Abstract
Objective:
To analyze the effect of the juice obtained from two varieties of sweet orange (Citrus sinensis L. Osbeck), Moro (a blood orange) and Navelina (a blond orange), on fat accumulation in mice fed a standard or a high-fat diet (HFD).
Methods:
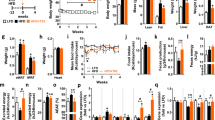
Obesity was induced in male C57/Bl6 mice by feeding a HFD. Moro and Navelina juices were provided instead of water. The effect of an anthocyanin-enriched extract from Moro oranges or purified cyanidin-3-glucoside (C3G) was also analyzed. Body weight and food intake were measured regularly over a 12-week period. The adipose pads were weighted and analyzed histologically; total RNA was also isolated for microarray analysis.
Results:
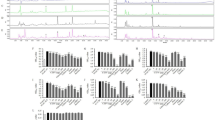
Dietary supplementation of Moro juice, but not Navelina juice significantly reduced body weight gain and fat accumulation regardless of the increased energy intake because of sugar content. Furthermore, mice drinking Moro juice were resistant to HFD-induced obesity with no alterations in food intake. Only the anthocyanin extract, but not the purified C3G, slightly affected fat accumulation. High-throughput gene expression analysis of fat tissues confirmed that Moro juice could entirely rescue the high fat-induced transcriptional reprogramming.
Conclusion:
Moro juice anti-obesity effect on fat accumulation cannot be explained only by its anthocyanin content. Our findings suggest that multiple components present in the Moro orange juice might act synergistically to inhibit fat accumulation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Diet, nutrition and the prevention of chronic diseases. World Health Organ Tech Rep Ser 2003; 916: i–viii, 1–149, backcover.
Welsh JA, Cogswell ME, Rogers S, Rockett H, Mei Z, Grummer-Strawn LM . Overweight among low-income preschool children associated with the consumption of sweet drinks: Missouri, 1999-2002. Pediatrics 2005; 115: e223–e229.
Malik VS, Schulze MB, Hu FB . Intake of sugar-sweetened beverages and weight gain: a systematic review. Am J Clin Nutr 2006; 84: 274–288.
McGinnis JM, Gootman JA, Kraak VI, Institute of Medicine (US). Committee on Food Marketing and the Diets of Children and Youth. Food Marketing to Children and Youth : Threat or Opportunity?. National Academies Press: Washington, DC, 2006, xx, 516, pp.
Vos MB, Kimmons JE, Gillespie C, Welsh J, Blanck HM . Dietary fructose consumption among US children and adults: the third national health and nutrition examination survey. Medscape J Med 2008; 10: 160.
West Europe Fruit Juice Market Research. Trends Analysis 2008. International Z: UK, 2008.
Neves MF . The Orange juice distribution channels: some characteristic, opportunities and threats. Ita Food Bev Tech 1999; 18: 15–28.
Perez-Cacho PR, Rouseff RL . Fresh squeezed orange juice odor: a review. Crit Rev Food Sci Nutr 2008; 48: 681–695.
Rampersaud GC . A comparison of nutrient density scores for 100% fruit juices. J Food Sci 2007; 72: S261–S266.
Rapisada P, Intrigliolo F . Anthocyanins in Blood Oranges: Composition and Biological Activity Vol 5. Pandalai SG, 2001.
Rapisarda P, Tomaino A, Lo Cascio R, Bonina F, De Pasquale A, Saija A . Antioxidant effectiveness as influenced by phenolic content of fresh orange juices. J Agric Food Chem 1999; 47: 4718–4723.
Reforgiato Recupero G . La propagazione dell’arancio Tarocco e del clementine Comune Vol 9 Italus Hortus, 2002.
Rapisarda P . Sample preparation for vitamin C analysis of pigmented orange juices. Ital J Food Sci 1996; 251–256.
Postorino E . I flavanoni dei succhi di arancia italiani. Essenze Deriv Agrum 1999; 149–158.
