Abstract
D5 dopamine receptor (D5R) knock-out mice (D5−/−) have a higher blood pressure (BP) and higher reactive oxygen species (ROS) production than their D5R wild-type littermates (D5+/+). We tested the hypothesis that the high BP and increased ROS production in D5−/− mice may be caused by decreased heme oxygenase-1 (HO-1) expression and activity. We found that renal HO-1 protein expression and HO enzyme activity were decreased (65 and 50%, respectively) in D5−/− relative to D5+/+ mice. A 24 h of administration of hemin, an HO-1 inducer, increased HO-1 expression and HO activity (6.8- and 1.9-fold, respectively) and normalized the increased ROS production and BP in D5−/− mice. Expression of HO-1 protein and HO activity were increased (2.3- and 1.5-fold, respectively) in HEK cells that heterologously expressed human wild-type D5R (HEK-hD5R), but not the empty vector-transfected HEK-293 cells. Fenoldopam (Fen), a D5R agonist, increased HO activity (3 h), HO-1 protein expression, HO-1 and D5R colocalization and co-immunoprecipitation in HEK-hD5R cells. Cellular NADPH oxidase activity was decreased by 35% in HEK-hD5R that was abrogated with silencing of the heme oxygenase 1 gene (HMOX1). HMOX1 siRNA also impaired the ability of Fen to decrease NADPH oxidase activity in HEK-hD5R cells. In summary, the D5R positively regulates HO-1 through direct protein/protein interaction in the short-term and by increasing HO-1 protein expression in the long-term. The impaired D5R regulation of HO-1 and ROS production contributes to the pathogenesis of hypertension in D5−/− mice.
Similar content being viewed by others
Introduction
The peripheral dopaminergic system regulates renal function, sodium balance and blood pressure (BP) through its actions on renal hemodynamics, epithelial ion transport, and humoral/hormonal agents.1, 2, 3 Dopamine exerts its actions via two families of cell surface receptors that belong to the superfamily of G protein-coupled receptors. These include D1-like (D1R and D5R) and D2-like receptors (D2R, D3R, and D4R) in mammals.4, 5, 6 There is abundant evidence that an intact dopaminergic system is necessary to maintain normal BP, and that genetic hypertension is associated with alterations in dopamine production and receptor function.4, 5, 6 The disruption of any of the dopamine receptor genes in mice produces dopamine receptor subtype-specific hypertension.6, 7, 8, 9 The loci of the human D5 dopamine receptor (D5R) and its pseudogenes have been linked to human essential hypertension.10, 11 Disruption of the D5 receptor gene (Drd5) in mice (D5−/−) causes hypertension.12
Reactive oxygen species (ROS) are ubiquitous, highly diffusible reactive molecules produced by reduction of molecular oxygen, and include a series of oxygen intermediates, such as the superoxide anion, hydrogen peroxide, hydroxyl radical and hypochlorous acid.13, 14, 15 There are several intracellular sources contributing to ROS generation, including mitochondrial respiration, cyclooxygenases, lipoxygenases, and NADPH oxidases, the major source of superoxide anion in vascular and renal tissues.16 Increased production of ROS has been reported in human essential hypertension, as well as in animal models of genetic and acquired hypertension.13, 17, 18, 19, 20, 21 Increased levels of ROS are caused not only by enhanced oxidant activity, but also by deficient activity of antioxidant enzymes, for example, heme oxygenase (HO).22, 23, 24, 25, 26
HO catalyzes the degradation of heme, resulting in the formation of iron, carbon monoxide (CO), and biliverdin, which is subsequently converted to bilirubin by biliverdin reductase.22, 23 Three HO isoforms have been reported: an inducible isoform, HO-1; and two constitutively expressed isoforms, HO-2 and HO-3.22, 23 HO-1 is highly inducible in response to various stimuli that are associated with oxidative stress and inflammation, including heme, hypoxia, ischemia, heavy metals, ultraviolet radiation, endotoxin, and proinflammatory cytokines.24, 25 A lack of HO is involved in the pathogenesis of hypertension associated with increased ROS,23, 24, 27, 28 and induction of HO inhibits NADPH oxidase activity and decreases ROS production.29, 30, 31
We have reported that D5−/− mice are hypertensive, in part, because of increased systemic production of ROS through increased expression and activity of NADPH oxidase.13 In the current study, we tested the hypothesis that the D5R inhibits ROS production, not only by inhibiting NADPH oxidase expression and activity, but also by increasing HO-1 expression and activity.
Methods
D5−/− Mice
The generation of D5−/− mice has been reported.12, 13, 17 In this study, adult (6-month old) sex-matched sixth generation progeny (F6, C57/BL/6 Taconic) D5−/− and D5R wild-type (D5+/+) littermates fed with a normal sodium diet were used. Six-month old mice were studied to make them comparable with our previous reports.12, 13, 17 Our protocol was approved by the Georgetown University Animal Care and Use Committee. The BPs were measured from the femoral artery of avertin-anesthetized mice. The BPs were recorded 1 h after the induction of anesthesia and when the BPs were stable. An intraperitoneal injection of hemin (50 μmol kg−1), an HO-1 inducer, was given.23 Then, the femoral artery was repaired and the skin sutured. The mice were injected intramuscularly with Buprenorphine (0.3 mg kg−1) and allowed to recover. Twenty-four hours later, the mice were again anesthetized with avertin and BP was measured using the other femoral artery. At the end of the experiment, the kidneys were harvested and stored at −70 °C until use; the mice were euthanized with an overdose of pentobarbital.
