Abstract
Diabetes and hypertension frequently coexist and constitute the most notorious combination for the pathogenesis of diabetic nephropathy and retinopathy. Large clinical trials have clearly demonstrated that tight control of glycemia and/or blood pressure significantly reduces the incidence and progression of diabetic retinopathy (DR) and nephropathy. However, the mechanism by which hypertension interacts with diabetes to induce and/or exacerbate nephropathy and retinopathy is very unclear. Substantial evidence implicates the involvement of chronic inflammation and oxidative stress in the pathogenesis of DR and nephropathy. In addition, hypertension causes oxidative stress and inflammation in the kidney and retina. In the present review, we summarized data obtained from our research along with those from other groups to better understand the role of hypertension in the pathogenesis of diabetic nephropathy and retinopathy. It is suggested that oxidative stress and inflammation may be common denominators of kidney and retinal damage in the concomitant presence of diabetes and hypertension.
Similar content being viewed by others
Introduction
The incidence and prevalence of hypertension among diabetic patients have been documented in various populations; overall, hypertension occurs 1.5–2.0 times more often in diabetics than in non-diabetic people.1 In the United States, the Third National Health and Nutrition Examination Survey (1988–1994) found 71% of adults with diabetes to have elevated blood pressure (mean blood pressure⩾130/85 mm Hg or using antihypertensive medication).2 In individuals with type 1 diabetes, blood pressure levels are usually normal at diagnosis, with the onset of hypertension closely correlated with the onset of diabetic renal disease.3 In patients with type 2 diabetes, approximately one-third of the individuals have elevated blood pressure when diabetes is first diagnosed,4 with the prevalence of hypertension increasing to close to 100% when renal disease is manifested.5 The concomitant presence of hypertension and diabetes significantly magnifies the risk of diabetic microvascular complications, namely diabetic nephropathy and retinopathy.6 In this setting, large clinical trials, such as the United Kingdom Prospective Diabetes Study and the action in diabetes and vascular disease: preterax and diamicron modified release controlled evaluation, have clearly demonstrated that intensive control of both blood pressure and glycemia is effective in reducing the incidence and progression of diabetic retinopathy (DR) and nephropathy.7, 8 However, the mechanisms by which hypertension and diabetes interact to exacerbate diabetic nephropathy and retinopathy are poorly established. In this article, we explore the possibility that exaggerated oxidative stress and inflammation may underlie the mechanism of kidney and retinal diseases in diabetes and hypertension.
Clinical and epidemiological evidence of the detrimental effect of hypertension on diabetic nephropathy and retinopathy
In individuals with diabetes mellitus (DM), the relationship between hypertension and poor vascular outcomes, including diabetic renal and retinal diseases, is unequivocal and independent of other confounding factors. The impact of hypertension on outcomes is exponential rather than linear.9 Even in normotensive diabetic patients, lowering blood pressure can be beneficial in controlling diabetic nephropathy and retinopathy.10 In particular, in patients with type 2 DM, the risk of diabetic nephropathy and retinopathy was independently and additively attributable to hypertension and hyperglycemia.6
Arterial hypertension is a main risk factor in the development and progression of diabetic nephropathy,9 and a sustained reduction in blood pressure is probably the most effective single intervention to slow the progression of nephropathy in type 1 and type 2 DM.7, 9, 10, 11
In the Wisconsin Epidemiology Study of DR, the presence of hypertension in type 1 diabetic patients was associated with a 73% increase in the risk of incident proliferative DR.12 More recently, the Wisconsin Epidemiology Study of DR demonstrated that the 25-year incidence of visual impairment was related to poorer glycemia control and the presence of arterial hypertension. In these patients, the presence of arterial hypertension increases the risk of visual impairment by 72%.13 In addition, a recent study has suggested that even in normotensive diabetic individuals, elevated blood pressure levels are associated with DR. It has been demonstrated that in type 1 diabetic subjects, each increment of 5 mm Hg in night-time systolic and diastolic blood pressure leads to an increase in about 40% in the risk of DR.14
Experimental evidence that hypertension aggravates diabetic nephropathy and retinopathy
Many studies have demonstrated that the presence of hypertension aggravates and can contribute to renal and retinal disease in experimental DM. To assess the contribution of hypertension to diabetic renal disease, studies were performed in different animal models, including diabetic hypertensive Dahl salt-sensitive rats,15 Goldblatt hypertension, wherein one renal artery is constricted and the contralateral kidney is left intact with streptozotocin-induced diabetes,16 and spontaneously hypertensive/National Institute of Health (NIH)-corpulent rats, which are a model of type 2 diabetic nephropathy.17 However, most of these studies were performed in a genetic model of hypertension, namely a spontaneously hypertensive rat (SHR) that was rendered diabetic by injection of streptozotocin. In these rats, studies by Cooper et al.18 were the first to demonstrate that when compared with the genetically diabetic normotensive rats, specifically Wistar Kyoto (WKY) rats, diabetic SHR showed features of accelerated nephropathy, as evidenced by a 10-fold increase in albuminuria and a more pronounced thickness of glomerular basement membrane. These investigators have concluded that pre-existing hypertension may have an important role in the progression of diabetic renal disease.18 Similarly, it has been demonstrated that the combination of diabetes