutz und Lebensmittelsicherheit (BVL). Data from the German Food Monitoring for 2014 (personal communication) 2016.
European Commission (COM). EU - Pesticides database: European Commission (COM). Available at http://ec.europa.eu/food/plant/pesticides/eu-pesticides-database. Accessed on 1 March 2016.
Regulation (EC) No 1107/2009 of the European Parliament and of the Council of 21 October 2009 concerning the placing of plant protection products on the market and repealing Council Directives 79/117/EEC and 91/414/EEC, 2009, O.J. L 309/1.
European Food Safety Authority (EFSA). Scientific Opinion on Exploring options for providing advice about possible human health risks based on the concept of Threshold of Toxicological Concern (TTC). Parma, Italy: EFSA, 2012 02.07.2012. Report No.: 10(7):2750.
European Food Safety Authority (EFSA). Conclusion on the peer review of the pesticide risk assessment of the active substance tricyclazole. EFSA J 2015; 13.
EFSA Scientific Panel on Dietetic Products NaAS. Tolerable Upper Intake Levels for Vitamins and Minerals. European Food Safety Authority (EFSA) 2006.
Vanden Bilcke C . The stockholm convention on persistent organic pollutants. Rev Eur Commun Int Envtl L 2002; 11: 328–342.
European Food Safety Authority (EFSA). Conclusion regarding the peer review of the pesticide risk assessment of the active substance dimethoate. EFSA 2006; 4.
European Food Safety Authority (EFSA). Review of the existing maximum residue levels (MRLs) for chlorpropham according to Article 12 of Regulation (EC) No 396/2005. EFSA J 2012; 10.
European Food Safety Authority (EFSA). Refined risk assessment regarding certain maximum residue levels (MRLs) of concern for the active substance chlorpyrifos. EFSA J 2015; 13.
European Food Safety Authority (EFSA). Reasoned opinion on the review of the existing maximum residue levels (MRLs) for lambda‐cyhalothrin according to Article 12 of Regulation (EC) No 396/2005. EFSA J 2014; 12.
European Food Safety Authority (EFSA). Review of the existing maximum residue levels for deltamethrin according to Article 12 of Regulation (EC) No 396/2005. EFSA J 2015; 13.
European Food Safety Authority (EFSA). Conclusion regarding the peer review of the pesticide risk assessment of the active substance diazinon. EFSA J 2006; 4.
European Food Safety Authority (EFSA). Conclusion regarding the peer review of the pesticide risk assessment of the active substance ethephon. EFSA J 2006; 4.
Food and Agriculture Organization of the United Nations (FAO). Pesticide Residues in Food - Report of the 2015 Joint FAO/WHO Meeting. Rome: FAO, 2015 No. 223.
European Food Safety Authority (EFSA). Conclusion on the peer review of the pesticide risk assessment of the active substance imazalil. EFSA J 2010; 8.
Federal Institute for Risk Assessment (BfR). Variability factors for the acute dietary risk assessment of pesticides Berlin, Germany: BfR; 2010. Available at http://www.bfr.bund.de/cm/349/variability_factors_for_the_acute_dietary_risk_assessment_of_pesticides.pdf. Accessed on 1 March 2016.
European Food Safety Authority (EFSA). Review of the existing maximum residue levels (MRLs) for maleic hydrazide according to Article 12 of Regulation (EC) No 396/2005. EFSA J 2011; 9.
European Food Safety Authority (EFSA). Conclusion on the peer review of the pesticide risk assessment of the active substance prochloraz. EFSA J 2011; 9.
Food and Agriculture Organization of the United Nations (FAO). Pesticide Residues in Food - Report of the 2004 Joint FAO/WHO Meeting. Rome, Italy: FAO, 2004 No. 182/1.
World Health Organization (WHO). Environmental Health Criteria 195 - Hexachlorobenzene. Geneva, Switzerland: World Health Organization (WHO) 1997.
U.S. Environmental Protection Agency (EPA). Integrated Risk Information System (IRIS) - Mirex. U.S. Environmental Protection Agency (EPA) 1992.
Australian Government - Department of Health Office of Chemical Safety. Acceptable Daily Intakes for Agricultural and Veterinary Chemicals as of 31 December 2015. Updated on 31 December 2015. Available at http://www.health.gov.au/internet/main/publishing.nsf/content/ocs-adi-list.htm.Accessed on 1 March 2016.
Acknowledgements
We would like to thank the MCRA team at the RIVM, especially Dr. J. van Klaveren and G. van Donkersgoed and Dr. O. Lindtner and C. Sommerfeld from the BfR for their support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Disclaimer
The material presenting is original research, has not been published previously and has not been submitted for the publication elsewhere while under consideration.
Supplementary Information accompanies the paper on the Journal of Exposure Science and Environmental Epidemiology website
Supplementary information
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/4.0/
About this article
Cite this article
Sieke, C., Michalski, B. & Kuhl, T. Probabilistic dietary risk assessment of pesticide residues in foods for the German population based on food monitoring data from 2009 to 2014. J Expo Sci Environ Epidemiol 28, 46–54 (2018). https://doi.org/10.1038/jes.2017.7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/jes.2017.7
Keywords
This article is cited by
-
Dietary zinc intake and body mass index as modifiers of the association between household pesticide exposure and infertility among US women: a population-level study
Environmental Science and Pollution Research (2022)
-
The dietary risk index system: a tool to track pesticide dietary risks
Environmental Health (2020)