Cell culture, treatment and transfection
HEK-293 cells, stably expressing full-length human D5R (hD5R) tagged withV5/HIS (HEK-hD5R) (passage 2–6), or empty vector-transfected HEK-293 (HEK-vector) cells were cultured in DMEM (Invitrogen, Carlsbad, CA, USA) containing 10% fetal bovine serum, 100 μg ml−1 streptomycin and 100 Uml−1 penicillin at 37 °C in an incubator with a humidified atmosphere of 95% air and 5% CO2, as reported.13, 17 Cells were pretreated for 2 h with serum-free medium and then treated with 1 μM fenoldopam (Fen) at different time points (0–24 h). Fen is selective for both D1R and D5R, but is specific for D5R in HEK-hD5R because D1R is not expressed.13, 17 Stable transfection of hD5R cDNA was verified by the presence of D5R protein (immunoblotting for V5/His and D5R).
HO-1 gene silencing with siRNA
HEK-hD5R (30–50% confluence) cells were split into 6-well plates the day before the transfection. Twenty microlitres of 10 μmol l−1 human HO-1 siRNA,31 diluted with 180 μl of Opti-Mem I, were mixed with 5 μl oligofectamine transfection reagent (Invitrogen). The mixture was incubated at room temperature for 20 min to form liposome–siRNA complexes prior to transfection of the cells (10 nM, 24 h).
Measurement of HO activity
Microsomal HO activity was assessed by bilirubin formation in the presence of purified biliverdin reductase.32 The amount of bilirubin formed was calculated using an extinction coefficient of 40 mmol l−1 cm−1 at a wavelength from 465 to 530 nm. HO activity was expressed as nanomol of bilirubin formed per milligram of protein per hour (nmol bilirubin per mg protein per h).
Immunoblotting analysis
Kidney homogenates or cell lysates were used to measure HO-1, HO-2 and β-actin expressions by immunoblotting with monoclonal anti-HO-1, anti-HO-2 (Stressgen Biotechnologies; British Columbia, Canada) and anti-β-actin antibodies (Millipore, Billerica, MA, USA). The membrane was washed three times with TBS-T buffer and subsequently probed with a secondary horseradish peroxidase-conjugated goat anti-mouse antibody (Santa Cruz Biotechnology, Santa Cruz, CA, USA). Chemiluminescence was detected using an ECL detection kit (Pierce Biotechnology Inc., Rockford, IL, USA), followed by autoradiography. Protein loading and membrane transfer were determined by immunoblotting for β-actin.
Immunoprecipitation
Immunoprecipitation was performed as described.33 Equal amounts of lysate protein of HEK-hD5R cells treated with Fen (1 μmol l−1, 3 and 12 h) were incubated with 2 μg ml−1 of anti-V5 antibody (Santa Cruz Biotechnology) for 1 h and protein-G agarose at 4 °C overnight. The immunoprecipitate was pelleted, washed four times with PBS, resuspended in sample buffer, boiled and immunoblotted with the HO-1 antibody. To determine the specificity of the bands, normal mouse IgG (negative control) and HO-1 antibody (positive control) were used as immunoprecipitants in lieu of the V5 antibody.
Immunofluorescence microscopy
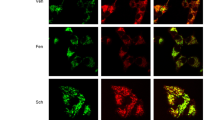
HEK-hD5R cells treated with vehicle or Fen (1 μmol l−1, 3 and 12 h) were used for immunofluorescence. The D5R was immunostained with monoclonal anti-V5 antibody and HO-1 with monoclonal anti-HO-1 antibody. For a negative control, the primary antibodies were replaced with normal mouse serum at an appropriate dilution. Colocalization of the D5R and HO-1 was identified by the appearance of a yellow color in the merged images.
Real-Time quantitative PCR
Total RNA extracted from mouse kidney or HEK-hD5R cells with RNeasy Mini Kit (Qiagen, Valencia, CA, USA) was reverse transcribed using M-MLV reverse transcriptase (Invitrogen). Two microlitres of the cDNA were used for real-time PCR (SYBR Green qPCR Supermix UDG Kit, Invitrogen): 50 °C for 2 min and 95 °C for 10 min, followed by 40 cycles at 95 °C for 15 s and 63 °C for 60 s. Real-time PCR reactions were carried out in a total volume of 25 μl using predesigned QuantiTect Primers for HO-1 and β-actin (Qiagen). All measurements were performed in triplicate to ensure reproducibility. The ratio of mean±s.e. of the expression of HO-1 to β-actin was calculated for sample-to-sample comparison.