and hypertension (diabetic SHRs) lead to early and more severe markers of DR, both functional and morphological. Dosso et al.19 have shown that basement membrane thickness and increased permeability to serum albumin were observed in normotensive diabetic rats (diabetic WKY); however, these abnormalities were significantly enhanced when diabetes was combined with hypertension (diabetic SHRs). In addition, Hammes et al.20 have shown that the frequency of acellular capillaries, a morphological gold-standard marker of DR, was nearly twice as high in diabetic SHRs as in diabetic WKY. Furthermore, it has been shown that the presence of diabetes (diabetic WKY) increased retinal albumin clearance, a marker of retinal capillary permeability, by 40%, whereas an approximately 100% increase in retinal albumin clearance was noticed when diabetes was combined with hypertension (diabetic SHRs).21 In these scenarios, the importance of hypertension in renal and retinal disease was further supported by the observation that control of blood pressure ameliorates nephropathy and retinopathy in diabetic SHR.21
Collectively, the above-mentioned studies support the concept that elevated blood pressure contributes to diabetic nephropathy and retinopathy in SHR rats. However, they do not provide insights into the nature of the interaction of hypertension and diabetes in exacerbating diabetic nephropathy and retinopathy. To better understand this issue and separate the respective contributions of hypertension and diabetes to renal and retinal disease, it may be necessary to evaluate early renal and retinal changes relevant to the lesions of diabetic nephropathy and retinopathy. To this end, we have investigated whether the presence of a short period of mild genetic hypertension and diabetes would modify the amount of renal fibronectin, an extracellular matrix component that increases in the early phase of diabetic renal disease. We have shown that, in experimental-induced diabetes, the presence of genetic hypertension promotes earlier accumulation of renal fibronectin, a matrix protein thought to be involved in the development of renal glomerulosclerosis.22 Molecular abnormalities were also detected in the retinas of diabetic SHR following a short period of diabetes. It has been shown that a 15-day induction of diabetes in SHR leads to an increase in retinal expression of fibronectin, accompanied by an elevation in retinal capillary permeability, which has been shown to be associated with a higher expression of retinal vascular endothelial growth factor (VEGF).23
It has been suggested that cell cycle regulators can be altered by the presence of diabetes and that such alterations contribute to the pathogenesis of diabetic nephropathy.24 More specifically, it has been shown that diabetes leads to an increase in cyclin-dependent kinase inhibitors, such as p27 and p21, which prompt the renal cells to halt the cell cycle, signaling hypertrophy instead of replication.24 It has been suggested that an increase in kidney cyclin-dependent kinase inhibitors contributes to glomerular hypertrophy observed in the early stages of diabetic nephropathy in a model of spontaneous developing diabetes, the db/db mouse.25 The importance of the cyclin-dependent kinase inhibitor p27 in diabetic nephropathy is further demonstrated by the observation that knocking down p27 confers renal protection to diabetic mice.26 In particular, it has been shown that induction of diabetes in SHR for only 10 days leads to kidney hypertrophy, accompanied by a decrease in renal cell replication secondary to an increase in the expression of cyclin-dependent kinase inhibitor p27.27 These abnormalities were not present in diabetic WKY rats. It was suggested that this cellular abnormality observed in diabetic SHRs may be involved in the mechanism by which hypertension interacts with diabetes to promote renal lesion.
A decrease in retinal cell replication was also observed in estreptozotocin (STZ)-induced diabetic SHR.23 The retinal cells that displayed a decrease in replication were identified as progenitor cells, and this abnormality was accompanied by an elevation in the expression of p27.23 Whether and how these alterations in the cell cycle in the retina of diabetic SHR that were associated with classical features of DR contribute to diabetic retinal disease requires further studies.
In vitro studies searching for the mechanism of the interaction of hypertension and diabetes in diabetic nephropathy and retinopathy
Clinical and experimental works suggest that systemic hypertension leading to increased intraglomerular pressure contributes to glomerular sclerosis in several renal diseases. Studies in experimental animals indicate that dilation of the afferent (precapillary) glomerular arteriole has an important role in increasing intraglomerular pressure and the glomerular filtration rate that may contribute to renal insult in diabetic nephropathy.28 Dilation of the afferent glomerular arteriole will also facilitate the transmission of elevated systemic hypertension to the glomerulus. Hyperglycemia amplifies the deleterious effects of blood pressure within the glomerulus by impairing the autoregulation of glomerular microcirculation.28 Intraglomerular hypertension, by expanding glomerular structures, induces mesangial cell stretch, leading to overproduction of the extracellular matrix and cytokines.29
Loss of autoregulation in diabetes has also been discovered in human retinal vasculature.30 This alteration would facilitate transmission of systemic hypertension to the retinal capillary bed, thus contributing to the mechanism of exacerbation of DR by hypertension.30 Interestingly, there is a positive correlation between loss of autoregulation and levels of glucose in the human retinal vasculature.30
Recently, molecular studies have identified several mechanisms that may be involved in the process of renal and retinal damage associated with hypertension and hyperglycemia. Most of these studies were based on an in vitro model of mechanical stimuli, such as stretch, mimicking the effect of increased pressure in glomerular and retinal cells in vivo.
