Abstract
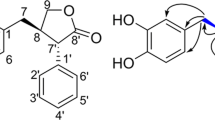
The novel 11β-hydroxysteroid dehydrogenase type 1 (11β-HSD1) inhibitors known as sterenin A, B, C and D were found in a solid-state culture of the producing basidiomycetes identified as Stereum sp. SANK 21205. Purification of the 50% aq Me2CO extract of the culture was performed by EtOAc extraction, reversed phase opencolumn chromatography and successive ODS HPLC preparation. These compounds, whose structures were determined by several spectroscopic methods, were found to be novel isoindolinone alkaloids which exhibited potent selective inhibitory activities against 11β-HSD1.
Similar content being viewed by others
Article PDF
References
Seckl JR, Walker BR . 11β-Hydroxysteroid dehydrogenase type 1 — A tissue-specific amplifier of glucocorticoid action. Endocrinology 142: 1371–1376 ( 2001)
Montague CT, O'Rahilly S . The perils of portliness: causes and consequences of visceral adiposity. Diabetes 49: 883–888 ( 2000)
Livingstone DEW, Kenyon CJ, Walker BR . Mechanisms of dysregulation of 11β-hydroxysteroid dehydrogenase type 1 in obese Zucker rats. J Endocrinol 167: 533–539 ( 2000)
Rask E, Olsson T, Soderberg S, Andrew R, Livingstone DEW, Johnson O, Walker BR . Tissue-specific dysregulation of cortisol metabolism in human obesity. J Clin Endocrinol Metab 86: 1418–1421 ( 2001)
Paulmyer-Lacroix O, Boullu S, Oliver C, Marie-Christine Alessi MC, Grino M . Expression of the mRNA coding for 11β-hydroxysteroid dehydrogenase type 1 in adipose tissue from obese patients: An in situ hybridization study. J Clin Endocrinol Metab 87: 2701–2705 ( 2002)
Kotelevtsev Y, Holmes MC, Burchell A, Houston PM, Schmoll D, Jamieson P, Best R, Brown R, Edwards CRW, Seckl JR, Mullins JJ . 11β-Hydroxysteroid dehydrogenase type 1 knockout mice show attenuated glucocorticoid-inducible responses and resist hyperglycemia on obesity or stress. Proc Natl Acad Sci USA 94: 14924–14929 ( 1997)
Masuzaki H, Paterson J, Shinyama H, Morton NM, Mullins JJ, Seckl JR, Flier JS . A transgenic model of visceral obesity and the metabolic syndrome. Science 294: 2166–2170 ( 2001)
Chamuris GP . The non-stipitate stereoid fungi in the northeastern United States and adjacent Canada. Mycological Memoir 14: 93–98 ( 1988)
Frincke JM, Faulkner DJ . Antimicrobial metabolites of the sponge Reniera sp. J Am Chem Soc 104: 265–269 ( 1982)
Xu X, de Guzman FS, Gloer JB, Shearer CA . Stachybotrins A and B: Novel bioactive metabolites from a brackish water isolate of the fungus Stachybotrys sp. J Org Chem 57: 6700–6703 ( 1992)
Shinobara C, Hasumi K, Hasumi W, Endo A . Staplabin, a novel triprenyl phenol which stimulates the binding of plasminogen to fibrin and U937 cells. J Antibiot 49: 961–966 ( 1996)
Kohyama T, Hasumi K, Hamanaka A, Endo A . SMTP-1 and -2, novel analogs of staplabin produced by Stachybotrys microspora IFO30018. J Antibiot 50: 172–174 ( 1997)
Hasumi K, Ohyama S, Kohyama T, Ohsaki Y, Takayasu R, Endo A . Isolation of SMTP-3, -4, -5 and -6, novel analogs of staplabin, and their effects on plasminogen activation and fibrinolysis. J Antibiot 51: 1059–1068 ( 1998)
Hu W, Ohyama S, Hasumi K . Activation of fibrinolysis by SMTP-7 and -8, novel staplabin analogs with a pseudosymmetric structure. J Antibiot 53: 241–247 ( 2000)
Hu W, Kitano Y, Hasumi K . SMTP-4D -5D, -6D -7D and -8D, a new series of the non-lysine-analog plasminogen modulators with a D amino acid moiety. J Antibiot 56: 832–837 ( 2003)
Hasumi K, Hasegawa K, Kitano Y . Isolation and absolute configuration of SMTP-0, a simplest congener of the SMTP family nonlysine-analog plasminogen modulators. J Antibiot 60: 463–468 ( 2007)
Kurasawa S, Takeuchi T, Umezawa H . Studies on glyoxalase inhibitor: Isolation of a new active agent, MS-3, from a mushroom. Agric Biol Chem 39: 2003–2008 ( 1975)
Kurasawa S, Naganawa H, Takeuchi T, Umezawa H . The structure of MS-3: A glyoxalase I inhibitor produced by a mushroom. Agric Biol Chem 39: 2009–2014 ( 1975)
Nakamura H, Iitaka Y, Kurasawa S, Takeuchi T, Umezawa H . The crystal structure of a dibromo-derivative of MS-3: A glyoxalase I inhibitor produced by a mushroom, Stereum hirsutum. Agric Biol Chem 40: 1781–1784 ( 1976)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ito-Kobayashi, M., Aoyagi, A., Tanaka, I. et al. Sterenin A, B, C and D, Novel 11β-Hydroxysteroid Dehydrogenase Type 1 Inhibitors from Stereum sp. SANK 21205. J Antibiot 61, 128–135 (2008). https://doi.org/10.1038/ja.2008.121
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ja.2008.121
Keywords
This article is cited by
-
Fruitful decade of fungal metabolites as anti-diabetic agents from 2010 to 2019: emphasis on α-glucosidase inhibitors
Phytochemistry Reviews (2021)
-
Lanostane triterpenoids from fruiting bodies of basidiomycete Stereum sp., structures and biological activities
The Journal of Antibiotics (2017)
-
Colletoic Acid, a Novel 11β-Hydroxysteroid Dehydrogenase Type 1 Inhibitor from Colletotrichum gloeosporioides SANK 21404
The Journal of Antibiotics (2008)