Abstract
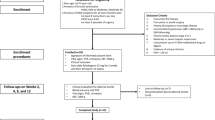
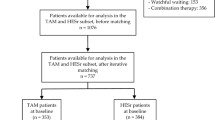
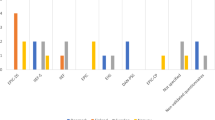
The sexuality and the management of benign prostatic hyperplasia (BPH) with alfuzosin (SAMBA) trial evaluated the effect of alfuzosin on sexual function in men treated for BPH using two sexual function scales: male sexual health questionnaire (MSHQ) and international index of erectile function (IIEF-15). A total of 148 patients with BPH were treated with alfuzosin for 24 weeks. The patients were followed at baseline, 4, 12 and 24 weeks after medication with alfuzosin. MSHQ was collected at every visit, whereas Qmax, IPSS and IIEF-15 were checked at baseline and end point. At the end point, Qmax (+4.7 ml s−1, P<0.01) and IPSS (−5.3, P<0.01) had improved significantly. Alfuzosin also significantly improved the total MSHQ (19.2%, 79.1–94.3, P<0.01) and the MSHQ ejaculatory scores (26.0%, 22.3–28.1, P=<0.01) versus baseline. Alfuzosin for the treatment of patients with BPH is effective in improving sexual function, as well as lower urinary tract symptoms (LUTSs) and quality of life, and is well tolerated.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 8 print issues and online access
$259.00 per year
only $32.38 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Hoesl CE, Woll EM, Burkart M, Altwein JE . Erectile dysfunction (ED) is prevalent, bothersome and underdiagnosed in patients consulting urologists for benign prostatic syndrome (BPS). Eur Urol 2005; 47: 511–517.
Braun M, Wassmer G, Klotz T, Reifenrath B, Mathers M, Engelmann U . Epidemiology of erectile dysfunction: results of the ‘Cologne Male Survey’. Int J Impot Res 2000; 12: 305–311.
Rosen RC, Giuliano F, Carson CC . Sexual dysfunction and lower urinary tract symptoms (LUTS) associated with benign prostatic hyperplasia (BPH). Eur Urol 2005; 47: 824–837.
Vallancien G, Emberton M, Harving N, van Moorselaar RJ . Sexual dysfunction in 1,274 European men suffering from lower urinary tract symptoms. J Urol 2003; 169: 2257–2261.
Brannigan RE . Ejaculatory disorders and lower urinary tract symptoms. Curr Urol Rep 2004; 5: 280–286.
Terai A, Ichioka K, Matsui Y, Yoshimura K . Association of lower urinary tract symptoms with erectile dysfunction in Japanese men. Urology 2004; 64: 132–136.
Kirby RS . Clinical uroselectivity of alfuzosin in the treatment of benign prostatic hyperplasia. Eur Urol 1998; 33 (Suppl 2): 19–27.
Roehrborn CG . Efficacy and safety of once-daily alfuzosin in the treatment of lower urinary tract symptoms and clinical benign prostatic hyperplasia: a randomized, placebo-controlled trial. Urology 2001; 58: 953–959.
van Kerrebroec P, Jardin A, van Cangh P, Laval KU . Long-term safety and efficacy of a once-daily formulation of alfuzosin 10 mg in patients with symptomatic benign prostatic hyperplasia: open-label extension study. Eur Urol 2002; 41: 54–60; discussion 60–61.
Rosen R, Seftel A, Roehrborn CG . Effects of alfuzosin 10 mg once daily on sexual function in men treated for symptomatic benign prostatic hyperplasia. Int J Impot Res 2007; 19: 480–485.
van Moorselaar RJ, Hartung R, Emberton M, Harving N, Matzkin H, Elhilali M et al. Alfuzosin 10 mg once daily improves sexual function in men with lower urinary tract symptoms and concomitant sexual dysfunction. BJU Int 2005; 95: 603–608.
Rosen R, Altwein J, Boyle P, Kirby RS, Lukacs B, Meuleman E et al. Lower urinary tract symptoms and male sexual dysfunction: the multinational survey of the aging male (MSAM-7). Eur Urol 2003; 44: 637–649.
Kirby RS, O'Leary MP, Carson C . Efficacy of extended-release doxazosin and doxazosin standard in patients with concomitant benign prostatic hyperplasia and sexual dysfunction. BJU Int 2005; 95: 103–109; discussion 109.
Zlotta AR, Teillac P, Raynaud JP, Schulman CC . Evaluation of male sexual function in patients with Lower Urinary Tract Symptoms (LUTS) associated with Benign Prostatic Hyperplasia (BPH) treated with a phytotherapeutic agent (Permixon), Tamsulosin or Finasteride. Eur Urol 2005; 48: 269–276.
Rosen RC, Catania J, Pollack L, Althof S, O'Leary M, Seftel AD . Male Sexual Health Questionnaire (MSHQ): scale development and psychometric validation. Urology 2004; 64: 777–782.
Oh CY, Lee JS, Chung BH . A validation and reliability study for the Korean version of the male sexual health questionnaire. Korean J Urol 2005; 46: 1308–1326.
Hofner K, Claes H, De Reijke TM, Folkestad B, Speakman MJ . Tamsulosin 0.4 mg once daily: effect on sexual function in patients with lower urinary tract symptoms suggestive of benign prostatic obstruction. Eur Urol 1999; 36: 335–341.
Nickel JC, Elhilali M, Emberton M, Vallancien G . The beneficial effect of alfuzosin 10 mg once daily in ‘real-life’ practice on lower urinary tract symptoms (LUTS), quality of life and sexual dysfunction in men with LUTS and painful ejaculation. BJU Int 2006; 97: 1242–1246.
Traish AM, Netsuwan N, Daley J, Padman-Nathan H, Goldstein I, Saenz de Tejada I . A heterogeneous population of alpha 1 adrenergic receptors mediates contraction of human corpus cavernosum smooth muscle to norepinephrine. J Urol 1995; 153: 222–227.
van Dijk MM, de la Rosette JJ, Michel MC . Effects of alpha(1)-adrenoceptor antagonists on male sexual function. Drugs 2006; 66: 287–301.
Giuliano F, Bernabe J, Droupy S, Alexandre L, Allard J . A comparison of the effects of tamsulosin and alfuzosin on neurally evoked increases in bladder neck and seminal vesicle pressure in rats. BJU Int 2004; 93: 605–608.
Acknowledgements
The SAMBA trial was sponsored by Sanofi-Aventis/Handok pharmacy Inc., Korea.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chung, BH., Lee, JY., Kim, CI. et al. Sexuality and the management of BPH with alfuzosin (SAMBA) trial. Int J Impot Res 21, 68–73 (2009). https://doi.org/10.1038/ijir.2008.62
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ijir.2008.62
Keywords
This article is cited by
-
Efficacy and safety of the simultaneous administration of mirodenafil and an α-blocker in men with BPH-LUTS: a multicenter open-label prospective study
International Journal of Impotence Research (2013)
-
Can open-label studies demonstrate efficacy in improving sexual function?
International Journal of Impotence Research (2009)
-
Measurement of benign prostatic hyperplasia treatment effects on male sexual function
International Journal of Impotence Research (2009)
-
Alfuzosin improves sexual function
Nature Reviews Urology (2009)