Abstract
Background:
Cellular glucose uptake can be enhanced by upregulating Ras signaling in either insulin-dependent or -independent manner. In presence of insulin and intact insulin signaling, Ras has a negligible role in glucose uptake. Conversely, when insulin signaling is impaired in obesity or diabetes, the insulin-independent Ras pathway may be valuable for enhancing glucose disposal. We previously reported that Ad36, a human adenovirus, enhances cellular glucose uptake by upregulating the Ras/Glut4 pathway. Here, we investigated if Ad36-upregulated Ras via the insulin-independent pathway, to enhance glucose uptake. Furthermore, uncontrolled upregulation of Ras is linked with oncogenic cell transformation, if the tumor-suppressor gene p53 is also downregulated. Hence, we determined if upregulation of Ras by Ad36 would induce oncogenic cell transformation. Finally, we determined the relevance of Ad36 to insulin resistance in humans.
Methods:
Insulin receptor (IR) was knocked down with small interfering RNA in 3T3-L1 adipocytes, to determine if Ad36 increases the Ras/Glut4 pathway and glucose uptake without IR-signaling. Next, the effects of Ad36 on cell transformation and p53 abundance were determined. Finally, overweight or obese women were screened for seropositivity to Ad36, as an indicator of natural Ad36 infection. Associations of Ad36 infection with adiposity and C-reactive proteins (CRPs)—two key markers of insulin resistance, and with glucose disposal, were determined.
Results:
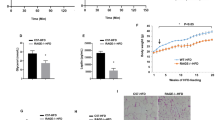
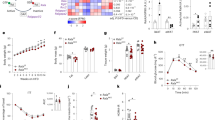
Unaffected by IR knock-down, Ad36 significantly increased the Ras pathway, Glut4 translocation and glucose uptake in 3T3-L1 adipocytes. Despite Ras upregulation, Ad36 did not transform 3T3-L1 cells. This may be because Ad36 significantly increased p53 protein in 3T3-L1 cells or mice adipose tissue. Ad36 seropositivity was associated with greater adiposity and CRP levels, yet a significantly higher systemic glucose disposal rate.
Conclusions:
Overall, the study offers Ras/Glut4 pathway as an alternate to enhance glucose disposal when insulin signaling is impaired, and, importantly, provides Ad36 as a tool to understand the modulation of that pathway.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Jiang ZY, Zhou QL, Coleman KA, Chouinard M, Boese Q, Czech MP . Insulin signaling through Akt/protein kinase B analyzed by small interfering RNA-mediated gene silencing. Proc Natl Acad Sci USA 2003; 100: 7569–7574.
Goodyear LJ, Giorgino F, Sherman LA, Carey J, Smith RJ, Dohm GL . Insulin receptor phosphorylation, insulin receptor substrate-1 phosphorylation, and phosphatidylinositol 3-kinase activity are decreased in intact skeletal muscle strips from obese subjects. J Clin Invest 1995; 95: 2195–2204.
Pessin JE, Saltiel AR . Signaling pathways in insulin action: molecular targets of insulin resistance. J Clin Invest 2000; 106: 165–169.
Sesti G, Federici M, Hribal ML, Lauro D, Sbraccia P, Lauro R . Defects of the insulin receptor substrate (IRS) system in human metabolic disorders. FASEB J 2001; 15: 2099–2111.
Klip A . The many ways to regulate glucose transporter 4. Appl Physiol Nutr Metab 2009; 34: 481–487.
Tanaka T, Nakatani K, Morioka K, Urakawa H, Maruyama N, Kitagawa N et al. Nitric oxide stimulates glucose transport through insulin-independent GLUT4 translocation in 3T3-L1 adipocytes. Eur J Endocrinol 2003; 149: 61–67.
Perdomo G, Martinez-Brocca MA, Bhatt BA, Brown NF, O’Doherty RM, Garcia-Ocana A . Hepatocyte growth factor is a novel stimulator of glucose uptake and metabolism in skeletal muscle cells. J Biol Chem 2008; 283: 13700–13706.
