Abstract
Background:
The association between the development of obesity and its metabolic comorbidities, and chronic consumption of high-fat diet (HFD) has been well-demonstrated. Interestingly, emerging evidence indicates that obesity is also associated with an increased risk for psychiatric disorders including anxiety and depression. Although HFD feeding is associated with anxiety-related behaviors, previous studies have reported inconsistent findings on the direction of this relationship. Therefore, in this study we sought to investigate the link between HFD feeding, body weight, energy states and anxiety levels in mice and specifically to determine if the duration of HFD exposure has distinct effects on anxiety-related behaviors.
Methods:
To disentangle the temporal dynamic effects of HFD feeding on anxiety-related behaviors, mice were fed a HFD or regular chow (RC) diet and were assayed periodically for anxiety-related behaviors by using behavioral tests (open field test; OFT) and the elevated plus maze. To determine if obesity phenotypes correlate with anxiety-related behaviors, changes in anxiety-related behaviors in OFTs were correlated with changes in both body weight and glucose sensitivity following various levels of HFD and RC exposure.
Results:
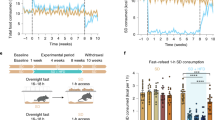
Our results demonstrate a time-dependent biphasic effect of HFD feeding on anxiety-related behaviors. At 5 weeks, mice fed HFD show a reduction in anxiety-related behaviors when compared to pair-fed RC mice. At 8 weeks of HFD or RC feeding, anxiety levels were the same in both groups. Following 15 weeks of HFD and RC feeding, however, mice displaying metabolic symptoms of obesity showed increased anxiety-related behaviors relative to mice resilient to obesity phenotypes, independent of feeding conditions.
Conclusions:
Taken together our findings suggest that HFD bi-directionally effects anxiety-related behaviors such that short-term exposure to a HFD reduces anxiety levels, while longer exposure to a HFD promotes anxiety levels selectively in mice that display metabolic symptoms of obesity. Regardless of diet (HFD or RC), heavier animals display increased anxiety-like behaviors. These findings indicate diverse overlapping roles for HFD and body weight in modulating anxiety-related behaviors, and may partly resolve previous inconsistencies in studies examining the relationship between HFD feeding and anxiety.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Phillips LK, Prins JB . The link between abdominal obesity and the metabolic syndrome. Curr Hypertens Rep 2008; 10: 156–164.
Seidell JC, Halberstadt J . Obesity: the obesity epidemic in the USA — no end in sight? Nat Rev Endocrinol 2016; 12: 499–500.
Faith MS, Matz PE, Jorge MA . Obesity-depression associations in the population. J Psychosom Res 2002; 53: 935–942.
Stunkard AJ, Faith MS, Allison KC . Depression and obesity. Biol Psychiatry 2003; 54: 330–337.
Heo M, Pietrobelli A, Fontaine KR, Sirey JA, Faith MS . Depressive mood and obesity in US adults: comparison and moderation by sex, age, and race. Int J Obes 2006; 30: 513–519.
Simon GE, Von Korff M, Saunders K, Miglioretti DL, Crane PK, van Belle G et al. Association between obesity and psychiatric disorders in the US adult population. Arch Gen Psychiatry 2006; 63: 824.
Johnston E, Johnson S, McLeod P, Johnston M . The relation of body mass index to depressive symptoms. Can J Public Health 2004; 95: 179–183.
Peterhänsel C, Petroff D, Klinitzke G, Kersting A, Wagner B . Risk of completed suicide after bariatric surgery: a systematic review. Obes Rev 2013; 14: 369–382.
Stunkard AJ, Rush J . Dieting and depression reexamined. A critical review of reports of untoward responses during weight reduction for obesity. Ann Intern Med 1974; 81: 526–533.
Teff KL, Kim SF . Atypical antipsychotics and the neural regulation of food intake and peripheral metabolism. Physiol Behav 2011; 104: 590–598.
Schwartz GJ . A satiating signal. Science 2016; 351: 1268–1269.
Hariri N, Thibault L . High-fat diet-induced obesity in animal models. Nutr Res Rev 2010; 23: 270–299.
Maniam J, Antoniadis CP, Le V, Morris MJ . A diet high in fat and sugar reverses anxiety-like behaviour induced by limited nesting in male rats: impacts on hippocampal markers. Psychoneuroendocrinology 2016; 68: 202–209.
Ohland CL, Pankiv E, Baker G, Madsen KL . Western diet-induced anxiolytic effects in mice are associated with alterations in tryptophan metabolism. Nutr Neurosci 2016; 19: 337–345.
McNeilly AD, Stewart CA, Sutherland C, Balfour DJK . High fat feeding is associated with stimulation of the hypothalamic-pituitary-adrenal axis and reduced anxiety in the rat. Psychoneuroendocrinology 2015; 52: 272–280.
Prasad A, Prasad C . Short-term consumption of a diet rich in fat decreases anxiety response in adult male rats. Physiol Behav 1996; 60: 1039–1042.
de Noronha SR, Campos GV, Abreu AR, de Souza AA, Chianca DA, de Menezes RC . High fat diet induced-obesity facilitates anxiety-like behaviors due to GABAergic impairment within the dorsomedial hypothalamus in rats. Behav Brain Res 2017; 316: 38–46.
Gainey SJ, Kwakwa KA, Bray JK, Pillote MM, Tir VL, Towers AE et al. Short-term high-fat diet (HFD) induced anxiety-like behaviors and cognitive impairment are improved with treatment by glyburide. Front Behav Neurosci 2016; 10: 156.
