Abstract
Background/Objectives:
A major risk factor of type 2 diabetes mellitus (T2DM) is a positive family history of diabetes. First degree relatives (FDR) of patients with T2DM are more insulin resistant and are reported to have larger abdominal subcutaneous adipocytes than adults without a family history. Our objectives were to assess whether FDR of T2DM are associated with larger abdominal adipocytes independent of age, sex and abdominal subcutaneous fat and to assess whether a family history of T2DM is also independently related to femoral adipocyte size, as well as visceral fat and fasting plasma triglyceride (TG) concentrations.
Methods:
We extracted adipocyte size, body composition, plasma TG and demographic data of non-diabetic research participants of previous studies conducted in our laboratory. We ascertained the family history of T2DM from the electronic medical records. Multivariate regression analysis was used to assess whether FDR of T2DM are more likely to have other risk factors after adjusting for known covariates.
Results:
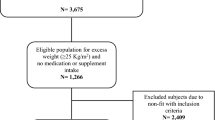
Of 604 participants, 148 were FDR of T2DM. Although abdominal and femoral adipocyte size was greater in FDR of T2DM than those without a family history (0.74±0.33 vs 0.63±0.33 μg lipid per cell, P<0.001; 0.81±0.29 vs 0.72±0.33 μg lipid per cell, P=0.01, respectively), this was confounded by FDR of T2DM being older, having greater body mass index and percent body fat. A family history of T2DM was a significant predictor of abdominal adipocyte size after adjustment for age and body fat distribution parameters in females (total R2=0.5, P<0.0001), but not in males. A family history of T2DM was not independently predictive of femoral adipocyte size, visceral fat area or TG.
Conclusions:
Female FDR of T2DM have larger abdominal, but not femoral, adipocytes, even after accounting for age and body fat distribution.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Kahn CR, Vicent D, Doria A . Genetics of non-insulin-dependent (type-II) diabetes mellitus. Annu Rev Med 1996; 47: 509–531.
Scott RA, Langenberg C, Sharp SJ, Franks PW, Rolandsson O, Drogan D et al. The link between family history and risk of type 2 diabetes is not explained by anthropometric, lifestyle or genetic risk factors: the EPIC-InterAct study. Diabetologia 2013; 56: 60–69.
Wagner R, Thorand B, Osterhoff MA, Muller G, Bohm A, Meisinger C et al. Family history of diabetes is associated with higher risk for prediabetes: a multicentre analysis from the German Center for Diabetes Research. Diabetologia 2013; 56: 2176–2180.
Vaag A, Henriksen JE, Beck-Nielsen H . Decreased insulin activation of glycogen synthase in skeletal muscles in young nonobese Caucasian first-degree relatives of patients with non-insulin-dependent diabetes mellitus. J Clin Invest 1992; 89: 782–788.
Hirsch J, Knittle JL . Cellularity of obese and nonobese human adipose tissue. Fed Proc 1970; 29: 1516–1521.
Krotkiewski M, Bjorntorp P, Sjostrom L, Smith U . Impact of obesity on metabolism in men and women: importance of regional adipose tissue distribution. J Clin Invest 1983; 72: 1150–1162.
Arner E, Westermark PO, Spalding KL, Britton T, Ryden M, Frisen J et al. Adipocyte turnover: relevance to human adipose tissue morphology. Diabetes 2010; 59: 105–109.
Arner P, Arner E, Hammarstedt A, Smith U . Genetic predisposition for Type 2 diabetes, but not for overweight/obesity, is associated with a restricted adipogenesis. PLoS One [Electronic Resource] 2011; 6: e18284.
Yang J, Eliasson B, Smith U, Cushman SW, Sherman AS . The size of large adipose cells is a predictor of insulin resistance in first-degree relatives of type 2 diabetic patients. Obesity (Silver Spring) 2012; 20: 932–938.
Henninger AM, Eliasson B, Jenndahl LE, Hammarstedt A . Adipocyte hypertrophy, inflammation and fibrosis characterize subcutaneous adipose tissue of healthy, non-obese subjects predisposed to type 2 diabetes. PloS One 2014; 9: e105262.
Despres J-P, Lemieux I . Abdominal obesity and metabolic syndrome. Nature 2006; 444: 881–887.
Britton KA, Massaro JM, Murabito JM, Kreger BE, Hoffmann U, Fox CS . Body fat distribution, incident cardiovascular disease, cancer, and all-cause mortality. J Am Coll Cardiol 2013; 62: 921–925.
McLaughlin T, Lamendola C, Liu A, Abbasi F . Preferential fat deposition in subcutaneous versus visceral depots is associated with insulin sensitivity. J Clin Endocrinol Metab 2011; 96: E1756–E1760.
Neeland IJ, Turer AT, Ayers CR, Powell-Wiley TM, Vega GL, Farzaneh-Far R et al. Dysfunctional adiposity and the risk of prediabetes and type 2 diabetes in obese adults. JAMA 2012; 308: 1150–1159.
Bouchard C, Rice T, Lemieux S, Despres JP, Perusse L, Rao DC . Major gene for abdominal visceral fat area in the Quebec Family Study. Int J Obes Relat Metab Disord 1996; 20: 420–427.
Wajchenberg BL . Subcutaneous and visceral adipose tissue: their relation to the metabolic syndrome. Endocr Rev 2000; 21: 697–738.
