Abstract
OBJECTIVE:
The objective was to evaluate the association of body size and fat distribution with the risk of colorectal cancer (CRC) in Chinese men and women.
DESIGN:
This was a population-based, prospective cohort study.
SUBJECTS:
The analysis included 134 255 Chinese adults enrolled in the Shanghai Women’s Health Study and the Shanghai Men’s Health Study, with an average follow-up of 11.0 and 5.5 years, respectively.
MEASUREMENTS:
Waist circumference (WC), body mass index (BMI) and waist-to-hip ratio (WHR) were measured by trained interviewers at baseline. Multivariable Cox models were used to calculate adjusted hazard ratios (HRs) for incident CRC.
RESULTS:
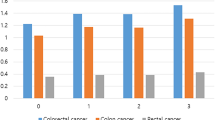
A total of 935 incident CRC cases were identified. Both measures of general adiposity (measured by BMI) and central adiposity (measured by WHR and WC) were significantly associated with an increased risk of colon cancer in men but not in women. Multivariable-adjusted HRs for colon cancer in men in the highest compared with the lowest quintiles were 2.15 (95% confidence interval (CI): 1.35–3.43; P for trend=0.0006) for BMI, 1.97 (95% CI: 1.19–3.24; P for trend=0.0004) for WHR and 2.00 (95% CI: 1.21–3.29; P for trend=0.0002) for WC. The BMI-associated risk was attenuated in analyses stratified by WHR, whereas the WHR-associated risk remained significant in the high BMI stratum (HR for comparison of extreme tertiles of WHR: 3.38, 95% CI: 1.47–7.75; P for trend =0.0002). None of these anthropometric measures were significantly associated with rectal cancer.
CONCLUSION:
Obesity, particularly central obesity, was associated with an increased risk of colon cancer in men.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Ferlay J, Shin HR, Bray F, Forman D, Mathers C, Parkin DM . GLOBOCAN 2008 v1.2, Cancer Incidence and Mortality Worldwide: IARC CancerBase No.10 [Internet]. International Agency for Research on Cancer: Lyon, France, 2010.
Gao Y, Lu W . Cancer Incidence, Mortality and Survival Rates in Urban Shanghai (1973-2000). Second Military Medical University Press: Shanghai, 2007.
Jin F, Devesa SS, Chow WH, Zheng W, Ji BT, Fraumeni JJ et al. Cancer incidence trends in urban Shanghai, 1972–1994: an update. Int J Cancer 1999; 83: 435–440.
Wang H, Du S, Zhai F, Popkin BM . Trends in the distribution of body mass index among Chinese adults, aged 20–45 years (1989–2000). Int J Obes (Lond) 2007; 31: 272–278.
IARC: International Agency for Research in Cancer. Weight control and physical activity. In: Vainio H, Bianchini F (eds). Handbooks of Cancer Prevention. IARC Press: Lyon, pp 85–94, 2002.
World Cancer Research Fund/American Institute for Cancer Research. Food, Nutrition, Physical Activity, and the Prevention of Cancer: A Global Perspective. AICR: Washington, DC, 2007.
Ning Y, Wang L, Giovannucci EL . A quantitative analysis of body mass index and colorectal cancer: findings from 56 observational studies. Obes Rev 2010; 11: 19–30.
Moghaddam AA, Woodward M, Huxley R . Obesity and risk of colorectal cancer: a meta-analysis of 31 studies with 70,000 events. Cancer Epidemiol Biomarkers Prev 2007; 16: 2533–2547.
Larsson SC, Wolk A . Obesity and colon and rectal cancer risk: a meta-analysis of prospective studies. Am J Clin Nutr 2007; 86: 556–565.
Dai Z, Xu YC, Niu L . Obesity and colorectal cancer risk: a meta-analysis of cohort studies. World J Gastroenterol 2007; 13: 4199–4206.