The response of different markers relevant to the pathogenesis of DR has been investigated using retinal cells subjected to mechanical stretch. VEGF is thought to have a central role in increased vascular retinal permeability and in the development of intraocular neovascularization in DR.31 The fact that retinal pigment epithelial cells respond to stretch by increasing the expression of VEGF was first demonstrated by Seko et al.32 This finding was confirmed and extended by Suzuma et al.,33 who showed that bovine retinal endothelial and pericyte cells exposed to cyclic stretch increase the expression of VEGF and its receptor, both protein and mRNA. Later, it was suggested that stretch-induced retinal VEGF expression was mediated by phosphatidylinositol 3-kinase and protein kinase C.34 Furthermore, it has been demonstrated that stretch induces apoptosis in porcine retinal pericytes that may be mediated by oxidative stress.35 In addition, it has been shown that stretch induces a reduction in cell proliferation, increased apoptosis and profound morphological changes in the actin cytoskeleton assembly in bovine retinal pericytes.36 These alterations induced by stretch were magnified when the pericytes were exposed to high levels of glucose.36 Pericyte loss is a hallmark of DR and it can be secondary to pericyte apoptosis37 or migration of these cells out of capillaries.38
Numerous studies have investigated the effects of mechanical stretch in glomerular cells. It has been shown that mesangial cells exposed to continuous cycles of stretch/relaxation increase gene and/or protein expression of extracellular matrix components, including fibronectin, collagen I, III and IV and laminin.39 Furthermore, this accumulation of extracellular matrix, which occurs as a result of mechanical stretch, is markedly enhanced in a milieu of high glucose concentration.40 Of interest here is that mechanical stretch may promote extracellular matrix accumulation in cultured mesangial cells, not only by increasing synthesis of extracellular matrix components, but also by decreasing the activity of degradative enzymes.39 In addition, mechanical stretch induces both gene and protein expression of transforming growth factor (TGF)-β1,39, 41 a cytokine involved in the accumulation of extracellular matrix in diabetic nephropathy.41 Mechanical stretch has also been shown to induce gene expression, but not protein secretion of connective growth factor, a downstream mediator of TGF-β1 signaling.42
Mechanical stretch may also induce extracellular matrix production by interacting with inflammatory pathways. Indeed, human mesangial cells exposed to stretch showed enhanced monocyte chemoattractant protein-1 (MCP-1) and monocyte chemoattractant activity, and these results were accompanied by a reduction in both gene and protein expression of CC chemokine receptor 2, the cognate MCP-1 receptor.43 These effects, which were further magnified by simultaneous exposure of mesangial cells to stretch and high glucose, were mediated by nuclear factor-kappa B (NF-κB).44 Furthermore, both stretch and MCP-1 independently induce intercellular adhesion molecule-1 (ICAM-1) expression.43 This would increase phagocytic leukocyte adhesion to mesangial cells44 and amplify the inflammatory cascade45 and may contribute to glomerular injury by inducing the release of cytotoxic reactive oxygen species.46
Angiotensin II (Ang II), a powerful vasoconstrictor and trophic hormone, is central to renal damage in diabetes. In mesangial cells, it produces effects similar to those seen with mechanical stretch. Gruden et al.47 have demonstrated that both Ang II and mechanical stretch can independently induce the production of VEGF. In addition, mechanical stretch upregulates the Ang II type-1 (AT1) receptor, enhancing the cellular response to Ang II.47 It has been shown that, in mesangial cells, VEGF induces proliferation48 and enhances collagen synthesis.49
Mechanical stretch, when applied to human mesangial cells, upregulates protein expression of the facilitative glucose transporter-1, as well as basal glucose transport, through a protein kinase C-TGF-β1-dependent mechanism.50 This, in turn, results in an excessive production of extracellular matrix.50 In particular, overexpression of glucose transporter-1 in mesangial cells leads to increased extracellular matrix production even when the cells were grown in a normal glucose medium.51 Therefore, mechanical stretch-induced glucose transporter-1 expression may represent a mechanism whereby mechanical forces interact with glucose-mediated pathways in the pathogenesis of glomerular injury.50
Recently, several lines of evidence have suggested that podocytes, terminally differentiated epithelial glomerular cells, have a critical role in glomerular disease, including diabetic nephropathy. Podocytes are essential in maintaining the integrity of the glomerular capillary barrier by restricting the passage of macromolecules, such as albumin into the ultrafiltrate.52 Podocytes are also involved in the synthesis of glomerular extracellular matrix proteins and in counteracting capillary wall distension.52 In patients with DM, it has been demonstrated that a reduction in the number of podocytes is associated with proteinuria.53 Podocyte loss in diabetes may be secondary to podocyte apoptosis, necrosis or decreased cell adhesion to the basement membrane and detachment of viable podocytes.52 As for mesangial cells, several studies have explored the response of podocytes in culture to mechanical stress.