NADPH oxidase assay and ROS production
NADPH oxidase activity was determined by NADPH-induced chemiluminescence in the presence of lucigenin (5 μmol l−1) and NADPH (100 μmol l−1).13 The specificity of the NADPH-dependent superoxide anion production was verified by treatment with diphenylene iodonium. NADPH oxidase activity was expressed as arbitrary units corrected by the protein concentration (AU per mg protein). ROS production in renal homogenates was quantified using the superoxide dismutase-inhibitable cytochrome c reduction assay.13
Statistical analysis
Data are expressed as mean±s.e. Comparison among and within groups of more than two was made by factorial and repeated-measures ANOVA and Newman–Keuls (multiple comparisons), respectively, and Student’s t-test was used for the comparison between two groups. A value of P<0.05 was considered statistically significant.
Results
In vivo studies
Blood pressure and heart rate in D5−/− mice
Pentobarbital-anesthetized D5−/− mice had higher systolic BP (123±1 vs. 96±1 mm Hg, P<0.01), diastolic BP(89±2 vs. 69±2 mm Hg, P<0.01) and mean arterial BP(100±2 vs. 78±1 mm Hg, P<0.01) than their D5+/+ littermates. The high BP of D5−/− mice was normalized 24 h after the administration of hemin, whereas the BP of D5+/+ littermates was not affected by hemin (Figures 1a,b and c). The heart rates were similar in pentobarbital-anesthetized D5+/+ and D5−/− mice before and after hemin treatment (Figure 1d).
Blood pressure, heart rate and ROS in D5−/− mice. Vehicle or hemin (50 μmol kg−1), an inducer of HO-1, was injected intraperitoneally and the systolic (a), diastolic (b), and mean arterial blood pressures (c), and heart rates (d) were measured before and ∼24 h after the injection of hemin. Results are mean±s.e. n=4 per group, *P<0.01, ANOVA, Newman–Keuls test. (e) Renal ROS production measured by cytochrome c reduction in D5−/− and D5+/+ mice 24 h after treatment with vehicle or hemin as above. Results are mean±s.e., n=4, *P<0.05, ANOVA, Newman–Keuls test.
ROS production in D5−/− mice
Renal ROS production was increased by 1.8-fold in D5−/− mice relative to D5+/+ littermates (D5−/−: 35.8±0.5 vs. D5+/+: 20.1±2.2; pmol per mg protein per min, P<0.05). The increased renal ROS production in D5−/− mice was almost completely abolished by hemin treatment (Figure 1e).
HO-1 expression in D5−/− mice
The renal protein expression of HO-1 was decreased by 65% in D5−/− relative to D5+/+ mice (D5−/−: 35±4%; D5+/+: 100±26%, P<0.05). Hemin treatment increased HO-1 protein expression by 6.8-fold in D5−/− mice (hemin: 237±8 and vehicle: 35±4%, respectively, P<0.01) without affecting its expression in D5+/+ mice (Figure 2a). In contrast, HO-2 protein did not differ between D5−/− and D5+/+ mice (Supplementary Figure S1A). We next determined if the increased HO-1 expression caused by hemin was due to increased gene transcription. We found that renal HO-1 mRNA expression (real-time QRT-PCR) was similar in D5−/− mice and D5+/+ littermates in the basal state; hemin treatment increased HO-1 mRNA expression to a similar extent in D5+/+ (hemin: 270±50%; vehicle: 100±3%, P<0.05) and D5−/− mice (hemin: 300±48%; vehicle: 98±5%, P<0.05) (Figure 2b). These data indicate that the cause of the differential HO-1 protein expression between the two mouse strains is at the post-transcriptional level.
HO-1 expression and HO enzyme activity in D5−/− mice. (a) Immunoblot analysis of HO-1 protein in renal homogenates from D5−/− and D5+/+ mice. Immunoblotting used a monoclonal antibody against HO-1, which recognizes a 32 kDa band. Results were normalized by β-actin and expressed as percent change relative to D5+/+ mice treated with vehicle. Results are mean±s.e., n=4, *P<0.05; #P<0.01, ANOVA, Newman–Keuls test. Insets: immunoblots of HO-1 and β-actin, two per group. (b) Real-time PCR analysis of HO-1 mRNA in renal homogenates from D5−/− and D5+/+ mice. Reverse transcription (RT) and real-time PCR were performed using SYBR Green qPCR Supermix system. Results were normalized by β-actin mRNA and expressed as percent change relative to D5+/+ mice treated with vehicle. Results are mean±s.e., n=4, *P<0.05, ANOVA, Newman–Keuls test. (c) Microsomal HO activity in kidneys from D5−/− and D5+/+ mice. Renal microsomal HO activity was measured as the rate of bilirubin formation. Results are mean±s.e., n=4, *P<0.01, ANOVA, Newman–Keuls test.
HO enzyme activity in D5−/− mice
Microsomal HO activity was decreased by 50% in the kidneys of D5−/− relative to D5+/+ mice (D5−/−: 3.5±0.3; D5+/+: 7.1±0.4, nmol bilirubin per mg protein per h, P<0.01). Hemin treatment increased renal HO activity (1.9-fold) in D5−/− mice (hemin: 6.7±0.4; vehicle: 3.5±0.3, nmol bilirubin per mg protein per h, P<0.01), but not in D5+/+ littermates (Figure 2c).