Frevert EU, Kahn BB . Differential effects of constitutively active phosphatidylinositol 3-kinase on glucose transport, glycogen synthase activity, and DNA synthesis in 3T3-L1 adipocytes. Mol Cell Biol 1997; 17: 190–198.
Krishnapuram R, Dhurandhar EJ, Dubuisson O, Kirk-Ballard H, Bajpeyi S, Butte N et al. Template to improve glycemic control without reducing adiposity or dietary fat. Am J Physiol Endocrinol Metab 2011; 300: E779–E789.
Rogers PM, Mashtalir N, Rathod MA, Dubuisson O, Wang ZQ, Dasuri K et al. Metabolically favorable remodeling of human adipose tissue by human adenovirus Ad-36. Diabetes 2008; 57: 2321–2331.
Wang ZQ, Cefalu WT, Zhang XH, Yongmei Y, Qin J, Son L et al. Human adenovirus type 36 enhances glucose uptake in diabetic and non-diabetic human skeletal muscle cells independent of insulin signaling. Diabetes 2008; 57: 1805–1813.
Pasarica M, Shin AC, Yu M, Ou Yang HM, Rathod M, Jen KL et al. Human adenovirus 36 induces adiposity, increases insulin sensitivity, and alters hypothalamic monoamines in rats. Obesity (Silver Spring) 2006; 14: 1905–1913.
Jiang S, Gavrikova TA, Pereboev A, Messina JL . Adenovirus infection results in alterations of insulin signaling and glucose homeostasis. Am J Physiol Endocrinol Metab 2010; 298: E1295–E1304.
Shaw RJ, Cantley LC . Ras, PI(3)K and mTOR signalling controls tumour cell growth. Nature 2006; 441: 424–430.
Suire S, Condliffe AM, Ferguson GJ, Ellson CD, Guillou H, Davidson K et al. Gbetagammas and the Ras binding domain of p110gamma are both important regulators of PI(3)Kgamma signalling in neutrophils. Nat Cell Biol 2006; 8: 1303–1309.
Sasaki AT, Chun C, Takeda K, Firtel RA . Localized Ras signaling at the leading edge regulates PI3K, cell polarity, and directional cell movement. J Cell Biol 2004; 167: 505–518.
Benito M, Porras A, Nebreda AR, Santos E . Differentiation of 3T3-L1 fibroblasts to adipocytes induced by transfection of ras oncogenes. Science 1991; 253: 565–568.
Kozma L, Baltensperger K, Klarlund J, Porras A, Santos E, Czech MP . The ras signaling pathway mimics insulin action on glucose transporter translocation. Proc Natl Acad Sci USA 1993; 90: 4460–4464.
Dorrestijn J, Ouwens DM, Van den Berghe N, Bos JL, Maassen JA . Expression of a dominant-negative Ras mutant does not affect stimulation of glucose uptake and glycogen synthesis by insulin. Diabetologia 1996; 39: 558–563.
Houseknecht KL, Zhu AX, Gnudi L, Hamann A, Zierath JR, Tozzo E et al. Overexpression of Ha-ras selectively in adipose tissue of transgenic mice. Evidence for enhanced sensitivity to insulin. J Biol Chem 1996; 271: 11347–11355.
Ceresa BP, Pessin JE . Insulin regulation of the Ras activation/inactivation cycle. Mol Cell Biochem 1998; 182: 23–29.
Finlay CA, Hinds PW, Levine AJ . The p53 proto-oncogene can act as a suppressor of transformation. Cell 1989; 57: 1083–1093.
Rathod M, Rogers PM, Vangipuram SD, McAllister EJ, Dhurandhar NV . Adipogenic cascade can be induced without adipogenic media by a human adenovirus. Obesity 2009; 17: 657–664.
Brochu M, Malita MF, Messier V, Doucet E, Strychar I, Lavoie JM et al. Resistance training does not contribute to improving the metabolic profile after a 6-month weight loss program in overweight and obese postmenopausal women. J Clin Endocrinol Metab 2009; 94: 3226–3233.
