Abstract
Background/Objectives:
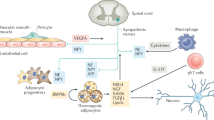
The identification of brown/beige adipose tissue in adult humans has motivated the search for methods aimed at increasing its thermogenic activity as an approach to treat obesity. In rodents, the brown adipose tissue is under the control of sympathetic signals originating in the hypothalamus. However, the putative connection between the depots of brown/beige adipocytes and the hypothalamus in humans has never been explored. The objective of this study was to evaluate the response of the hypothalamus and brown/beige adipose tissue to cold stimulus in obese subjects undergoing body mass reduction following gastric bypass.
Subjects/Methods:
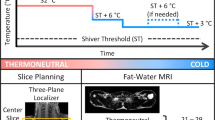
We evaluated twelve obese, non-diabetic subjects undergoing Roux-in-Y gastric bypass and 12 lean controls. Obese subjects were evaluated before and approximately 8 months after gastric bypass. Lean subjects were evaluated only at admission. Subjects were evaluated for hypothalamic activity in response to cold by functional magnetic resonance, whereas brown/beige adipose tissue activity was evaluated using a (F 18) fluorodeoxyglucose positron emisson tomography/computed tomography scan and real-time PCR measurement of signature genes.
Results:
Body mass reduction resulted in a significant increase in brown/beige adipose tissue activity in response to cold; however, no change in cold-induced hypothalamic activity was observed after body mass reduction. No correlation was found between brown/beige adipose tissue activation and hypothalamus activity in obese subjects or in lean controls.
Conclusions:
In humans, the increase in brown/beige adipose tissue activity related to body mass reduction occurs independently of changes in hypothalamic activity as determined by functional magnetic resonance.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Morton GJ, Cummings DE, Baskin DG, Barsh GS, Schwartz MW . Central nervous system control of food intake and body weight. Nature 2006; 443: 289–295.
Zeltser LM, Seeley RJ, Tschop MH . Synaptic plasticity in neuronal circuits regulating energy balance. Nat Neurosci 2012; 15: 1336–1342.
Dietrich MO, Horvath TL . Hypothalamic control of energy balance: insights into the role of synaptic plasticity. Trends Neurosci 2013; 36: 65–73.
Velloso LA, Schwartz MW . Altered hypothalamic function in diet-induced obesity. Int J Obes (Lond) 2011; 35: 1455–1465.
Thaler JP, Guyenet SJ, Dorfman MD, Wisse BE, Schwartz MW . Hypothalamic inflammation: marker or mechanism of obesity pathogenesis? Diabetes 2013; 62: 2629–2634.
Cai D . Neuroinflammation and neurodegeneration in overnutrition-induced diseases. Trends Endocrinol Metab 2013; 24: 40–47.
Cannon B, Nedergaard J . Metabolic consequences of the presence or absence of the thermogenic capacity of brown adipose tissue in mice (and probably in humans). Int J Obes 2010; 34: S7–S16.
Romanatto T, Roman EA, Arruda AP, Denis RG, Solon C, Milanski M et al. Deletion of tumor necrosis factor-alpha receptor 1 (TNFR1) protects against diet-induced obesity by means of increased thermogenesis. J Biol Chem 2009; 284: 36213–36222.
Arruda AP, Milanski M, Velloso LA . Hypothalamic inflammation and thermogenesis: the brown adipose tissue connection. J Bioenerg Biomembr 2011; 43: 53–58.
Hamann A, Flier JS, Lowell BB . Decreased brown fat markedly enhances susceptibility to diet-induced obesity, diabetes, and hyperlipidemia. Endocrinology 1996; 137: 21–29.
Kong D, Tong Q, Ye C, Koda S, Fuller PM, Krashes MJ et al. GABAergic RIP-Cre neurons in the arcuate nucleus selectively regulate energy expenditure. Cell (Lond) 2012; 151: 645–657.
van Marken Lichtenbelt WD, Vanhommerig JW, Smulders NM, Drossaerts JM, Kemerink GJ, Bouvy ND et al. Cold-activated brown adipose tissue in healthy men. N Engl J Med 2009; 360: 1500–1508.
