Abstract
Objective:
The SH3-domain GRB2-like (endophilin)-interacting protein 1 (SGIP1) gene has been shown to be differentially expressed in the hypothalamus of lean versus obese Israeli sand rats (Psammomys obesus), and is suspected of having a role in regulating food intake. The purpose of this study was to assess the role of genetic variation in SGIP1 in human disease.
Subjects:
We performed single-nucleotide polymorphism (SNP) genotyping in a large family pedigree cohort from the island of Mauritius. The Mauritius Family Study (MFS) consists of 400 individuals from 24 Indo-Mauritian families recruited from the genetically homogeneous population of Mauritius. We measured markers of the metabolic syndrome, including diabetes and obesity-related phenotypes such as fasting plasma glucose, waist:hip ratio, body mass index and fat mass.
Results:
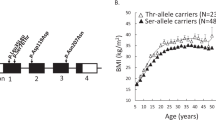
Statistical genetic analysis revealed associations between SGIP1 polymorphisms and fat mass (in kilograms) as measured by bioimpedance. SNP genotyping identified associations between several genetic variants and fat mass, with the strongest association for rs2146905 (P=4.7 × 10−5). A strong allelic effect was noted for several SNPs where fat mass was reduced by up to 9.4% for individuals homozygous for the minor allele.
Conclusions:
Our results show association between genetic variants in SGIP1 and fat mass. We provide evidence that variation in SGIP1 is a potentially important determinant of obesity-related traits in humans.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
WHO. World Health Organisation Factsheet No.311. WHO: Geneva, 2005.
Shaw JE, Sicree RA, Zimmet PZ . Global estimates of the prevalence of diabetes for 2010 and 2030. Diabetes Res Clin Pract 2010; 87: 4–14.
Barsh GS, Schwartz MW . Genetic approaches to studying energy balance: perception and integration. Nat Rev Genet 2002; 3: 589–600.
Schwartz MW, Woods SC, Porte Jr D, Seeley RJ, Baskin DG . Central nervous system control of food intake. Nature 2000; 404: 661–671.
Benzinou M, Chevre JC, Ward KJ, Lecoeur C, Dina C, Lobbens S et al. Endocannabinoid receptor 1 gene variations increase risk for obesity and modulate body mass index in European populations. Hum Mol Genet 2008; 17: 1916–1921.
Jackson RS, Creemers JW, Ohagi S, Raffin-Sanson ML, Sanders L, Montague CT et al. Obesity and impaired prohormone processing associated with mutations in the human prohormone convertase 1 gene. Nat Genet 1997; 16: 303–306.
Lange LA, Norris JM, Langefeld CD, Nicklas BJ, Wagenknecht LE, Saad MF et al. Association of adipose tissue deposition and beta-2 adrenergic receptor variants: the IRAS family study. Int J Obes (Lond) 2005; 29: 449–457.
Loos RJ, Rankinen T, Tremblay A, Perusse L, Chagnon Y, Bouchard C . Melanocortin-4 receptor gene and physical activity in the Quebec Family Study. Int J Obes (Lond) 2005; 29: 420–428.
Ribases M, Gratacos M, Fernandez-Aranda F, Bellodi L, Boni C, Anderluh M et al. Association of BDNF with restricting anorexia nervosa and minimum body mass index: a family-based association study of eight European populations. Eur J Hum Genet 2005; 13: 428–434.
Herbert A, Gerry NP, McQueen MB, Heid IM, Pfeufer A, Illig T et al. A common genetic variant is associated with adult and childhood obesity. Science 2006; 312: 279–283.
Liu YJ, Liu XG, Wang L, Dina C, Yan H, Liu JF et al. Genome-wide association scans identified CTNNBL1 as a novel gene for obesity. Hum Mol Genet 2008; 17: 1803–1813.
Meyre D, Delplanque J, Chevre JC, Lecoeur C, Lobbens S, Gallina S et al. Genome-wide association study for early-onset and morbid adult obesity identifies three new risk loci in European populations. Nat Genet 2009; 41: 157–159.
Hindorff LA, Sethupathy P, Junkins HA, Ramos EM, Mehta JP, Collins FS et al. Potential etiologic and functional implications of genome-wide association loci for human diseases and traits. Proc Natl Acad Sci USA 2009; 106: 9362–9367.
Barnett M, Collier GR, Collier FM, Zimmet P, O’Dea K . A cross-sectional and short-term longitudinal characterisation of NIDDM in Psammomys obesus. Diabetologia 1994; 37: 671–676.
