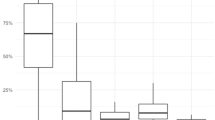
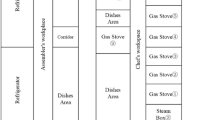
Abstract
Personal exposure to ultrafine particles (UFP) can occur while people are cooking, driving, smoking, operating small appliances such as hair dryers, or eating out in restaurants. These exposures can often be higher than outdoor concentrations. For 3 years, portable monitors were employed in homes, cars, and restaurants. More than 300 measurement periods in several homes were documented, along with 25 h of driving two cars, and 22 visits to restaurants. Cooking on gas or electric stoves and electric toaster ovens was a major source of UFP, with peak personal exposures often exceeding 100,000 particles/cm3 and estimated emission rates in the neighborhood of 1012 particles/min. Other common sources of high UFP exposures were cigarettes, a vented gas clothes dryer, an air popcorn popper, candles, an electric mixer, a toaster, a hair dryer, a curling iron, and a steam iron. Relatively low indoor UFP emissions were noted for a fireplace, several space heaters, and a laser printer. Driving resulted in moderate exposures averaging about 30,000 particles/cm3 in each of two cars driven on 17 trips on major highways on the East and West Coasts. Most of the restaurants visited maintained consistently high levels of 50,000–200,000 particles/cm3 for the entire length of the meal. The indoor/outdoor ratios of size-resolved UFP were much lower than for PM2.5 or PM10, suggesting that outdoor UFP have difficulty in penetrating a home. This in turn implies that outdoor concentrations of UFP have only a moderate effect on personal exposures if indoor sources are present. A time-weighted scenario suggests that for typical suburban nonsmoker lifestyles, indoor sources provide about 47% and outdoor sources about 36% of total daily UFP exposure and in-vehicle exposures add the remainder (17%). However, the effect of one smoker in the home results in an overwhelming increase in the importance of indoor sources (77% of the total).
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 6 print issues and online access
$259.00 per year
only $43.17 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout









Similar content being viewed by others
References
Abt E., Suh H.H., Catalano P., and Koutrakis P. Relative contribution of outdoor and indoor particle sources to indoor concentrations. Environ Sci Tech 2000: 34: 3579–3587.
Afshari A., Matson U., and Ekberg L.E. Characterization of indoor sources of fine and ultrafine particles: a study conducted in a full-scale chamber. Indoor Air 2005: 15: 141–150.
ATUS. American Time Use Survey, Bureau of Labor Statistics, Washington, DC. 2009: http://www.bls.gov/news.release/atus.nr0.htm (accessed 4th July 2009).
Bräuner E.V., Forchhammer L., Møller P., Barregard L., Gunnarsen L., Afshari A., Wåhlin P., Glasius M., Dragsted L.O., Basu S., Raaschou-Nielsen O., and Loft S. Indoor particles affect vascular function in the aged: an air filtration–based intervention study. Am J Resp Crit Care Med 2007a: 177: 419–425.
Bräuner E.V., Forchhammer L., Møller P., Simonsen J., Glasius M., Wåhlin P., Raaschou-Nielsen O., and Loft S. Exposure to ultrafine particles from ambient air and oxidative stress–induced DNA damage. Environ Health Persp 2007b: 115: 1177–1182.
Dennekamp M., Howarth S., Dick C.A., Cherrie J.H.W., Donaldson K., and Seaton A. Ultrafine particles and nitrogen oxides generated by gas and electric cooking. Occup Environ Med 2001: 58: 511–516.
Fruin S., Westerdahl D., Sax T., Sioutas C., and Fine P.M. Measurements and predictors of on-road ultrafine particle concentrations and associated pollutants in Los Angeles. Atmos Environ 2008: 42: 207–219.
Hämeri K., Koponen I.K., Aalto P.P., and Kulmala M. The particle detection efficiency of the TSI-3007 condensation particle counter. J Aerosol Science 2002: 33: 1463–1469.
He C., Morawska L., Hitchins J., and Gilbert D. Contribution of indoor sources to particle number and mass concentrations in residential houses. Atmos Environ 2004: 38: 3405–3415.
Hoek G., Kos G., Harrison R., de Hartog J., Meliefste K., ten Brink H., Katsouyanni K., Karakatsani A., Liangou M., Kotronarou A., and Hämeri K., et al. Indoor-outdoor relationships of particle number and mass in four European cities. Atmos Eviron 2008: 42: 156–169.
Howard-Reed C.H., Wallace L.A., and Ott W.R. The effect of opening windows on air change rates in two homes. J Air Waste Manage Assoc 2002: 52: 147–159.
Klepeis N.E., Apte M.G., Gundel L.A., Sextro R.G., and Nazaroff W.W. Determining size-specific emission factors for environmental tobacco smoke particles. Aerosol Sci Technol 2003: 37: 780–790.
Long C.M., Suh H.H., Catalano P., and Koutrakis P Using time- and size-resolved particulate data to quantify indoor penetration and deposition behavior. Environ Sci Technol 2001: 35: 2089–2099.
Oberdörster G., Oberdörster E., and Oberdörster J. Nanotoxicology: an emerging discipline evolving from studies of ultrafine particles. Environ Health Persp 2005: 113: 823–839.