It has been demonstrated that podocytes in culture respond to mechanical stress.54 Specifically, mechanical stress reduces the size of the cell body of the podocyte and induces a reversible reorganization of the actin cytoskeleton, resulting in the disappearance of transverse stress fibers and formation of radial stress fibers connected to an actin-rich center.54 It has been hypothesized that podocytes may assume a state of intermediate adhesion as an adaptive strategy to escape mechanical damage.55 However, intermediate adhesion may be maladaptive in the long term, as it may lead to progressive loss of podocytes because of their greater ease of detachment from the glomerular basement membrane.55 To this end, it has recently been demonstrated that podocytes exposed to mechanical stretch or TGF-β1 reduce both the expression of integrin α3β1 and podocyte adhesion to extracellular matrix substrates.56 α3β1 integrin, which is abundantly expressed in the podocyte membrane,57 is the principal mechanism by which podocytes adhere to the glomerular basement membrane.58
Studies in cultured podocytes have demonstrated that these cells possess an independent renin–Ang system (RAS) that is activated by high glucose levels59 and mechanical stretch.60 In turn, activation of the podocyte RAS leads to an increase in podocyte apoptosis60 and also to a reduction in the expression of nephrin.61 Nephrin is a key component of the slit diaphragm,52, 61 a junction connecting the foot process of neighboring podocytes, which represents the major restriction site of protein filtration.52, 61 A decrease in nephrin expression occurs in vitro in podocytes exposed to glucose-modified proteins known as advanced glycation end-products62 and in vivo in streptozotocin-induced diabetic rats.63, 64
Contribution of inflammation and oxidative stress to DR and nephropathy
Both oxidative stress and inflammation have been strongly implicated in the pathogenesis of diabetic complications. The mitochondrial respiratory chain, nicotinamide adenine dinucleotide phosphate (NADPH) oxidase enzyme system, Nox4 (homologue of gp91phox subunit of NADPH oxidase) and advanced glycation-end product–receptor for advanced glycation end-product interaction have been found to induce oxidative stress in high-glucose conditions.65, 66, 67
Oxidative stress is conventionally defined as an imbalance between pro-oxidant stress and antioxidant defense. However, recent evidence indicates that the disruption of redox signaling is an important aspect of oxidative stress, sometimes more important than the pro-oxidant–antioxidant imbalance or the tissue damage induced by such imbalance.68 Therefore, a new definition of oxidative stress has been proposed as ‘an imbalance between oxidants and antioxidants in favor of the oxidants, leading to a disruption of redox signaling and control and/or molecular damage’.68 Consequences of oxidative stress can be very subtle to very serious (including oxidative damage to biomolecules, disruption of signal transduction, mutation and cell death), depending on the balance between reactive species generation and antioxidant defense.69 Oxidative stress can be estimated by production of a pro-oxidant, such as superoxide; expression or activity of an antioxidant, like superoxide dismutase or damage induced by oxidative stress to biomolecules, such as protein (nitration of tyrosine residues—nitrotyrosine), lipids (aldehydes from lipid peroxidation) and DNA (8-hydroxy-2′-deoxyguanosine, 8-OHdG).69
Studies in humans have shown an association between oxidative stress markers and the presence of DR and nephropathy. It has been shown that in type 2 diabetic patients with diabetic nephropathy and/or retinopathy, levels of 8-OHdG are significantly higher than in diabetic patients without these complications.70, 71 Furthermore, in a prospective study, it was demonstrated that in type 2 diabetic patients, higher levels of urinary 8-OHdG were associated with the progression of diabetic nephropathy.72
A recent study demonstrated that low doses of erythropoietin inhibit oxidative stress and early vascular changes in the experimental diabetic retina.73 The investigators of this study have concluded that erythropoietin may prevent microvascular damage in the diabetic retina.
Glomerular and tubulointerstitial macrophage infiltration has also been demonstrated in human progressive diabetic nephropathy, and the interstitial macrophage infiltration is strongly correlated with serum creatinine, proteinuria and interstitial fibrosis.74 In addition to the association between renal inflammation and features of diabetic nephropathy, several studies have evaluated the effects of anti-inflammatory agents on the prevention of diabetic kidney disease. Diabetes-induced glomerular macrophage infiltration, ICAM-1 expression and NF-κB activation associated with albuminuria, mesangial expansion, glomerular hypertrophy and glomerular expression of collagen and TGF-β in the kidney have been partly or completely prevented by anti-inflammatory agents, such as mycophenolate mofetil, erythromycin or methotrexate.75, 76, 77 Prevention of diabetes-induced renal inflammation and associated renal damage in ICAM-1- or MCP-1-deficient animals provides further evidence of the participation of the inflammatory process in the pathogenesis of diabetic nephropathy.78, 79
Several lines of evidence have been shown to support the concept that a local inflammatory process is involved in the pathogenesis of DR.80 Studies in humans using material from a vitrectomy or post-mortem specimens from diabetic donors have demonstrated an association between inflammatory markers and the presence of DR. An increased expression of ICAM-1, which facilitates the trafficking of leukocytes into the retina, has been demonstrated in retinal blood vessels of post-mortem specimens from diabetic donors81 and also in vitreous tissue of diabetic patients.82 Concordantly, elevation in the intra-vitreous levels of two pro-inflammatory cytokines Interleukin-8 and MCP-1 was observed in diabetic patients with proliferative DR compared with non-diabetic subjects.83 Interestingly, intra-vitreous levels of cytokines were correlated with proliferative DR activity, suggesting that these cytokines may be pathogenically important in proliferative DR.83
To this end, our group demonstrated that the presence of Ectodermal dysplasia 1 (ED-1)-positive cells in the retina, which indicate microglial activation, is increased in SHR rats, mainly in diabetic SHR rats, and this increment was accompanied by an increase in ICAM-1 and the NF-κB p65 transcription factor.84
Inflammation and oxidative stress as the underlying mechanism for the interaction between hypertension and DR and nephropathy
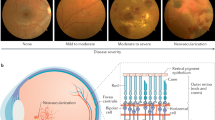