Matsuo K, Mizoue T, Tanaka K, Tsuji I, Sugawara Y, Sasazuki S et al. Association between body mass index and the colorectal cancer risk in Japan: pooled analysis of population-based cohort studies in Japan. Ann Oncol 2012; 23: 479–490.
Giovannucci E . Obesity, gender, and colon cancer. Gut 2002; 51: 147.
MacInnis RJ, English DR, Hopper JL, Gertig DM, Haydon AM, Giles GG . Body size and composition and colon cancer risk in women. Int J Cancer 2006; 118: 1496–1500.
MacInnis RJ, English DR, Hopper JL, Haydon AM, Gertig DM, Giles GG . Body size and composition and colon cancer risk in men. Cancer Epidemiol Biomarkers Prev 2004; 13: 553–559.
Pischon T, Lahmann PH, Boeing H, Friedenreich C, Norat T, Tjonneland A et al. Body size and risk of colon and rectal cancer in the European Prospective Investigation Into Cancer and Nutrition (EPIC). J Natl Cancer Inst 2006; 98: 920–931.
Wang Y, Jacobs EJ, Patel AV, Rodriguez C, McCullough ML, Thun MJ et al. A prospective study of waist circumference and body mass index in relation to colorectal cancer incidence. Cancer Causes Control 2008; 19: 783–792.
Moore LL, Bradlee ML, Singer MR, Splansky GL, Proctor MH, Ellison RC et al. BMI and waist circumference as predictors of lifetime colon cancer risk in Framingham Study adults. Int J Obes Relat Metab Disord 2004; 28: 559–567.
Adams KF, Leitzmann MF, Albanes D, Kipnis V, Mouw T, Hollenbeck A et al. Body mass and colorectal cancer risk in the NIH-AARP cohort. Am J Epidemiol 2007; 166: 36–45.
Lin J, Zhang SM, Cook NR, Rexrode KM, Lee IM, Buring JE . Body mass index and risk of colorectal cancer in women (United States). Cancer Causes Control 2004; 15: 581–589.
Otani T, Iwasaki M, Inoue M . Body mass index, body height, and subsequent risk of colorectal cancer in middle-aged and elderly Japanese men and women: Japan public health center-based prospective study. Cancer Causes Control 2005; 16: 839–850.
Terry PD, Miller AB, Rohan TE . Obesity and colorectal cancer risk in women. Gut 2002; 51: 191–194.
Thygesen LC, Gronbaek M, Johansen C, Fuchs CS, Willett WC, Giovannucci E . Prospective weight change and colon cancer risk in male US health professionals. Int J Cancer 2008; 123: 1160–1165.
Zheng W, Chow WH, Yang G, Jin F, Rothman N, Blair A et al. The Shanghai Women’s Health Study: rationale, study design, and baseline characteristics. Am J Epidemiol 2005; 162: 1123–1131.
Cai H, Zheng W, Xiang YB, Xu WH, Yang G, Li H et al. Dietary patterns and their correlates among middle-aged and elderly Chinese men: a report from the Shanghai Men’s Health Study. Br J Nutr 2007; 98: 1006–1013.
Zhang X, Shu XO, Gao YT, Yang G, Matthews CE, Li Q et al. Anthropometric predictors of coronary heart disease in Chinese women. Int J Obes Relat Metab Disord 2004; 28: 734–740.
Chan AT, Giovannucci EL . Primary prevention of colorectal cancer. Gastroenterology 2010; 138: 2029–2043.
Siddiqui AA . Metabolic syndrome and its association with colorectal cancer: a review. Am J Med Sci 2011; 341: 227–231.
Donohoe CL, Doyle SL, Reynolds JV . Visceral adiposity, insulin resistance and cancer risk. Diabetol Metab Syndr 2011; 3: 12.
Giovannucci E . Nutrition, insulin, insulin-like growth factors and cancer. Horm Metab Res 2003; 35: 694–704.