In vitro studies
HO-1 protein expression and HO activity in HEK-hD5R cells
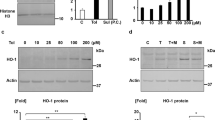
To evaluate directly the role of D5R in the regulation of HO-1 expression and activity, studies were performed in HEK-293 cells, which do not endogenously express D1R or D5R but were generated to express heterologously the human wild-type D5R (HEK-hD5R).13, 17 Expression of HO-1 protein was increased by 2.3-fold in HEK-hD5R cells relative to HEK-vector cells (n=3, P<0.01) (Figure 3a). Fen treatment increased HO-1 expression in a time- and concentration-dependent manner (Figures 3b and c) in HEK-hD5R cells. In contrast, Fen treatment did not alter HO-1 expression in HEK-vector cells (Supplementary Figure S1B). The Fen-mediated increase in HO-1 protein expression in HEK-hD5R cells was completely abolished by the addition of SCH23390 (D5R receptor antagonist in the absence of D1R), which, by itself, had no effect on HO-1 expression (Figure 3d). Microsomal HO activity was increased by 1.5-fold in HEK-hD5R cells relative to HEK-vector cells (control: 0.008±0.001; 0 min: 0.012±0.001, nmol bilirubin per mg protein per h, P<0.05) (Figure 3e), suggesting that the presence of the constitutively active D5R can increase HO activity. In HEK-hD5R cells, HO activity was increased by 2.2-fold after 3 h of Fen treatment (3 h: 0.027±0.001, nmol bilirubin per mg protein per h, P<0.01) (Figure 3e) and decreased thereafter, but remained at least twice the basal levels at 24 h.
HO-1 protein expression and HO activity in HEK-hD5R cells. (a) Immunoblot analysis of HO-1 protein in HEK-hD5R cells. Immunoblotting used monoclonal antibody against HO-1. Results were normalized by expression of β-actin and expressed as percent change relative to HEK-vector cells. Results are mean±s.e., n=3, *P<0.01, t-test. Insets: representative immunoblots of HO-1 and β-actin. (b) Time-dependent effect of Fen (1 μmol l−1) on HO-1 protein expression in HEK-hD5R cells. Results were normalized by expression of β-actin and expressed as percent change relative to control (0 h). Results are mean±s.e., n=5, *P<0.05 vs. control (0 h), ANOVA, Newman–Keuls test. Insets: representative immunoblots of HO-1 and β-actin. (c) Concentration-dependent effect of Fen (24 h) on HO-1 protein expression in HEK-hD5R cells. Results were normalized by expression of β-actin and expressed as percent change relative to control. Results are mean±s.e., n=5, *P<0.05 vs. control (Ctr), ANOVA, Newman–Keuls test. Insets: representative immunoblots of HO-1 and β-actin. (d) Effect of Fen and SCH23390 (D5R antagonist in the absence of D1R) on HO-1 protein in HEK-hD5R cells. Cells were pretreated with vehicle or SCH23390 (1 μmol l−1) for 1 h and then treated with Fen (1 μmol l−1, 12 h). Results were normalized by β-actin and expressed as percent change relative to HEK-hD5R cells treated with vehicle. Results are mean±s.e., n=4, *P<0.05, ANOVA, Newman–Keuls test. Insets: representative immunoblots of HO-1 and β-actin. (e) Effect of Fen on HO activity in HEK-hD5R cells. Cells were treated with Fen (1 μmol l−1) at different time points and then harvested. Microsomal HO activity was measured as the rate of bilirubin formation and shown as mean±s.e., n=6, *P<0.05, #P<0.01 vs. 0 min, ANOVA, Newman–Keuls test.
Colocalization and co-immunoprecipitation of hD5R and HO-1 in HEK-hD5R cells
To determine the mechanism for the increased HO-1 activity (from 3 h) prior to the increase in HO-1 protein expression (from 12 h) by D5R, we next determined if D5R and HO-1 could directly interact with each other in HEK-hD5R cells by colocalization and co-immunoprecipitation studies. Using confocal immunofluorescence microscopy, we found that in vehicle-treated cells, the D5R was mainly located at the plasma membrane, whereas HO-1 was primarily located in the cytosol; there was some colocalization between D5R and HO-1 in either plasma membrane or cytosol (Figure 4a). Fen treatment (1 μmol l−1, 3 h) promoted D5R internalization and, along with HO-1, redistribution to the perinuclear area where they colocalized. Colocalization was also observed at the plasma membrane (Figure 4a). Colocalization continued to be observed after 12 h of treatment. There was physical interaction between D5R and HO-1 because co-immunoprecipitation was increased at 3 h of Fen (1 μmol l−1) treatment, and decreased after 12 h of treatment (Figure 4b), similar to that observed with HO activity (Figure 3e).
Interaction between D5R and HO-1. (a) D5R and HO-1 co-localization in HEK-hD5R cells. The cells were treated with vehicle or Fen (1 μmol l−1, 3 h and 12 h), and then D5R and HO-1 colocalization was determined by confocal microscopy. Colocalization appears as yellow after merging the images of D5R (red) and HO-1 (green). Images are representative of three experiments using different cell preparations. (b) D5R and HO-1 co-immunoprecipitation in HEK-hD5R cells. Cells were treated with vehicle or Fen (1 μmol l−1, 3 h and 12 h). Lysates of HEK-hD5R cells were immunoprecipitated (IP) and immunoblotted (IB) as shown. To determine the specificity of the bands, normal IgG was used as negative control and immunoblots of whole-cell lysate with HO-1 antibody were used as positive control. The studies were performed three times with similar results. A full color version of this figure is available at the Hypertension Research journal online.