Rapisarda P, Carollo G, Fallico B, Tomaselli F, Maccarone E . Hydroxycinnamic acids as markers of Italian blood orange juices. J Agric Food Chem 1998; 46: 464–470.
Acquaviva R, Russo A, Galvano F, Galvano G, Barcellona ML, Li Volti G et al. Cyanidin and cyanidin 3-O-beta-D -glucoside as DNA cleavage protectors and antioxidants. Cell Biol Toxicol 2003; 19: 243–252.
Shih PH, Yeh CT, Yen GC . Anthocyanins induce the activation of phase II enzymes through the antioxidant response element pathway against oxidative stress-induced apoptosis. J Agric Food Chem 2007; 55: 9427–9435.
Hou DX, Kai K, Li JJ, Lin S, Terahara N, Wakamatsu M et al. Anthocyanidins inhibit activator protein 1 activity and cell transformation: structure-activity relationship and molecular mechanisms. Carcinogenesis 2004; 25: 29–36.
Xia M, Ling W, Zhu H, Ma J, Wang Q, Hou M et al. Anthocyanin attenuates CD40-mediated endothelial cell activation and apoptosis by inhibiting CD40-induced MAPK activation. Atherosclerosis 2009; 202: 41–47.
Guo H, Ling W, Wang Q, Liu C, Hu Y, Xia M . Cyanidin 3-glucoside protects 3T3-L1 adipocytes against H2O2- or TNF-alpha-induced insulin resistance by inhibiting c-Jun NH2-terminal kinase activation. Biochem Pharmacol 2008; 75: 1393–1401.
Berniakovich I, Trinei M, Stendardo M, Migliaccio E, Minucci S, Bernardi P et al. p66Shc-generated oxidative signal promotes fat accumulation. J Biol Chem 2008; 283: 34283–34293.
Carpene C, Iffiu-Soltesz Z, Bour S, Prevot D, Valet P . Reduction of fat deposition by combined inhibition of monoamine oxidases and semicarbazide-sensitive amine oxidases in obese Zucker rats. Pharmacol Res 2007; 56: 522–530.
Tsuda T, Horio F, Uchida K, Aoki H, Osawa T . Dietary cyanidin 3-O-beta-D-glucoside-rich purple corn color prevents obesity and ameliorates hyperglycemia in mice. J Nutr 2003; 133: 2125–2130.
Jayaprakasam B, Olson LK, Schutzki RE, Tai MH, Nair MG . Amelioration of obesity and glucose intolerance in high-fat-fed C57BL/6 mice by anthocyanins and ursolic acid in Cornelian cherry (Cornus mas). J Agric Food Chem 2006; 54: 243–248.
Tsuda T, Ueno Y, Yoshikawa T, Kojo H, Osawa T . Microarray profiling of gene expression in human adipocytes in response to anthocyanins. Biochem Pharmacol 2006; 71: 1184–1197.
Tsuda T . Regulation of adipocyte function by anthocyanins; possibility of preventing the metabolic syndrome. J Agric Food Chem 2008; 56: 642–646.
Rapisarda P, Fanella F, Maccarone E . Reliability of analytical methods for determining anthocyanins in blood orange juices. J Agric Food Chem 2000; 48: 2249–2252.
Rouseff RL, Youtsey CO . Quantitative survey of narirutin, naringin, hesperidin and neohesperidin in Citrus. J Agric Food Chem 1987; 35: 1027–1030.
Terpstra AH, Beynen AC, Everts H, Kocsis S, Katan MB, Zock PL . The decrease in body fat in mice fed conjugated linoleic acid is due to increases in energy expenditure and energy loss in the excreta. J Nutr 2002; 132: 940–945.
Huang DW, Sherman BT, Lempicki RA . Systematic and integrative analysis of large gene lists using DAVID Bioinformatics Resources. Nature Protoc 2009; 4: 44–57.