Both hypertension and diabetes induce oxidative stress and inflammation, which, in turn, contribute to diabetic nephropathy and retinopathy (Figure 1). Oxidative stress and inflammation are closely related events. The generation of reactive species by the inflammatory cells can induce oxidative stress. On the other hand, oxidative stress may induce inflammation through NF-κB-mediated pro-inflammatory gene expression.85 This intricate relationship between inflammation and oxidative stress is not just a theoretical link. Rather, numerous studies have demonstrated the simultaneous presence of inflammation and oxidative stress in an inseparable manner in different organs, particularly in the kidney and retina.80, 86
Schematic representation of mechanisms whereby diabetes mellitus and hypertension may induce diabetic nephropathy and retinopathy. Green indicates mechanisms related to the pathophysiology of nephropathy; pink indicates mechanisms related to the pathophysiology of retinopathy; and orange indicates the mechanisms equally important for both nephropathy and retinopathy. AGE, advanced glycation end-products; BRB, blood retinal barrier; ECM, extracellular matrix; GLUT-1, glucose transporter-1; ICAM, intercellular adhesion molecule; NADPH oxidase, nicotinamide adenine dinucleotide phosphate-oxidase; NF-κB, nuclear factor-kappa B; RAGE, receptor for advanced glycation end-products; ROS, reactive oxygen species; TGF-β, transforming growth factor-beta; VCAM, vascular cellular adhesion molecule; VEGF, vascular endothelial growth factor. A full color version of this figure is available at the Hypertension Research journal online.
The interaction between hypertension and diabetes with inflammation and oxidative stress as a mechanism for renal and retinal disease has been assessed in SHR and WKY rats with streptozotocin-induced DM. In these models, it has been shown that the renal hypertrophy and albumin excretion rate, along with glomerular and tubulointerstitial macrophage infiltrations, was significantly increased only 10 days after the induction of STZ-induced diabetes in both normotensive WKY and hypertensive SHRs.87 However, nitrotyrosine and 8-OHdG, which are markers of oxidative stress-induced protein and DNA modifications, respectively, were elevated only in hypertensive diabetic rats (diabetic SHRs) after 10 days of induction of diabetes.87 This elevated renal cortical oxidative stress was associated with increased superoxide generation through NADPH oxidase and decreased antioxidant defense in hypertensive diabetic rats. In normotensive diabetic rats, although superoxide generation increased 10 days after of induction of diabetes (but not as much as in hypertensive diabetic rats), antioxidant defenses were intact, and the overall level of renal cortical oxidative stress did not increase, as evidenced by the levels of nitrotyrosine and 8-OHdG. These findings clearly demonstrate that the presence of hypertension has a profound role in the elevation of renal cortical oxidative stress in the very early stages of diabetes.87 However, long-term diabetes alone is sufficient to increase renal oxidative stress, as shown in many different studies.67, 88, 89 Interestingly, it has been shown that tempol, an antioxidant mimetic of superoxide dismutase, is renal protective when administered to diabetic SHRs. In STZ-induced DM SHRs, the use of tempol for 20 days normalized renal cortical extracellular superoxide dismutase expression and decreased superoxide generation and oxidative stress-induced DNA damage. Along with the reversal of the redox balance, tempol treatment improved renal functional and structural alterations, as evidenced by decreased albumin excretion rate and renal cortical expression of collagen IV.90 In a different study, it was found that long-term green tea consumption ameliorates renal injury in hypertensive diabetic rats by inhibiting oxidative stress through downregulation of Nox4 expression.91
The presence of hypertension exacerbates retinopathy in diabetic animals. We have demonstrated that the concomitance of both diabetes and hypertension leads to earlier and more pronounced retinal inflammatory alterations characterized by increased expression levels of ICAM-1, VEGF and NF-κB p65 in the retina when compared with those of normotensive diabetic rats.92 Further corroborating the contribution of hypertension to retinal damage in diabetic SHR, we have demonstrated that anti-hypertensive treatment either with losartan (an AT1 receptor blocker, ARB) or with triple therapies (hydralazine, reserpine and hydrochlorothiazide) abolished these effects.84
In the retina, a number of studies have shown that there is an increase in oxidative markers after the induction of DM,67, 93, 94, 95 but the concomitance of diabetes and hypertension evoked earlier oxidative retinal damage characterized by an increase in nitrotyrosine and 8-OHdG in retinal tissue from short-term STZ-induced DM in SHRs.96, 97, 98, 99 These oxidative markers were the consequence of an increase in superoxide production and depletion of the gluthatione-reduced antioxidant system in the retinal tissue.96, 97, 98 Interestingly, the administration of a superoxide dismutase mimetic tempol to diabetic SHRs re-established the redox status and improved early molecular markers of DR.97 In these models, with long-term diabetes (12 weeks of duration), we have demonstrated that hypertension could account for exacerbation of retinopathy in diabetic SHRs. It was observed that higher levels of apoptotic neural cells were found in retinas from diabetic SHR, accompanied by increased glial reaction, both of which are accepted as early markers of DR. The oxidative imbalance, evaluated through increased superoxide production and decreased gluthatione levels, was accentuated in diabetic SHR compared with normotensive diabetic controls. As a consequence, the levels of nitrotyrosine and 8-OHdG were elevated mainly in diabetic SHRs. In addition, the mitochondrial integrity was evaluated through expression of B-cell lymphoma 2 and uncoupling protein-2, which are mitochondrial proteins involved in cytochrome c release, apoptosis, mitochondrial energy metabolism and neurodegeneration. The presence of diabetes dramatically affected these mitochondrial parameters. Treatment with losartan re-established all of the above-mentioned parameters.98 An AT1 receptor blockade with losartan also reduces retinal damage by decreasing inflammation and oxidative stress by means of a reduction in retinal inducible nitric oxide.99 These findings indicate that the blockage of RAS exerts a variety of effects, including antioxidant, anti-apoptotic and anti-inflammatory properties.98, 99, 100
Collectively, the above findings suggest that oxidative stress and inflammation may be common denominators of kidney and retinal damage in the concomitant presence of diabetes and hypertension (Figure 1).