NADPH oxidase activity in HEK-hD5R cells
We next examined the effect of the D5R on pro-oxidant (NADPH oxidase) and antioxidant (HO-1) enzymes in HEK-hD5R cells. As shown in Figure 5a, NADPH oxidase activity, determined by lucigenin chemiluminescence, was decreased by 35% in HEK-hD5R cells compared to HEK-vector cells (HEK-vector: 4600±239; HEK-hD5R: 2974±134 AU per mg protein, P<0.05).
NADPH oxidase activity in HEK-hD5R cells. (a) NADPH oxidase activity in HEK-hD5R cells. NADPH oxidase activity was measured using the lucigenin chemiluminescence method and expressed as AU per mg protein. Results are mean±s.e., n=4, *P<0.05, t-test. (b) Microsomal HO activity in HEK-vector and HEK-hD5R cells transfected with HO-1 siRNA or non-silencing siRNA for 24 h. HO activity was measured as the rate of bilirubin formation and shown as mean±s.e., n=3, *P<0.05, ANOVA, Newman–Keuls test. (c) NADPH oxidase activity in HEK-vector and HEK-hD5R cells. Cells were transfected with HO-1 siRNA or non-silencing siRNA for 24 h. n=3, *P<0.05, ANOVA, Newman–Keuls test. (d) Effect of D5R agonist on NADPH oxidase activity in HEK-hD5R cells transfected with HO-1 siRNA. Cells transfected with non-silencing siRNA or HO-1 siRNA for 12 h, and then treated with Fen (1 μmol l−1) for 12 h. n=4, *P<0.01, ANOVA, Newman–Keuls test.
To prove that HO-1 mediates, in part, the antioxidative effect of D5R, we studied the consequence of silencing HO-1 gene expression on the inhibitory effect of D5R on NADPH oxidase. As expected, 24 h after HO-1 siRNA transfection, mRNA and protein expressions of HO-1 were significantly decreased (70 and 76%, respectively) (Supplementary Figures S1C and S1D). HO activity was decreased to same level in HEK-vector cells (non-silencing siRNA: 0.017±0.001; HO-1 siRNA: 0.011±0.001, nmol bilirubin per mg protein per h, P<0.05) and HEK-hD5R cells (non-silencing siRNA: 0.023±0.001; HO-1 siRNA: 0.012±0.001, nmol bilirubin per mg protein per h, P<0.05) (Figure 5b), corroborating the results shown in Figures 3e and 5a regarding the constitutive activity of D5R and increased HO activity. As shown in Figure 5c, silencing HO-1, alone increased the NADPH oxidase activity by 1.6-fold in HEK-vector cells (non-silencing siRNA: 1074±83; HO-1 siRNA: 1602±106, AU per mg protein, P<0.05) and completed abolished the inhibitory effect of D5R on basal NADPH oxidase activity in HEK-hD5R cells, that is, NADPH oxidase activity was not constitutively decreased by D5R when HO-1 was silenced.
In HEK-hD5R cells, Fen (1 μmol l−1, 12 h) treatment decreased NADPH oxidase activity by 72% in non-silencing siRNA-transfected cells (vehicle: 3166±193; Fen: 886±92, AU per mg protein, P<0.01) (Figure 5d). In contrast, in HO-1 siRNA-transfected HEK-hD5R cells, the ability of Fen to decrease NADPH oxidase activity was reduced by about 50% (Fen+HO-1 siRNA: 2216±120, AU per mg protein, P<0.01) (Figure 5d), indicating that HO-1 mediated, in part, the ability of D5R to attenuate NADPH oxidase.
Discussion
Oxidative stress is increased in both humans with essential hypertension and several animal models of hypertension.13, 16, 17, 18, 19, 20, 24, 26, 27, 28, 34 We have reported that the increased BP in D5−/− mice is, in part, due to an increase in oxidative stress as a consequence of the activation of NADPH oxidase.13 The D5R constitutively inhibits the expression and activity of the pro-oxidant enzyme NADPH oxidase.13 We now show that the increase in oxidative stress and BP in D5−/− mice is also caused by a decrease in HO-1 protein. HO-1 but not HO-2 protein expression and microsomal HO activity are lower in D5−/− than D5+/+ mice. Hemin, an inducer of HO-1, increases HO-1 but not HO-2 expression, abolishes the oxidative stress and normalizes BP in D5−/− mice. The renal cellular effects of Drd5 deletion and HO-1 induction with hemin are consistent with those observed in human kidney cells; HO-1 protein and HO activity are increased and NADPH oxidase activity is decreased in HEK-hD5R cells, relative to empty vector-transfected HEK-293 cells, indicating that these effects are due to constitutive activity of D5R. In addition, D5R stimulation with Fen increases HO-1 protein expression and HO activity in HEK-hD5R cells. Silencing of HO-1 increases NADPH oxidase activity and impairs the ability of Fen to inhibit NADPH oxidase activity. These findings suggest that activation of D5R constitutively and positively regulates renal HO-1 protein expression and activity, and the absence of D5R results in an increase in ROS production due to both an increase in NADPH oxidase subunit expression (gp91phox, p47phox and Nox 4) and activity,13 and a decrease in HO-1 protein and activity (current study). Thus, in this study, we demonstrate for the first time, that renal HO-1 is positively regulated by D5R in vivo and in vitro, and that HO-1 negatively regulates NADPH oxidase activity via the D5R.