Dutheil S, Ota KT, Wohleb ES, Rasmussen K, Duman RS . High-fat diet induced anxiety and anhedonia: impact on brain homeostasis and inflammation. Neuropsychopharmacology 2016; 41: 1874–1887.
Sivanathan S, Thavartnam K, Arif S, Elegino T, McGowan PO . Chronic high fat feeding increases anxiety-like behaviour and reduces transcript abundance of glucocorticoid signalling genes in the hippocampus of female rats. Behav Brain Res 2015; 286: 265–270.
Sweeney P, Yang Y . An inhibitory septum to lateral hypothalamus circuit that suppresses feeding. J Neurosci 2016; 36: 11185–11195.
Sweeney P, Yang Y . An excitatory ventral hippocampus to lateral septum circuit that suppresses feeding. Nat Commun 2015; 6: 10188.
Murray S, Tulloch A, Gold MS, Avena NM . Hormonal and neural mechanisms of food reward, eating behaviour and obesity. Nat Rev Endocrinol 2014; 10: 540–552.
Schwartz GJ, Zeltser LM . Functional organization of neuronal and humoral signals regulating feeding behavior. Annu Rev Nutr 2013; 33: 1–21.
Williams KW, Elmquist JK . From neuroanatomy to behavior: central integration of peripheral signals regulating feeding behavior. Nat Neurosci 2012; 15: 1350–1355.
Pang ZP, Han W . Regulation of synaptic functions in central nervous system by endocrine hormones and the maintenance of energy homoeostasis. Biosci Rep 2012; 32: 423–432.
Lim BK, Huang KW, Grueter BA, Rothwell PE, Malenka RC . Anhedonia requires MC4R-mediated synaptic adaptations in nucleus accumbens. Nature 2012; 487: 183–189.
Domingos AI, Vaynshteyn J, Voss HU, Ren X, Gradinaru V, Zang F et al. Leptin regulates the reward value of nutrient. Nat Neurosci 2011; 14: 1562–1568.
Andrews ZB . The extra-hypothalamic actions of ghrelin on neuronal function. Trends Neurosci 2011; 34: 31–40.
Beck B, Pourié G . Ghrelin, neuropeptide Y, and other feeding-regulatory peptides active in the hippocampus: role in learning and memory. Nutr Rev 2013; 71: 541–561.
Gautron L, Elmquist JK, Williams KW . Neural control of energy balance: translating circuits to therapies. Cell 2015; 161: 133–145.
Hardaway JA, Crowley NA, Bulik CM, Kash TL . Integrated circuits and molecular components for stress and feeding: implications for eating disorders. Genes Brain Behav 2015; 14: 85–97.
Maniam J, Morris MJ . The link between stress and feeding behaviour. Neuropharmacology 2012; 63: 97–110.
Morris MJ, Beilharz JE, Maniam J, Reichelt AC, Westbrook RF . Why is obesity such a problem in the 21st century? The intersection of palatable food, cues and reward pathways, stress, and cognition. Neurosci Biobehav Rev 2015; 58: 36–45.
Davidson TL, Sample CH, Swithers SE . An application of Pavlovian principles to the problems of obesity and cognitive decline. Neurobiol Learn Mem 2014; 108: 172–184.
Võikar V, Kõks S, Vasar E, Rauvala H . Strain and gender differences in the behavior of mouse lines commonly used in transgenic studies. Physiol Behav 2001; 72: 271–281.
Kokras N, Dalla C . Sex differences in animal models of psychiatric disorders. Br J Pharmacol 2014; 171: 4595–4619.
Tucker LB, McCabe JT . Behavior of male and female C57BL/6 J mice is more consistent with repeated trials in the elevated zero maze than in the elevated plus maze. Front Behav Neurosci 2017; 11: 13.
Johnston AL, File SE . Sex differences in animal tests of anxiety. Physiol Behav 1991; 49: 245–250.
Russo SJ, Murrough JW, Han M-H, Charney DS, Nestler EJ . Neurobiology of resilience. Nat Neurosci 2012; 15: 1475–1484.
Acknowledgements
We thank all the members of Yang laboratory for discussion and critical comments on this project. We also thank R. Quinn for mouse husbandry. This research was supported by the State University of New York and NIH (R01 MH109441A to YY). YY conceived the idea. PS, KO and YY designed this study. PS, KO, and ZX performed the experiments and analyzed the data. PS, KO and YY wrote the paper with the inputs from other authors.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on International Journal of Obesity website
Supplementary information
Rights and permissions
About this article
Cite this article
Sweeney, P., O'Hara, K., Xu, Z. et al. HFD-induced energy states-dependent bidirectional control of anxiety levels in mice. Int J Obes 41, 1237–1245 (2017). https://doi.org/10.1038/ijo.2017.112
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ijo.2017.112
This article is cited by
-
Hepatic glycogen participates in the regulation of hypothalamic pAkt/Akt ratio in high-sugar/high-fat diet-induced obesity
Metabolic Brain Disease (2022)
-
Lactobacillus delbrueckii subsp. bulgaricus strain TCI904 reduces body weight gain, modulates immune response, improves metabolism and anxiety in high fat diet-induced obese mice
3 Biotech (2022)
-
Palatable high-fat diet intake influences mnemonic and emotional aspects in female rats in an estrous cycle-dependent manner
Metabolic Brain Disease (2021)
-
Identification of a neurocircuit underlying regulation of feeding by stress-related emotional responses
Nature Communications (2019)
-
AGRP neurons modulate fasting-induced anxiolytic effects
Translational Psychiatry (2019)