Koohestani N, Tran TT, Lee W, Wolever TM, Bruce WR . Insulin resistance and promotion of aberrant crypt foci in the colons of rats on a high-fat diet. Nutr Cancer 1997; 29: 69–76.
Yamamoto S, Nakagawa T, Matsushita Y, Kusano S, Hayashi T, Irokawa M et al. Visceral fat area and markers of insulin resistance in relation to colorectal neoplasia. Diabetes Care 2010; 33: 184–189.
Hotamisligil GS . Inflammation and metabolic disorders. Nature 2006; 444: 860–867.
Ullman TA, Itzkowitz SH . Intestinal inflammation and cancer. Gastroenterology 2011; 140: 1807–1816.
MacInnis RJ, English DR, Haydon AM, Hopper JL, Gertig DM, Giles GG . Body size and composition and risk of rectal cancer (Australia). Cancer Causes Control 2006; 17: 1291–1297.
Oxentenko AS, Bardia A, Vierkant RA, Wang AH, Anderson KE, Campbell PT et al. Body size and incident colorectal cancer: a prospective study of older women. Cancer Prev Res (Phila) 2010; 3: 1608–1620.
Leitzmann MF, Moore SC, Koster A, Harris TB, Park Y, Hollenbeck A et al. Waist circumference as compared with body-mass index in predicting mortality from specific causes. PLoS One 2011; 6: e18582.
Kissebah AH, Vydelingum N, Murray R, Evans DJ, Hartz AJ, Kalkhoff RK et al. Relation of body fat distribution to metabolic complications of obesity. J Clin Endocrinol Metab 1982; 54: 254–260.
Racette SB, Evans EM, Weiss EP, Hagberg JM, Holloszy JO . Abdominal adiposity is a stronger predictor of insulin resistance than fitness among 50-95 year olds. Diabetes Care 2006; 29: 673–678.
Renehan AG, Tyson M, Egger M, Heller RF, Zwahlen M . Body-mass index and incidence of cancer: a systematic review and meta-analysis of prospective observational studies. Lancet 2008; 371: 569–578.
Tamimi RM, Hankinson SE, Colditz GA, Byrne C . Endogenous sex hormone levels and mammographic density among postmenopausal women. Cancer Epidemiol Biomarkers Prev 2005; 14: 2641–2647.
Nelson HD, Humphrey LL, Nygren P, Teutsch SM, Allan JD . Postmenopausal hormone replacement therapy: scientific review. JAMA 2002; 288: 872–881.
Acknowledgements
This work was supported by the United States Public Health Service Grants R01CA082729 (PI: X-O Shu), R37CA070867 (PI: W Zheng) and R01CA122364 (PI: G Yang). We are grateful to the participants and research staff of the Shanghai Women’s and Men’s Health Studies for their contributions to the study. We also thank Ms Bethanie Rammer for her assistance in preparing the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Li, H., Yang, G., Xiang, YB. et al. Body weight, fat distribution and colorectal cancer risk: a report from cohort studies of 134 255 Chinese men and women. Int J Obes 37, 783–789 (2013). https://doi.org/10.1038/ijo.2012.152
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ijo.2012.152
Keywords
This article is cited by
-
Abdominal obesity and digestive system cancer: a systematic review and meta-analysis of prospective studies
BMC Public Health (2023)
-
The underestimated impact of excess body weight on colorectal cancer risk: Evidence from the UK Biobank cohort
British Journal of Cancer (2023)
-
Is the association of overweight and obesity with colorectal cancer underestimated? An umbrella review of systematic reviews and meta-analyses
European Journal of Epidemiology (2023)
-
Adiposity and mortality in older Chinese: an 11-year follow-up of the Guangzhou Biobank Cohort Study
Scientific Reports (2020)
-
Mortality trends of colorectal cancer among overweight patients at the global and national levels
International Journal of Colorectal Disease (2019)