The levels of HO-1 mRNA are increased after 24 h treatment with hemin in both D5−/− and D5+/+ mice. However, whereas hemin does not affect HO-1 protein levels and activity in the wild-type (D5+/+) littermates, it increases HO-1 protein levels and normalizes HO-1 activity in D5−/− mice. The regulation of HO-1 expression is not only dependent on transcription but also on translation,35 and it is possible that the increased oxidative stress in the kidneys of D5−/− mice may have made them more sensitive to HO-1 protein induction than their wild-type counterparts.
In HEK-hD5R cells, D5R stimulation with Fen increases HO activity much earlier (3 h) (Figure 3e) than its ability to increase HO-1 protein expression (12 h) (Figure 3b). The D5R-mediated increase in HO-1 activity before there is an increase in HO-1 protein may be due to a direct effect of D5R on HO-1, because the colocalization and physical interaction between D5R and HO-1 are increased by Fen at 3 h when HO activity is also increased. These results suggest that the early increase in HO-1 activity after Fen treatment (3 h) may be due to protein–protein interaction, whereas the later increase (12 h) may be via increased protein expression.
Dopamine has been reported to increase HO-1 expression in neuronal and endothelial cells.36, 37 Fen has also been shown to increase HO-1 protein expression that may protect renal proximal tubule cells and kidneys from cold-ischemia and reperfusion injury.38 Our studies suggest that the dopamine receptor subtype involved is probably D5R. The mechanism by which D5R upregulates HO-1 protein expression is still unknown. Apomorphine, a dopamine agonist that activates both D1-like and D2-like receptors, stimulates the nuclear translocation of a basic leucine zipper transcription factor, nuclear factor-E2-related factor-2, increases the expression of HO-1 and prevents neuronal death.39 The antioxidant effect of D1-like receptor in human renal proximal tubule cells may also be modified by nuclear factor-E2-related factor-2.40 The Nrf2/HO-1 pathway can be regulated by the PI3K-Akt pathway,41, 42, 43 and D5R activation, via PI3K-Akt pathway, increases the phosphorylation of Akt44 in neurons. Thus, we propose that the activation of the PI3K-Akt pathway by D5R is involved in its ability to induce HO-1 protein expression.
In conclusion, we suggest that renal HO-1 is positively regulated by D5R and mediates, in part, the inhibitory effect of renal D5R on NADPH oxidase activity. D5R positively regulates HO-1 activity through direct protein–protein interaction in the short-term and via increasing HO-1 protein expression in the long-term. D5R, via HO-1, counter-regulates renal ROS production, thereby contributing to the maintenance of normal BP.45
References
Felder RA, Jose PA . Mechanisms of disease: the role of GRK4 in the etiology of essential hypertension and salt sensitivity. Nat Clin Pract Nephrol 2006; 2: 637–650.
Asghar M, Tayebati SK, Lokhandwala MF, Hussain T . Potential dopamine-1 receptor stimulation in hypertension management. Curr Hypertens Rep 2011; 13: 294–302.
Ortiz PA, Garvin JL . Intrarenal transport and vasoactive substances in hypertension. Hypertension 2001; 38: 621–624.
Beaulieu JM, Gainetdinov RR . The physiology, signaling, and pharmacology of dopamine receptors. Pharmacol Rev 2011; 63: 182–217.
Hussain T, Lokhandwala MF . Renal dopamine receptors and hypertension. Exp Biol Med 2003; 228: 134–142.
Zeng C, Sanada H, Watanabe H, Eisner GM, Felder RA, Jose PA . Functional genomics of the dopaminergic system in hypertension. Physiol Genomics 2004; 19: 233–246.
Bek MJ, Wang X, Asico LD, Jones JE, Zheng S, Li X, Eisner GM, Dk Grandy, Carey RM, Soares-da-Silva P, Jose PA . Angiotensin-II type 1 receptor-mediated hypertension in D4 dopamine receptor-deficient mice. Hypertension 2006; 47: 288–295.
Li XX, Bek M, Asico LD, Yang Z, Grandy DK, Goldstein DS, Rubinstein M, Eisner GM, Jose PA . Adrenergic and endothelin B receptor-dependent hypertension in dopamine receptor type-2 knockout mice. Hypertension 2001; 38: 303–308.
Ueda A, Ozono R, Oshima T, Yano A, Kambe M, Teranishi Y, Katsuki M, Chayama K . Disruption of the type 2 dopamine receptor gene causes a sodium-dependent increase in blood pressure in mice. Am J Hypertens 2003; 16: 853–858.
Cohn DH, Shohat T, Yahav M, Ilan T, Rechavi G, King L, Shohat M . A locus for an autosomal dominant form of progressive renal failure and hypertension at chromosome 1q21. Am J Hum Genet 2000; 67: 647–651.