Johanson EH, Jansson PA, Lonn L, Matsuzawa Y, Funahashi T, Taskinen MR et al. Fat distribution, lipid accumulation in the liver, and exercise capacity do not explain the insulin resistance in healthy males with a family history for type 2 diabetes. J Clin Endocrinol Metab 2003; 88: 4232–4238.
Mitchell BD, Zaccaro D, Wagenknecht LE, Scherzinger AL, Bergman RN, Haffner SM et al. Insulin sensitivity, body fat distribution, and family diabetes history: the IRAS Family Study. Obes Res 2004; 12: 831–839.
Nyholm B, Nielsen MF, Kristensen K, Nielsen S, Ostergard T, Pedersen SB et al. Evidence of increased visceral obesity and reduced physical fitness in healthy insulin-resistant first-degree relatives of type 2 diabetic patients. Eur J Endocrinol 2004; 150: 207–214.
Lorenzo C, Hartnett S, Hanley AJ, Rewers MJ, Wagenknecht LE, Karter AJ et al. Impaired fasting glucose and impaired glucose tolerance have distinct lipoprotein and apolipoprotein changes: the insulin resistance atherosclerosis study. J Clin Endocrinol Metab 2013; 98: 1622–1630.
Axelsen M, Smith U, Eriksson JW, Taskinen MR, Jansson PA . Postprandial hypertriglyceridemia and insulin resistance in normoglycemic first-degree relatives of patients with type 2 diabetes. Ann Intern Med 1999; 131: 27–31.
Normand-Lauziere F, Frisch F, Labbe SM, Bherer P, Gagnon R, Cunnane SC et al. Increased postprandial nonesterified fatty acid appearance and oxidation in type 2 diabetes is not fully established in offspring of diabetic subjects. PloS One 2010; 5: e10956.
Anthanont P, Jensen MD . Does basal metabolic rate predict weight gain? Am J Clin Nutr 2016; 104: 959–963.
Jensen MD, Kanaley JA, Reed JE, Sheedy PF . Measurement of abdominal and visceral fat with computed tomography and dual-energy x-ray absorptiometry. Am J Clin Nutr 1995; 61: 274–278.
Tchoukalova YD, Koutsari C, Karpyak MV, Votruba SB, Wendland E, Jensen MD . Subcutaneous adipocyte size and body fat distribution. Am J Clin Nutr 2008; 87: 56–63.
Palmer BF, Clegg DJ . The sexual dimorphism of obesity. Mol Cell Endocrinol 2015; 402: 113–119.
Snijder MB, Dekker JM, Visser M, Bouter LM, Stehouwer CD, Yudkin JS et al. Trunk fat and leg fat have independent and opposite associations with fasting and postload glucose levels: the Hoorn study. Diabetes Care 2004; 27: 372–377.
Despres JP, Nadeau A, Tremblay A, Ferland M, Moorjani S, Lupien PJ et al. Role of deep abdominal fat in the association between regional adipose tissue distribution and glucose tolerance in obese women. Diabetes 1989; 38: 304–309.
Pouliot M, Despres JP, Nadeau A, Moorjani S, Prud'Homme D, Lupien PJ et al. Visceral obesity in men. Associations with glucose tolerance, plasma insulin, and lipoprotein levels. Diabetes 1992; 41: 826–834.
Boyko EJ, Fujimoto WY, Leonetti DL, Newell-Morris L . Visceral adiposity and risk of type 2 diabetes: a prospective study among Japanese Americans. Diabetes Care 2000; 23: 465–471.
Eriksson J, Franssila-Kallunki A, Ekstrand A, Saloranta C, Widen E, Schalin C et al. Early metabolic defects in persons at increased risk for non-insulin-dependent diabetes mellitus. N Engl J Med 1989; 321: 337–343.
Perseghin G, Ghosh S, Gerow K, Shulman GI . Metabolic defects in lean nondiabetic offspring of NIDDM parents: a cross-sectional study. Diabetes 1997; 46: 1001–1009.
Stewart MW, Humphriss DB, Mitcheson J, Webster J, Walker M, Laker MF . Lipoprotein composition and serum apolipoproteins in normoglycaemic first-degree relatives of non-insulin dependent diabetic patients. Atherosclerosis 1998; 139: 115–121.
Laws A, Stefanick ML, Reaven GM . Insulin resistance and hypertriglyceridemia in nondiabetic relatives of patients with noninsulin-dependent diabetes mellitus. J Clin Endocrinol Metab 1989; 69: 343–347.
Lotta LA, Gulati P, Day FR, Payne F, Ongen H, van de Bunt M et al. Integrative genomic analysis implicates limited peripheral adipose storage capacity in the pathogenesis of human insulin resistance. Nat Genet 2017; 49: 17–26.
Acknowledgements
PA, KCH, PR and MDJ designed the research. PA, PR and KCH performed the research. PA and KH analyzed the data and wrote the paper. Drs Michael Jensen and Kazanna Hames are the guarantors of this work and, as such, had full access to all of the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis. This work was supported by grant NCRR UL1 TR000135, National Institutes of Health grants, DK-45343, DK-40484, DK-50456 and BIRCWH K12HD065987. Dr Anthanont was a postdoctoral research fellow supported by Thammasat University, Thailand.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Anthanont, P., Ramos, P., Jensen, M. et al. Family history of type 2 diabetes, abdominal adipocyte size and markers of the metabolic syndrome. Int J Obes 41, 1621–1626 (2017). https://doi.org/10.1038/ijo.2017.171
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ijo.2017.171