Lavoie ME, Rabasa-Lhoret R, Ziai S, Lavoie JC . Blood glutathione peroxidase activity in relation with the risk of cardiovascular diseases. J Diabetes Metab 2011; 2: 2–6.
Atkinson RL, Dhurandhar NV, Allison DB, Bowen RL, Israel BA, Albu JB et al. Human adenovirus-36 is associated with increased body weight and paradoxical reduction of serum lipids. Int J Obes (Lond) 2005; 29: 281–286.
Kilroy G, Burk DH, Floyd ZE . High efficiency lipid-based siRNA transfection of adipocytes in suspension. PLoS One 2009; 4: e6940.
Jialal I, Devaraj S, Venugopal SK . C-reactive protein: risk marker or mediator in atherothrombosis? Hypertension 2004; 44: 6–11.
Visser M, Bouter LM, McQuillan GM, Wener MH, Harris TB . Elevated C-reactive protein levels in overweight and obese adults. JAMA 1999; 282: 2131–2135.
Bluher M, Fasshauer M, Tonjes A, Kratzsch J, Schon MR, Paschke R . Association of interleukin-6, C-reactive protein, interleukin-10 and adiponectin plasma concentrations with measures of obesity, insulin sensitivity and glucose metabolism. Exp Clin Endocrinol Diabetes 2005; 113: 534–537.
Foy HM, Grayston JT . Adenoviruses. In: Evans Alfred S (ed). Viral Infections of Humans: Epidemiology and Control. Plenum Medical: New York, 1976, pp 53–70.
Horvath J, Palkonyay L, Weber J . Group C adenovirus DNA sequences in human lymphoid cells. J Virol 1986; 59: 189–192.
Hierholzer JC, Wigand R, Anderson LJ, Adrian T, Gold JW . Adenoviruses from patients with AIDS: a plethora of serotypes and a description of five new serotypes of subgenus D (types 43-47). J Infect Dis 1988; 158: 804–813.
Wigand R, Gelderblom H, Wadell G . New human adenovirus (candidate adenovirus 36), a novel member of subgroup D. Arch Virol 1980; 64: 225–233.
Atkinson R, Dhurandhar N, Allison D, Bower R, Israel B . Evidence for an association of an obesity virus with human obesity at three sites in the United States. Int J Obes 1998; 22: S57.
Dhurandhar NV, Israel BA, Kolesar JM, Mayhew GF, Cook ME, Atkinson RL . Increased adiposity in animals due to a human virus. Int J Obes Relat Metab Disord 2000; 24: 989–996.
Dhurandhar NV, Israel BA, Kolesar JM, Mayhew G, Cook ME, Atkinson RL . Transmissibility of adenovirus-induced adiposity in a chicken model. Int J Obes Relat Metab Disord 2001; 25: 990–996.
Dhurandhar NV, Whigham LD, Abbott DH, Schultz-Darken NJ, Israel BA, Bradley SM et al. Human adenovirus Ad-36 promotes weight gain in male rhesus and marmoset monkeys. J Nutr 2002; 132: 3155–3160.
Pasarica M, Loiler S, Dhurandhar NV . Acute effect of infection by adipogenic human adenovirus Ad36. Arch Virol 2008; 153: 2097–2102.
Dhurandhar NV . A framework for identification of infections that contribute to human obesity. Lancet Infect Dis 2011; 11: 963–969.
Bos JL . Ras oncogenes in human cancer: a review. Cancer Res 1989; 49: 4682–4689.
Slattery ML, Samowitz W, Curtin K, Ma KN, Hoffman M, Caan B et al. Associations among IRS1, IRS2, IGF1, and IGFBP3 genetic polymorphisms and colorectal cancer. Cancer Epidemiol Biomarkers Prev 2004; 13: 1206–1214.