Cooper RS, Luke A, Zhu X, Kan D, Adeyemo A, Rotimi C, Bouzekri N, Ward R, Rorimi C . Genome scan among Nigerians linking blood pressure to chromosomes 2, 3, and 19. Hypertension 2002; 40: 629–633.
Hollon TR, Bek MJ, Lachowicz JE, Ariano MA, Mezey E, Ramachandran R, Wersinger SR, Soares-da-Silva P, Liu ZF, Grinberg A, Drago J, Young WS, Westphal H, Jose PA, Sibley DR . Mice lacking D5 dopamine receptors have increased sympathetic tone and are hypertensive. J Neurosci 2002; 22: 10801–10810.
Yang Z, Asico LD, Yu P, Wang Z, Jones JE, Escano CS, Wang X, Quinn MT, Sibley DR, Romero GG, Felder RA, Jose PA . D5 dopamine receptor regulation of reactive oxygen species production, NADPH oxidase, and blood pressure. Am J Physiol Regul Integr Comp Physiol 2006; 290: R96–R104.
Banki K, Hutter E, Gonchoroff NJ, Perl A . Elevation of mitochondrial transmembrane potential and reactive oxygen intermediate levels are early events and occurs independently from activation of caspase in Fas signaling. J Immunol 1999; 162: 1466–1479.
Forman HJ, Torres M . Reactive oxygen species and cell signaling. Am J Respir Crit Care Med 2002; 166: S4–S8.
Harrison DG, Gongora MC . Oxidative stress and hypertension. Med Clin North Am 2009; 93: 621–635.
Yang Z, Asico LD, Yu P, Wang Z, Jones JE, Bai RK, Sibley DR, Felder RA, Jose PA . D5 dopamine receptor regulation of phospholipase D. Am J Physiol Heart Circ Physiol 2005; 288: H55–H61.
Quinn MT, Ammons MC, Deleo FR . The expanding role of NADPH oxidases in health and disease: no longer just agents of death and destruction. Clin Sci (Lond) 2006; 111: 1–20.
Briones AM, Touyz RM . Oxidative stress and hypertension: current concepts. Curr Hypertens Rep 2010; 12: 135–142.
Wang D, Strandgaard S, Iversen J, Wilcox CS . Asymmetric dimethylarginine, oxidative stress, and vascular nitric oxide synthase in essential hypertension. Am J Physiol Regul Integr Comp Physiol 2009; 296: R195–R200.
Vanecková I, Kramer HJ, Novotná J, Kazdová L, Opocenský M, Bader M, Ganten D, Cervenka L . Roles of nitric oxide and oxidative stress in the regulation of blood pressure and renal function in prehypertensive Ren-2 transgenic rats. Kidney Blood Press Res 2005; 28: 117–126.
Maines MD, Gibbs PE . 30 some years of heme oxygenase: from a "molecular wrecking ball" to a "mesmerizing" trigger of cellular events. Biochem Biophys Res Commun 2005; 338: 568–577.
Peterson S, Frishman WH, Abraham NG . Targeting heme oxygenase: therapeutic implications for diseases of the cardiovascular system. Cardiol Rev 2009; 17: 99–111.
Hosick PA, Stec DE . Heme oxygenase, a novel target for the treatment of hypertension and obesity? Am J Physiol Regul Integr Comp Physiol 2012; 302: R207–R214.
Ryter SW, Otterbein LE, Morse D, Choi AM . Heme oxygenase/carbon monoxide signaling pathways: regulation and functional significance. Mol Cell Biochem 2002; 234-235: 249–263.
Majid DS, Kopkan L . Nitric oxide and superoxide interactions in the kidney and their implication in the development of salt-sensitive hypertension. Clin Exp Pharmacol Physiol 2007; 34: 946–952.
Vera T, Kelsen S, Yanes LL, Reckelhoff JF, Stec DE . HO-1 induction lowers blood pressure and superoxide production in the renal medulla of angiotensin II hypertensive mice. Am J Physiol Regul Integr Comp Physiol 2007; 292: R1472–R1478.
Sabaawy HE, Zhang F, Nguyen X, Elhosseiny A, Nasjletti A, Schwartzman M, Dennery P, Kappas A, Abraham NG . Human heme oxygenase-1 gene transfer lowers blood pressure and promotes growth in spontaneously hypertensive rats. Hypertension 2001; 38: 210–215.
Taillé C, El-Benna J, Lanone S, Dang MC, Ogier-Denis E, Aubier M, Boczkowski J . Induction of heme oxygenase-1 inhibits NAD(P)H oxidase activity by down-regulating cytochrome b558 expression via the reduction of heme availability. J Biol Chem 2004; 27: 28681–28688.
Lanone S, Bloc S, Foresti R, Almolki A, Taillé C, Callebert J, Conti M, Goven D, Aubier M, Dureuil B, El-Benna J, Motterlini R, Boczkowski J . Bilirubin decreases NOS2 expression via inhibition of NAD(P)H oxidase: implications for protection against endotoxic shock in rats. FASEB J 2005; 13: 1890–1892.