Role of renin–Ang aldosterone system with emphasis on aldosterone, Ang-converting enzyme 2 and prorenin in DR and nephropathy
The link between hypertension and the pathogenesis and progression of DR and nephropathy most likely involves the renin–Ang aldosterone system (RAAS). A major role of local RAAS in the development and progression of diabetic nephropathy and retinopathy has been clearly demonstrated and reviewed elsewhere.101, 102 In addition, several observational and clinical trials have demonstrated that interference of the RAAS with Ang-converting enzyme (ACE) inhibitors or using ARB may be effective in the treatment of diabetic nephropathy and retinopathy. It has been shown that in type 1 diabetic patients, use of an ACE inhibitor was able to prevent end-stage renal disease and decrease the incidence of overt nephropathy (defined as development of persistent macroalbuminuria) with preserved renal function.103 However, a more recent trial has shown that use of ACE inhibitors was unable to prevent the development of microalbuminuria in normoalbuminuric type 1 diabetic patients.104 Two large placebo-controlled studies, DR Candersartan Trial (DIRECT)-Prevent 1 and DIRECT-Protect 1, failed to detect any protective effect against the development of microalbuminuria over a median of 4.7 years of treatment with the ARB candesartan in 3326 patients with type 1 diabetes.105 In type 2 diabetic patients, the use of an ACE inhibitor was associated with a reduction in the incidence of macroalbuminuria106 and the development of microalbuminuria in hypertensive normoalbuminuric patients.107 In type 2 diabetic patients, the first formal demonstration that overt diabetic nephropathy can be prevented using RAAS inhibition was provided using an AT1-R blocker, irbesartan.108 In this study, 590 hypertensive patients with type 2 diabetes and microalbuminuria were allocated to receive irbesartan 150 mg, either 300 mg per day or placebo. After a median of 2 years of treatment with 300 mg per day of the ARB irbesartan, there was a threefold reduction in the incidence of macroalbuminuria compared with placebo.108 In type 2 diabetic patients, use of an ARB, either losartan109 or irbesartan,110 versus placebo decreased the incidence of a composite end point of doubling of serum creatinine concentration, end-stage renal disease or death by 16 and 19%, respectively. The RAS study was designed to assess, over a 5-year period, the effect of a RAAS blockade with either an ACE inhibitor (enalapril) or an ARB (losartan) compared with placebo on both renal and retinal morphological features in normotensive patients with type 1 DM before the onset of albuminuria. Of 285 patients enrolled, 256 (90%) had renal percutaneous biopsies, and 223 (83%) had dilated seven standard-field Early Treatment Diabetic Retinopathy Study (ETDRS) photographs at baseline and at 5 years. No significant differences were observed in renal outcomes after 5 years. The odds of progression of DR by two or more steps was reduced by 65% with enalarpil (odds ratio 0.35, confidence interval 0.14–0.85) and 70% with losartan (odds ratio 0.30, confidence interval 0.12–0.73), independent of changes in blood pressure or glycemic control.104 The DIRECT enrolled 1905 individuals with type 2 DM and 3326 individuals with type 1 DM and followed them over a median of approximately 5 years.105, 111 Three outcomes were evaluated in this trial: the development of DR in type 1 DM (DIRECT-Prevent 1), the progression of DR in type 1 (DIRECT-Protect 1) and the progression of DR in type 2 DM (DIRECT-Protect 2). The groups did not differ in HbA1c levels, and treatment with candesartan resulted in a small but significant reduction in blood pressure. There was no beneficial effect of candesartan in the development of DR (two or more step progression) in patients with type 1 DM without baseline retinopathy (DIRECT-Prevent 1). However, when evaluating for a three-step or more progression of DR, a 35% relative risk reduction was observed with candesartan treatment (P=0.0034). DIRECT-Protect 1 and DIRECT-Protect 2 failed to show any significant differences in the prevention of DR progression. However, in DIRECT-Protect 2, a greater regression of established DR was observed with candesartan treatment (19%) than without (14%; P<0.009). The RASS and DIRECT studies demonstrated that treatment with either an ACE inhibitor or an ARB may result in a beneficial effect on DR in patients with type 1 or type 2 DM and that these effects may be independent of blood pressure lowering.