Ott W., Klepeis N., and Switzer P. Air change rates of motor vehicles and in-vehicle concentrations from secondhand smoke. J Exposure Science Environ Epidemiol 2008: 18 (3): 312–325.
Ott W., Wallace L., and Mage D. Predicting particulate (PM10) personal exposure distributions using a random component superposition statistical model. J Air Waste Manage Assoc 2000: 50: 1390–1406.
Solomon P.A., Hopke P.K., Froines J., and Scheffe R. Key scientific findings and policy and health-relevant insights from the U.S. Environmental Protection Agency's Particulate Matter Supersites Program and related studies: an integration and synthesis of results. J Air Waste Manage Assoc 2008: 58 (Suppl. 2008): S-3–S-92.
Stölzel M., Breitner S., Cyrys J., Pitz M., Wölke G., Kreyling W., Heinrich J., Wichmann H.-E., and Peters A. Daily mortality and particulate matter in different size classes in Erfurt, Germany. J Expo Sci Environ Epidemiol 2007: 17: 458–467.
Wallace L.A Real-time monitoring of particles, PAH, and CO in an occupied townhouse. Applied Occup Environ Hygiene 2000: 15 (1): 39–47.
Wallace L.A. Ultrafine particles from a vented gas clothes dryer. Atmos Environ 2005: 39: 5777–5786.
Wallace L.A. Indoor sources of ultrafine and accumulation mode particles: number concentrations and size distributions. Aerosol Sci Technol 2006: 40: 348–360.
Wallace L.A., Emmerich S.J., and Howard-Reed C. Emission rates of ultrafine and fine particles due to cooking with a gas stove. Environ Sci Technol 2004: 38: 2304–2311.
Wallace L.A., and Howard-Reed C. Continuous monitoring of ultrafine, fine, and coarse particles in a residence for 18 months in 1999–2000. J Air Waste Manage Assoc 2002: 52: 828–844.
Wallace LA, Wang F, Howard-Reed C, and Persily A. Contribution of gas and electric stoves to residential ultrafine particle concentrations between 2 and 64 nm: size distributions and emission and coagulation rates. Environ Sci Tech 2008: 42: 8641–8647.
Weichenthal S., Dufresne A, Infante-Rivard C., and Joseph L. Determinants of ultrafine particle exposures in transportation environments: findings of an 8-month survey conducted in Montreal, Canada. J Exposure Science Environ Epidemiol 2008: 18: 551–563.
Westerdahl D., Fruin S., Sax T., Fine P.M., and Sioutas C. Mobile platform measurements of ultrafine particles and associated pollutant concentrations on freeways and residential streets in Los Angeles. Atmos Environ 2005: 39: 3597–3610.
Wichmann H.E., Spix C., Tuch T., Wolke G., Peters A., Heinrich J., Kreyling W.G., and Heyder J. Daily mortality and fine and ultrafine particles in erfurt, Germany Part I: role of particle number and particle mass. Health Effects Inst 2000: 98: 5–86.
Wu Z., Hu M., Lin P., Liu S., Wehner B., and Wiedensohler A. Particle number size distribution in the urban atmosphere. Atmos Environ 2008: 42: 7967–7980.
Zhu Y., Personal communication on October 14 at the ISEA-ISEE 2008 Joint Annual Meeting in Pasadena, CA. 2008.
Zhu Y., Fung D.C, Kennedy N., Hinds W.C., and Eiguran-Fernandez A. Measurements of ultrafine particles and other vehicular pollutants inside a mobile exposure system on Los Angeles freeways. J Air Waste Manage Assoc 2008: 58 (3): 424–434.
Zhu Y., Hinds W.C., Kim S., and Sioutas C. Concentration and size distribution of ultrafine particles near a major highway. J Air Waste Manage Assoc 2002: 52 (9): 1032–1042.
Acknowledgements
One of us (LW) acknowledges Andrew Persily and members of his group at the National Institute of Standards and Technology for supplying the Model 3007 and the test house on the NIST campus where the indoor–outdoor measurements were made. WO gratefully acknowledges the assistance of Jane McAteer in monitoring several California restaurants, and the Flight Attendants’ Medical Research Institute for support. We also acknowledge TSI for loaning a second Model 3007 to determine the precision of the instrument.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Wallace, L., Ott, W. Personal exposure to ultrafine particles. J Expo Sci Environ Epidemiol 21, 20–30 (2011). https://doi.org/10.1038/jes.2009.59
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/jes.2009.59
Keywords
This article is cited by
-
Airway and systemic biomarkers of health effects after short-term exposure to indoor ultrafine particles from cooking and candles – A randomized controlled double-blind crossover study among mild asthmatic subjects
Particle and Fibre Toxicology (2023)
-
Cooking methods and kitchen ventilation availability, usage, perceived performance and potential in Canadian homes
Journal of Exposure Science & Environmental Epidemiology (2023)
-
Characteristics of outdoor pollutants intrusion and ventilation control in sentry buildings with normal openings
Building Simulation (2023)
-
Pot study using Chlorophytum comosum plants to biomonitor PAH levels in domestic kitchens
Environmental Science and Pollution Research (2023)
-
Exploring the influence of ventilation settings and fan strength on passenger car in-cabin particle number concentration
Air Quality, Atmosphere & Health (2022)