Xia ZW, Li CE, Jin YX, Shi Y, Xu LQ, Zhong WW, Li YZ, Yu SC, Zhang ZL . Reduction of bilirubin by targeting human heme oxygenase-1 through siRNA. Exp Biol Med (Maywood) 2007; 232: 495–502.
Maines MD, Trakshel GM, Kutty RK . Characterization of two constitutive forms of rat liver microsomal heme oxygenase. Only one molecular species of the enzyme is inducible. J Biol Chem 1986; 261: 411–419.
Zheng S, Yu P, Zeng C, Wang Z, Yang Z, Andrews PM, Felder RA, Jose PA . Gα12- and Gα13-protein subunit linkage of D5 dopamine receptors in the nephron. Hypertension 2003; 41: 604–610.
Armando I, Wang X, Villar VA, Jones JE, Asico LD, Escano C, Jose PA . Reactive oxygen species–dependent hypertension in dopamine D2 receptor–deficient mice. Hypertension 2007; 49: 672–678.
Sikorsk EM, Hock T, Hill-Kapturczak N, Agarwal A . The story so far: molecular regulation of the heme oxygenase. Am J Physiol Renal Physiol 2004; 286: F425–F441.
Berger SP, Hunger M, Yard B, Schnuelle P, Van Der Woude FJ . Dopamine induces the expression of heme oxygenase-1 by human endothelial cells in vitro. Kidney Int 2000; 58: 2314–2319.
Schmidt J, Mertz K, Morgan JI . Regulation of heme oxygenase-1 expression by dopamine in cultured C6 glioma and primary astrocytes. Brain Res Mol Brain Res 1999; 10: 50–59.
Salahudeen AK, Yang M, Huang H, Dore S, Stec DE . Fenoldopam preconditioning: role of heme oxygenase-1 in protecting human tubular cells and rodent kidneys against cold-hypoxic injury. Transplantation 2011; 91: 176–182.
Hara H, Ohta M, Adachi T . Apomorphine protects against 6-hydroxydopamine-induced neuronal cell death through activation of the Nrf2-ARE pathway. J Neurosci Res 2006; 84: 860–866.
George LE, Lokhandwala MF, Asghar M . Novel role of NF-κB-p65 in antioxidant homeostasis in human kidney-2 cells. Am J Physiol Renal Physiol 2012; 302: F1440–F1446.
Zhang Z, Cui W, Li G, Yuan S, Xu D, Hoi MP, Lin Z, Dou J, Han Y, Lee SM . Baicalein protects against 6-OHDA-induced neurotoxicity through activation of Keap1/Nrf2/ HO-1 and involving PKCα and PI3K/AKT signaling pathways. J Agric Food Chem 2012; 60: 8171–8182.
Kim KC, Kang KA, Zhang R, Piao MJ, Kim GY, Kang MY, Lee SJ, Lee NH, Surh YJ, Hyun JW . Up-regulation of Nrf2-mediated heme oxygenase-1 expression by eckol, a phlorotannin compound, through activation of Erk and PI3K/Akt. Int J Biochem Cell Biol 2010; 42: 297–305.
Lee YJ, Jeong HY, Kim YB, Lee YJ, Won SY, Shim JH, Cho MK, Nam HS, Lee SH . Reactive oxygen species and PI3K/Akt signaling play key roles in the induction of Nrf2-driven heme oxygenase-1 expression in sulforaphane-treated human mesothelioma MSTO-211H cells. Food Chem Toxicol 2012; 50: 116–123.
Perreault ML, Jones-Tabah J, O'Dowd BF, George SR . A physiological role for the dopamine D5 receptor as a regulator of BDNF and Akt signalling in rodent prefrontal cortex. Int J Neuropsychopharmacol 2012; 25: 1–7.
Asico L, Zhang X, Jiang J, Cabrera D, Escano CS, Sibley DR, Wang X, Yang Y, Mannon R, Jones JE, Armando I, Jose PA . Lack of renal dopamine D5 receptors promotes hypertension. J Am Soc Nephrol 2011; 22: 82–89.
Acknowledgements
These studies were supported, in part, by the NIH grants, HL023081, DK039308, HL068686, HL074940, AR042426 and NS41043.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on Hypertension Research website
Supplementary information
Rights and permissions
About this article
Cite this article
Lu, Q., Yang, Y., Villar, V. et al. D5 dopamine receptor decreases NADPH oxidase, reactive oxygen species and blood pressure via heme oxygenase-1. Hypertens Res 36, 684–690 (2013). https://doi.org/10.1038/hr.2013.9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/hr.2013.9
Keywords
This article is cited by
-
The biological function of ELABELA and APJ signaling in the cardiovascular system and pre-eclampsia
Hypertension Research (2019)
-
Genetic polymorphisms associated with reactive oxygen species and blood pressure regulation
The Pharmacogenomics Journal (2019)
-
Sex differences in the blood antioxidant defense system in juvenile rats with various genetic predispositions to hypertension
Hypertension Research (2016)
-
Regulation of blood pressure, oxidative stress and AT1R by high salt diet in mutant human dopamine D5 receptor transgenic mice
Hypertension Research (2015)
-
CYBA (p22phox) variants associate with blood pressure and oxidative stress markers in hypertension: a replication study in populations of diverse altitudes
Hypertension Research (2015)