Dennis G Jr SB, Hosack DA, Yang J, Gao W, Lane HC, Lempicki RA . DAVID: Database for annotation, visualization, and integrated discovery. Genome Biol 2003; 4: P3.
Guarnieri S, Riso P, Porrini M . Orange juice vs vitamin C: effect on hydrogen peroxide-induced DNA damage in mononuclear blood cells. Br J Nutr 2007; 97: 639–643.
Arbo MD, Larentis ER, Linck VM, Aboy AL, Pimentel AL, Henriques AT et al. Concentrations of p-synephrine in fruits and leaves of Citrus species (Rutaceae) and the acute toxicity testing of Citrus aurantium extract and p-synephrine. Food Chem Toxicol 2008; 46: 2770–2775.
Sale C, Harris RC, Delves S, Corbett J . Metabolic and physiological effects of ingesting extracts of bitter orange, green tea and guarana at rest and during treadmill walking in overweight males. Int J Obes (Lond) 2006; 30: 764–773.
Bui LT, Nguyen DT, Ambrose PJ . Blood pressure and heart rate effects following a single dose of bitter orange. Ann Pharmacother 2006; 40: 53–57.
Haller CA, Benowitz NL, Jacob III P . Hemodynamic effects of ephedra-free weight-loss supplements in humans. Am J Med 2005; 118: 998–1003.
Szkudelska K, Nogowski L, Szkudelski T . Genistein affects lipogenesis and lipolysis in isolated rat adipocytes. J Steroid Biochem Mol Biol 2000; 75: 265–271.
Pyo YH, Seong KS . Hypolipidemic effects of monascus-fermented soybean extracts in rats fed a high-fat and -cholesterol diet. J Agric Food Chem 2009; 57: 8617–8622.
Shisheva A, Shechter Y . Quercetin selectively inhibits insulin receptor function in vitro and the bioresponses of insulin and insulinomimetic agents in rat adipocytes. Biochemistry 1992; 31: 8059–8063.
Rayalam S, Della-Fera MA, Baile CA . Phytochemicals and regulation of the adipocyte life cycle. J Nutr Biochem 2008; 19: 717–726.
Acknowledgements
We thank Elena Beltrami for many helpful discussions and Paola Dalton for the help in writing the paper. We thank also Ivan Toschi, Valentina and Nicoletta Cesari for the help in energy expenditure analysis and Simone Paolo Minardi for the assistance in the Affymetrix analysis. This work was supported by the EU FP6 FLORA project (FOOD-CT-01730) awarded to MG, CT, PGP, SM and GRR. This work was financially supported by EU FP6 FLORA project (FOOD-CT-01730).
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on International Journal of Obesity website (http://www.nature.com/ijo)
Supplementary information
Rights and permissions
About this article
Cite this article
Titta, L., Trinei, M., Stendardo, M. et al. Blood orange juice inhibits fat accumulation in mice. Int J Obes 34, 578–588 (2010). https://doi.org/10.1038/ijo.2009.266
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ijo.2009.266
Keywords
This article is cited by
-
Pre- and Post-harvest Elicitation with Methyl Jasmonate and Salicylic Acid Followed by Cold Storage Synergistically Improves Red Colour Development and Health-Promoting Compounds in Blood Oranges
Journal of Plant Growth Regulation (2024)
-
Purple corn extract induces long-lasting reprogramming and M2 phenotypic switch of adipose tissue macrophages in obese mice
Journal of Translational Medicine (2019)
-
The dominant allele Aft induces a shift from flavonol to anthocyanin production in response to UV-B radiation in tomato fruit
Planta (2017)
-
Anthocyanins and phenolic acids from a wild blueberry (Vaccinium angustifolium) powder counteract lipid accumulation in THP-1-derived macrophages
European Journal of Nutrition (2016)
-
p66Shc deletion or deficiency protects from obesity but not metabolic dysfunction in mice and humans
Diabetologia (2015)