Next, we would like to focus on recent findings in the RAAS, specifically those concerning aldosterone, ACE2, prorenin and its recent cloned receptor.
Recent clinical trials have suggested that adding an inhibitor of the aldosterone system to an ACE inhibitor-based regime in patients with diabetic nephropathy may further reduce proteinuria and thereby afford additional renal protection.112 The beneficial effects of aldosterone blockade on diabetic nephropathy seem to be independent of the blood pressure reduction112 and RAAS blockade on diabetic nephropathy.113 The mechanism by which an aldosterone blocker improves diabetic nephropathy is not totally clear; however, anti-inflammatory114, 115 and/or anti-oxidant116 mechanisms have been suggested as possibilities. To this end, it has been shown that the induction of diabetes in rats leads to a reduction in glucose-6-phosphate dehydrogenase (G6PD) and oxidative stress.117 A reduction in G6PD activity leads to decreased NADPH and makes cells very sensitive to oxidant damage.118, 119 Deficiency in G6PD activity may lead not only to oxidative stress but also to nephropathy, as it has been shown that G6PD knockout mice have increased renal oxidative stress and elevated albuminuria.120 Finally, it has been shown that aldosterone induces a G6PD-deficient phenotype that can be improved by aldosterone antagonist or gene transfer of G6PD.121 Of great interest, it has recently been suggested that, similar to what has been described in the heart, brain and vasculature,122 the eye has a dynamic aldosterone-mineralocorticoid receptor system that has an important pathophysiological role in the development of retinal pathology. Wilkinson-Berka et al.123, using a rat model of oxygen-induced retinopathy (OIR) that has features of premature retinopathy in humans with neovascularization, have shown that mineralocorticoid receptor antagonism with spironolactone improves retinal angiogenesis by attenuating leukostasis and decreasing proinflamatory responses. These beneficial effects of spironolactone were attributed, at least in part, to a reduction in oxidative stress. Further observations in cultured retinal cells and in vivo in OIR suggested that reduction in oxidative stress may be secondary to a decrease in G6PD and retinal NADPH oxidase subunit Nox4 (Wilkinson-Berka et al.123). Together, these observations suggest that mineralocorticoid receptor antagonism may improve diabetic nephropathy and retinopathy by restoring G6PD activity and consequently reducing oxidative stress and inflammation. These experimental observations, however, need to be assessed in clinical settings.
Recent observations suggest that ACE2 is important in various pathophysiological conditions, including hypertension124 and diabetic nephropathy.125 The presence of the enzymatic activity of ACE2 has also been described in the rodent126 and the porcine127 retina; however, the pathophysiological significance of ACE2 in the retina has not yet been established.
ACE2 is part of the enzymatic cascade of RAAS. Specifically, it seems to act as a negative regulator of the RAAS, counterbalancing the function of ACE.128 The main product of ACE2 is Ang 1–7, which is now recognized as a vasodilatory peptide.128 ACE2 converts Ang I to Ang 1–9 and subsequently forms Ang 1–7 from Ang 1–9. ACE2 is also able to convert Ang II directly to Ang 1–7 (Burrell et al.128). Additionally, Ang 1–7 has its own receptor, Mas. As Ang 1–7 is a vasodilator, the overall effects that appears to be induced by ACE2 counterbalance many of the effects of ACE.
ACE2 is expressed in a variety of tissues, although its distribution is much less widespread than that of ACE. Sites of ACE2 expression, as well as that of its specific receptor Mas, include the heart and kidney.128 In the kidney, ACE2 has a distribution similar to ACE, with major localization in renal tubules.128, 129, 130
It has been suggested that both mRNA and protein expression of ACE2 are reduced in kidney biopsy specimens from type 2 diabetic patients with nephropathy compared with normal controls or patients with other renal disease.131 In another study, ACE2 levels in kidney biopsy of diabetic patients were reduced only at the protein level, not at the mRNA level, and only in the tubulointerstitial region, not in the glomeruli, compared with non-diabetic controls.132 In experimental models of diabetes-associated kidney disease, it has been shown that ACE2 may be reduced129 or elevated.133 Reasons for this discrepancy may be attributed to the duration of diabetes and different methodology in assessing ACE2, gene or protein expression, or their activity. Indeed, it has been suggested that the early phases are characterized by high levels of ACE2 (Wysocki et al.133) whereas a longer duration of diabetes (24 weeks) is associated with a reduction in ACE2 expression.129 A recent study using ACE2-knockout mice with streptozotocin-induced diabetes has shown that these mice displayed early and more severe features of diabetic nephropathy than wild-type diabetic mice.134 Further evidence of the importance of ACE2 on experimental diabetic nephropathy was obtained by the observation that the administration of human recombinant ACE2 to diabetic mice attenuates diabetic kidney injury in association with a reduction in blood pressure and a decrease in NADPH oxidase activity.125 In vitro studies suggested that the protective effect of human recombinant ACE2 is due to a reduction in Ang II and an increase in Ang 1–7 signaling.125 Further studies are still necessary to establish whether ACE2 represents a novel target for diabetic nephropathy and perhaps also for DR.