Cypess AM, Lehman S, Williams G, Tal I, Rodman D, Goldfine AB et al. Identification and importance of brown adipose tissue in adult humans. N Engl J Med 2009; 360: 1509–1517.
Virtanen KA, Lidell ME, Orava J, Heglind M, Westergren R, Niemi T et al. Functional brown adipose tissue in healthy adults. N Engl J Med 2009; 360: 1518–1525.
Wu J, Bostrom P, Sparks LM, Ye L, Choi JH, Giang AH et al. Beige adipocytes are a distinct type of thermogenic fat cell in mouse and human. Cell 2012; 150: 366–376.
Jespersen NZ, Larsen TJ, Peijs L, Daugaard S, Homoe P, Loft A et al. A classical brown adipose tissue mRNA signature partly overlaps with brite in the supraclavicular region of adult humans. Cell Metab 2013; 17: 798–805.
Almind K, Manieri M, Sivitz WI, Cinti S, Kahn CR . Ectopic brown adipose tissue in muscle provides a mechanism for differences in risk of metabolic syndrome in mice. Proc Natl Acad Sci USA 2007; 104: 2366–2371.
Cypess AM, Kahn CR . Brown fat as a therapy for obesity and diabetes. Curr Opin Endocrinol Diabetes Obes 2010; 17: 143–149.
Lidell ME, Enerback S . Brown adipose tissue—a new role in humans? Nat Rev Endocrinol 2010; 6: 319–325.
Nakamura K, Morrison SF . Central efferent pathways mediating skin cooling-evoked sympathetic thermogenesis in brown adipose tissue. Am J Physiol Regul Integr Comp Physiol 2007; 292: R127–R136.
Zhang Y, Kerman IA, Laque A, Nguyen P, Faouzi M, Louis GW et al. Leptin-receptor-expressing neurons in the dorsomedial hypothalamus and median preoptic area regulate sympathetic brown adipose tissue circuits. J Neurosci 2011; 31: 1873–1884.
Bartness TJ, Vaughan CH, Song CK . Sympathetic and sensory innervation of brown adipose tissue. Int J Obes (Lond) 2010; 34: S36–S42.
Carey AL, Formosa MF, Van Every B, Bertovic D, Eikelis N, Lambert GW et al. Ephedrine activates brown adipose tissue in lean but not obese humans. Diabetologia 2013; 56: 147–155.
Vosselman MJ, van der Lans AA, Brans B, Wierts R, van Baak MA, Schrauwen P et al. Systemic beta-adrenergic stimulation of thermogenesis is not accompanied by brown adipose tissue activity in humans. Diabetes 2012; 61: 3106–3113.
Cypess AM, Chen YC, Sze C, Wang K, English J, Chan O et al. Cold but not sympathomimetics activates human brown adipose tissue in vivo. Proc Natl Acad Sci USA 2012; 109: 10001–10005.
American Diabetes Association. Standards of medical care in diabetes–2011. Diabetes Care 2011; 34: S11–S61.
Gastrointestinal surgery for severe obesity: National Institutes of Health Consensus Development Conference Statement Am J Clin Nutr 1992; 55: 615S–619S.
de Carvalho CP, Marin DM, de Souza AL, Pareja JC, Chaim EA, de Barros Mazon S et al. GLP-1 and adiponectin: effect of weight loss after dietary restriction and gastric bypass in morbidly obese patients with normal and abnormal glucose metabolism. Obes Surg 2009; 19: 313–320.
Milanski M, Arruda AP, Coope A, Ignacio-Souza LM, Nunez CE, Roman EA et al. Inhibition of hypothalamic inflammation reverses diet-induced insulin resistance in the liver. Diabetes 2012; 61: 1455–1462.
Bell AW, Gardner JW, Manson W, Thompson GE . Acute cold exposure and the metabolism of blood glucose, lactate and pyruvate, and plasma amino acids in the hind leg of the fed and fasted young ox. Br J Nutr 1975; 33: 207–217.
Vallerand AL, Frim J, Kavanagh MF . Plasma glucose and insulin responses to oral and intravenous glucose in cold-exposed humans. J Appl Physiol 1988; 65: 2395–2399.
Gasparetti AL, de Souza CT, Pereira-da-Silva M, Oliveira RL, Saad MJ, Carneiro EM et al. Cold exposure induces tissue-specific modulation of the insulin-signalling pathway in Rattus norvegicus. J Physiol 2003; 552: 149–162.