Bader AG, Kang S, Zhao L, Vogt PK . Oncogenic PI3K deregulates transcription and translation. Nat Rev Cancer 2005; 5: 921–929.
Pylayeva Y, Gillen KM, Gerald W, Beggs HE, Reichardt LF, Giancotti FG . Ras- and PI3K-dependent breast tumorigenesis in mice and humans requires focal adhesion kinase signaling. J Clin Invest 2009; 119: 252–266.
Hanahan D, Weinberg RA . The hallmarks of cancer. Cell 2000; 100: 57–70.
Donehower LA, Harvey M, Slagle BL, McArthur MJ, Montgomery Jr CA, Butel JS et al. Mice deficient for p53 are developmentally normal but susceptible to spontaneous tumours. Nature 1992; 356: 215–221.
Ventura A, Kirsch DG, McLaughlin ME, Tuveson DA, Grimm J, Lintault L et al. Restoration of p53 function leads to tumour regression in vivo. Nature 2007; 445: 661–665.
Boiko AD, Porteous S, Razorenova OV, Krivokrysenko VI, Williams BR, Gudkov AV . A systematic search for downstream mediators of tumor suppressor function of p53 reveals a major role of BTG2 in suppression of Ras-induced transformation. Genes Dev 2006; 20: 236–252.
Mistry T, Digby JE, Desai KM, Randeva HS . Leptin and adiponectin interact in the regulation of prostate cancer cell growth via modulation of p53 and bcl-2 expression. BJU Int 2008; 101: 1317–1322.
Lipscombe LL, Gomes T, Levesque LE, Hux JE, Juurlink DN, Alter DA . Thiazolidinediones and cardiovascular outcomes in older patients with diabetes. JAMA 2007; 298: 2634–2643.
McLaughlin T, Lamendola C, Liu A, Abbasi F . Preferential fat deposition in subcutaneous versus visceral depots is associated with insulin sensitivity. J Clin Endocrinol Metab 2011; 96: E1756–E1760.
Acknowledgements
The research was funded in part by the American Diabetes Association 1-09-IN-13 and the Mathile Institute for the Advancement of Human Nutrition, awarded to NVD. This work used Genomics core facilities at the Pennington Biomedical Research Center, that are supported in part by COBRE (NIH P20-RR021945) and NORC (NIH 1P30-DK072476) center grants from the National Institutes of Health. The human studies were supported by CIHR (Canadian Institute for Health Research) grants: 63279 MONET study (Montreal Ottawa New Emerging Team) as well and from Genome Canada-Quebec (CAO, Complications Associated with Obesity project) to RRL. RRL holds scholarships from the Fonds de Recherche en Santé du Québec (FRSQ) and the J-A DeSève chair in clinical research.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest associated with this paper other than the following: (a) Patent filed: NV Dhurandhar and TC Holland. Ad-36 E4orf1, E1A and obesity and diabetes. November 2006. File No. 05P09. (b) Provisional patent filed: adenovirus Ad36 E4orf1 protein for prevention and treatment of non-alcoholic fatty liver disease (July 2010). (c) United States patent granted: adenovirus 36 E4orf1 gene and protein and their uses (US 8,008,436B2, dated 30 August 2011).
Rights and permissions
About this article
Cite this article
Krishnapuram, R., Kirk-Ballard, H., Dhurandhar, E. et al. Insulin receptor-independent upregulation of cellular glucose uptake. Int J Obes 37, 146–153 (2013). https://doi.org/10.1038/ijo.2012.6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ijo.2012.6
Keywords
This article is cited by
-
What we know and what we need to know about adenovirus 36-induced obesity
International Journal of Obesity (2020)
-
Obesity and Diabetes in an Arab population: Role of Adenovirus 36 Infection
Scientific Reports (2020)
-
E4orf1 protein reduces the need for endogenous insulin
Nutrition & Diabetes (2019)
-
Insulin-sparing and fungible effects of E4orf1 combined with an adipocyte-targeting sequence in mouse models of type 1 and type 2 diabetes
International Journal of Obesity (2017)