Abstract
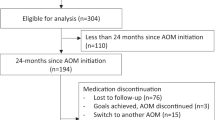
Phentermine is the most widely prescribed obesity medication in adults, yet studies of its use in the pediatric population are limited. We conducted a retrospective chart review of adolescents with obesity treated in a pediatric weight management clinic to examine the weight loss effectiveness of phentermine added to standard of care (SOC) lifestyle modification therapy versus SOC alone. All patients receiving phentermine plus SOC (n=25) were matched with a comparison group receiving only SOC (n=274). Differences at 1, 3 and 6 months were evaluated using generalized estimated equations adjusting for age, sex and baseline body mass index (BMI) and robust variance standard error estimates for confidence intervals and P-values. Phentermine use was associated with a greater percent change in BMI at 1 month (−1.6%; 95% confidence interval (CI): −2.6, −0.6%; P=0.001), 3 months (−2.9%; 95% CI: −4.5, −1.4%; P<0.001) and 6 months (−4.1%; 95% CI: −7.1, −1.0%; P=0.009) compared with SOC alone, with no differences in systolic or diastolic blood pressure between groups. Heart rate was higher at all time-points in the phentermine plus SOC compared with SOC-only group. These data suggest that short-term use of phentermine added to SOC may enhance weight loss in adolescents with obesity in the clinical setting.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Kelly AS, Fox CK, Rudser KD, Gross AC, Ryder JR . Pediatric obesity pharmacotherapy: Current state of the field, review of the literature, and clinical trial considerations. Int J Obes (Lond) 2016; 40: 1043–1050.
Barlow SE . Expert Committee Recommendations Regarding the Prevention, Assessment, and Treatment of Child and Adolescent Overweight and Obesity: Summary Report. Pediatrics 2007; 120: S164–S192.
Sherafat-Kazemzadeh R, Yanovski SZ, Yanovski JA . Pharmacotherapy for childhood obesity: present and future prospects. Int J Obes (Lond) 2013; 37: 1–15.
Hendricks EJ, Rothman RB, Greenway FL . How physician obesity specialists use drugs to treat obesity. Obesity (Silver Spring) 2009; 17: 1730–1735.
Stafford RS, Radley DC . National trends in antiobesity medication use. Arch Intern Med 2003; 163: 1046–1050.
Lorber J . Obesity in childhood. A controlled trial of anorectic drugs. Arch Dis Childhood 1966; 41: 309–312.
Rauh JL, Lipp R . Chlorphentermine as an anorexigenic agent in adolescent obesity. Report of its efficacy in a double-blind study of 30 teenagers. Clin Pediatr 1968; 7: 138–140.
Team RCR: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing, 2015.
Kang JG, Park CY, Kang JH, Park YW, Park SW . Randomized controlled trial to investigate the effects of a newly developed formulation of phentermine diffuse-controlled release for obesity. Diabetes Obes Metab 2010; 12: 876–882.
Hendricks EJ, Greenway FL, Westman EC, Gupta AK . Blood pressure and heart rate effects, weight loss and maintenance during long-term phentermine pharmacotherapy for obesity. Obesity (Silver Spring) 2011; 19: 2351–2360.
Kelly AS, Rudser KD, Nathan BM, Fox CK, Metzig AM, Coombes BJ et al. The effect of glucagon-like peptide-1 receptor agonist therapy on body mass index in adolescents with severe obesity: a randomized, placebo-controlled, clinical trial. JAMA Pediatr 2013; 167: 355–360.
Kelly AS, Metzig AM, Rudser KD, Fitch AK, Fox CK, Nathan BM et al. Exenatide as a weight-loss therapy in extreme pediatric obesity: a randomized, controlled pilot study. Obesity (Silver Spring) 2012; 20: 364–370.
Yanovski JA, Krakoff J, Salaita CG, McDuffie JR, Kozlosky M, Sebring NG et al. Effects of metformin on body weight and body composition in obese insulin-resistant children: a randomized clinical trial. Diabetes 2011; 60: 477–485.
Chanoine JP, Hampl S, Jensen C, Boldrin M, Hauptman J . Effect of orlistat on weight and body composition in obese adolescents: a randomized controlled trial. JAMA 2005; 293: 2873–2883.
Wilson DM, Abrams SH, Aye T, Lee PD, Lenders C, Lustig RH et al. Metformin extended release treatment of adolescent obesity: a 48-week randomized, double-blind, placebo-controlled trial with 48-week follow-up. Arch Pediatr Adolesc Med 2010; 164: 116–123.
Antelmi I, de Paula RS, Shinzato AR, Peres CA, Mansur AJ, Grupi CJ . Influence of age, gender, body mass index, and functional capacity on heart rate variability in a cohort of subjects without heart disease. Am J Cardiol 2004; 93: 381–385.
Hendricks EJ, Srisurapanont M, Schmidt SL, Haggard M, Souter S, Mitchell CL et al. Addiction potential of phentermine prescribed during long-term treatment of obesity. Int J Obes (Lond) 2014; 38: 292–298.
Acknowledgements
Dr Ryder is supported by an individual training grant from NIH/NHLBI (F32-HL127851). Dr Rudser and Kaizer are supported, in part, by the National Center for Advancing Translational Sciences/NIH (UL1TR000114). The other authors received no funding for this project.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Dr Kelly serves as a consultant for Novo Nordisk Pharmaceuticals, but does not receive personal or professional income for these activities. Dr Kelly receives research support in the form of drug/placebo from Astra Zeneca Pharmaceuticals. Dr Fox serves as a site principal investigator for a clinical trial sponsored by Novo Nordisk Pharmaceuticals. The other authors declare no conflict of interest. No honorarium, grant or other form of payment was given to anyone to produce the manuscript.
Additional information
Supplementary Information accompanies this paper on International Journal of Obesity website
Supplementary information
Rights and permissions
About this article
Cite this article
Ryder, J., Kaizer, A., Rudser, K. et al. Effect of phentermine on weight reduction in a pediatric weight management clinic. Int J Obes 41, 90–93 (2017). https://doi.org/10.1038/ijo.2016.185
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ijo.2016.185
This article is cited by
-
Current and future state of pharmacological management of pediatric obesity
International Journal of Obesity (2024)
-
Pharmacological Interventions of Atypical Antipsychotics Induced Weight Gain in the Pediatric Population: A Systemic Review of Current Evidence
Child Psychiatry & Human Development (2024)
-
Updates in the Treatment of Pediatric Obesity
Current Treatment Options in Pediatrics (2023)
-
The treatment of obesity in children and adolescents: consensus position statement of the Italian society of pediatric endocrinology and diabetology, Italian Society of Pediatrics and Italian Society of Pediatric Surgery
Italian Journal of Pediatrics (2023)
-
The adolescent with obesity: what perspectives for treatment?
Italian Journal of Pediatrics (2022)