Ouellet V, Labbe SM, Blondin DP, Phoenix S, Guerin B, Haman F et al. Brown adipose tissue oxidative metabolism contributes to energy expenditure during acute cold exposure in humans. J Clin Invest 2012; 122: 545–552.
Harms M, Seale P . Brown and beige fat: development, function and therapeutic potential. Nat Med 2013; 19: 1252–1263.
Parysow O, Mollerach AM, Jager V, Racioppi S, San Roman J, Gerbaudo VH . Low-dose oral propranolol could reduce brown adipose tissue F-18 FDG uptake in patients undergoing PET scans. Clin Nucl Med 2007; 32: 351–357.
Soderlund V, Larsson SA, Jacobsson H . Reduction of FDG uptake in brown adipose tissue in clinical patients by a single dose of propranolol. Eur J Nucl Med Mol Imaging 2007; 34: 1018–1022.
De Araujo IE, Rolls ET . Representation in the human brain of food texture and oral fat. J Neurosci 2004; 24: 3086–3093.
Liu Y, Gao JH, Liu HL, Fox PT . The temporal response of the brain after eating revealed by functional MRI. Nature 2000; 405: 1058–1062.
van de Sande-Lee S, Pereira FR, Cintra DE, Fernandes PT, Cardoso AR, Garlipp CR et al. Partial reversibility of hypothalamic dysfunction and changes in brain activity after body mass reduction in obese subjects. Diabetes 2011; 60: 1699–1704.
Thaler JP, Yi CX, Schur EA, Guyenet SJ, Hwang BH, Dietrich MO et al. Obesity is associated with hypothalamic injury in rodents and humans. J Clin Invest 2012; 122: 153–162.
McKitrick DJ . Expression of fos in the hypothalamus of rats exposed to warm and cold temperatures. Brain Res Bull 2000; 53: 307–315.
Vijgen GH, Bouvy ND, Teule GJ, Brans B, Hoeks J, Schrauwen P et al. Increase in brown adipose tissue activity after weight loss in morbidly obese subjects. J Clin Endocrinol Metab 2012; 97: E1229–E1233.
Lidell ME, Betz MJ, Enerback S . Two types of brown adipose tissue in humans. Adipocyte 2014; 3: 63–66.
Orava J, Nummenmaa L, Noponen T, Viljanen T, Parkkola R, Nuutila P et al. Brown adipose tissue function is accompanied by cerebral activation in lean but not in obese humans. J Cereb Blood Flow Metab 2014; 34: 1018–1023.
Acknowledgements
We thank E. Roman and G. Ferraz from the University of Campinas for technical assistance. Support for the study was provided by the Fundação de Amparo a Pesquisa do Estado de São Paulo, Conselho Nacional de Desenvolvimento Cientifico e Tecnologico and grants from the Trust in Science Initiative from Glaxo-Smithkline, UK. The Laboratories of Cell Signaling and Experimental Endocrinology belong to the Obesity and Comorbidities Research Center and the National Institute of Science and Technology–Diabetes and Obesity.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on International Journal of Obesity website
Supplementary information
Rights and permissions
About this article
Cite this article
Rachid, B., van de Sande-Lee, S., Rodovalho, S. et al. Distinct regulation of hypothalamic and brown/beige adipose tissue activities in human obesity. Int J Obes 39, 1515–1522 (2015). https://doi.org/10.1038/ijo.2015.94
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ijo.2015.94
This article is cited by
-
Hypothalamic control of energy expenditure and thermogenesis
Experimental & Molecular Medicine (2022)
-
Caloric restriction and Roux-en-Y Gastric Bypass promote white adipose tissue browning in mice
Journal of Endocrinological Investigation (2022)
-
The Role of Positron Emission Tomography in Bariatric Surgery Research: a Review
Obesity Surgery (2021)
-
Radiologic evidence that hypothalamic gliosis is improved after bariatric surgery in obese women with type 2 diabetes
International Journal of Obesity (2020)
-
Elevated Levels of Circulating miR-92a Are Associated with Impaired Glucose Homeostasis in Patients with Obesity and Correlate with Metabolic Status After Bariatric Surgery
Obesity Surgery (2020)