Collier GR, Walder K, Lewandowski P, Sanigorski A, Zimmet P . Leptin and the development of obesity and diabetes in Psammomys obesus. Obes Res 1997; 5: 455–458.
Trevaskis J, Walder K, Foletta V, Kerr-Bayles L, McMillan J, Cooper A et al. Src homology 3-domain growth factor receptor-bound 2-like (endophilin) interacting protein 1, a novel neuronal protein that regulates energy balance. Endocrinology 2005; 146: 3757–3764.
Walder KR, Fahey RP, Morton GJ, Zimmet PZ, Collier GR . Characterization of obesity phenotypes in Psammomys obesus (Israeli sand rats). Int J Exp Diabetes Res 2000; 1: 177–184.
Jowett JB, Elliott KS, Curran JE, Hunt N, Walder KR, Collier GR et al. Genetic variation in BEACON influences quantitative variation in metabolic syndrome-related phenotypes. Diabetes 2004; 53: 2467–2472.
Collier GR, McMillan JS, Windmill K, Walder K, Tenne-Brown J, de Silva A et al. Beacon: a novel gene involved in the regulation of energy balance. Diabetes 2000; 49: 1766–1771.
Curran JE, Jowett JB, Elliott KS, Gao Y, Gluschenko K, Wang J et al. Genetic variation in selenoprotein S influences inflammatory response. Nat Genet 2005; 37: 1234–1241.
Uezu A, Horiuchi A, Kanda K, Kikuchi N, Umeda K, Tsujita K et al. SGIP1alpha is an endocytic protein that directly interacts with phospholipids and Eps15. J Biol Chem 2007; 282: 26481–26489.
Shmulewitz D, Heath SC, Blundell ML, Han Z, Sharma R, Salit J et al. Linkage analysis of quantitative traits for obesity, diabetes, hypertension, and dyslipidemia on the island of Kosrae, Federated States of Micronesia. Proc Natl Acad Sci USA 2006; 103: 3502–3509.
Jowett JB, Diego VP, Kotea N, Kowlessur S, Chitson P, Dyer TD et al. Genetic influences on type 2 diabetes and metabolic syndrome related quantitative traits in Mauritius. Twin Res Hum Genet 2009; 12: 44–52.
Boehnke M . Allele frequency estimation from data on relatives. Am J Hum Genet 1991; 48: 22–25.
Almasy L, Blangero J . Multipoint quantitative-trait linkage analysis in general pedigrees. Am J Hum Genet 1998; 62: 1198–1211.
Blangero J, Goring HH, Kent Jr JW, Williams JT, Peterson CP, Almasy L et al. Quantitative trait nucleotide analysis using Bayesian model selection. Hum Biol 2005; 77: 541–559.
Boerwinkle E, Chakraborty R, Sing CF . The use of measured genotype information in the analysis of quantitative phenotypes in man. I. Models and analytical methods. Ann Hum Genet 1986; 50: 181–194.
Abecasis GR, Cookson WO, Cardon LR . Pedigree tests of transmission disequilibrium. Eur J Hum Genet 2000; 8: 545–551.
Havill LM, Dyer TD, Richardson DK, Mahaney MC, Blangero J . The quantitative trait linkage disequilibrium test: a more powerful alternative to the quantitative transmission disequilibrium test for use in the absence of population stratification. BMC Genet 2005; 6 (Suppl 1): S91.
Li J, Ji L . Adjusting multiple testing in multilocus analyses using the eigenvalues of a correlation matrix. Heredity 2005; 95: 221–227.
Nyholt DR . A simple correction for multiple testing for single-nucleotide polymorphisms in linkage disequilibrium with each other. Am J Hum Genet 2004; 74: 765–769.
Cummings N, Dyer TD, Kotea N, Kowlessur S, Chitson P, Zimmet P et al. Genome-wide scan identifies a quantitative trait locus at 4p15.3 for serum urate. Eur J Hum Genet 2010; 18: 1243–1247.
Cheverud JM . A simple correction for multiple comparisons in interval mapping genome scans. Heredity 2001; 87: 52–58.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Cummings, N., Shields, K., Curran, J. et al. Genetic variation in SH3-domain GRB2-like (endophilin)-interacting protein 1 has a major impact on fat mass. Int J Obes 36, 201–206 (2012). https://doi.org/10.1038/ijo.2011.67
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ijo.2011.67