Discovery of the (pro)renin receptor ((P)RR) in the human kidney in 2002 (Nguyen et al.135) led to the concept that prorenin, previously considered an inactive precursor of renin, could be involved in different pathological conditions, including DR and nephropathy.135, 136, 137, 138, 139 Renal juxtaglomerular cells are the only known site of renin production, whereas the kidney and a number of extrarenal tissues, including the adrenal gland, ovary, testis, placenta and retina, produce prorenin.140, 141, 142 Plasma prorenin concentrations are 10–20-fold higher than those of rennin.143 The (P)RR binds both renin and prorenin and is reported to increase the catalytic efficiency of renin and activate prorenin.135 Thus, binding of renin and prorenin not only stimulates the (P)RR, but also increases Ang II formation, leading to AT1 receptor stimulation.139 Ichihara et al.144 have demonstrated that the administration of synthetic handle region peptides—which interfere with the binding of prorenin to its receptor—to diabetic rats completely prevented the development of nephropathy. The same group has shown that administration of handle region peptides to diabetic rats can also cause regression of established diabetic nephropathy.145 In cultured mesangial cells, it has been suggested that high glucose-induced prorenin upregulation leads to the production of inflammatory cytokines, such as interleukin-1β and cyclooxygenase-2 (Huang and Siragy146). In the retinal vasculature of diabetic mice, Satofuka et al.147 have shown that handle region peptides, also called (P)RR blockers (PRRBs), reduced leukostasis to a greater extent than the AT1-R blocker losartan in wild-type mice and also reduced leukostasis in AT1A receptor gene knockout mice. Furthermore, PRRB reduced the diabetes-induced elevation in retinal expression of phosphorylated extracellular signal-regulated kinases 1/2 in AT1A receptor gene knockout mice. The finding that PRRB reduced phosphorylated extracellular signal-regulated kinases 1/2 expression in cultured brain capillary endothelial cells stimulated with prorenin, but not in cells stimulated with angiotenin II, was consistent with a specific effect of PRRB on the actions of prorenin. Recently, Wilkinson-Berka et al.136 have reported that a (P)RR antagonist—RILLKKMPSV—clearly exhibited antiangiogenic and anti-inflammatory effects in a model of OIR. However, (P)RR antagonist also compromised the electroretinogram in shams and OIR and increased phosphorylated extracellular signal-regulated kinases 1/2 immunolabeling in shams but not OIR. These results hold promise for using PRRB as a new therapeutic intervention in diabetic nephropathy and retinopathy, although more research in this field is clearly required.
Concluding remarks
Blood glucose control, normalization of arterial pressure and use of drugs that interfere in the RAAS are efficient approaches for the prevention and treatment of diabetic nephropathy and retinopathy. Experimental and clinical studies have demonstrated that these maneuvers are also able to reduce superoxide generation and/or increase anti-oxidant defenses.98, 99, 148, 149 In this review, we have provided evidence that local generation of oxidative stress and inflammation can be a common mechanism of renal and retinal lesion in the presence of diabetes and hypertension. Although efficient, blood glucose control, normalization of arterial pressure and drugs that interfere in RAAS are currently unable to completely arrest diabetic renal and retinal diseases.150, 151, 152 It has been suggested that a multifactorial approach, including lifestyle modification, blood glucose control, blood pressure normalization, and the use of drugs that interfere with the RAS, statin and an anti-oxidant, vitamin E (alpha tocoferol), is more efficient than standard treatment to slow the progression of diabetic nephropathy and retinopathy.153, 154 The effect of this multifactorial approach in oxidative stress was not investigated. In type 2 diabetic patients, a glass of red wine (118 ml, 4-oz) per day was associated with reduction of proteinura and urinary 8-OHdG, a marker of oxidative stress, which was not observed in the placebo group or in patients who received white wine.155 Consumption of dark chocolate is associated with blood pressure reduction associated with a reduction in oxidative stress and increase in nitric oxide bioavailability,156 both abnormalities that have been involved in the pathogenesis of diabetic nephropathy and retinopathy.67, 85, 86, 87, 88, 93, 94, 95, 97, 98, 99, 156 Finally, in experimental diabetes, the use of green tea was associated with an improvement in diabetic nephropathy and retinopathy and associated with a reduction in oxidative stress.91, 157 The usefulness of these dietary approaches in diabetic patients with hypertension and nephropathy or retinopathy that are on standard treatment deserves to be tested in larger clinical trials. Finally, a better understanding and identification of the basic mechanism of action of different antioxidants in diabetes can lead to better optimization of renal and retinal protection.
References
Anonymous . National High Blood Pressure Education Program Working Group report on hypertension in diabetes. Hypertension 1994; 23: 145–158.
Geiss LS, Rolka DB, Engelgau MM . Elevated blood pressure among U.S. adults with diabetes, 1988–1994. Am J Prev Med 2002; 22: 42–48.
Jensen T, Borch-Johnsen K, Deckert T . Changes in blood pressure and renal function in patients with type I (insulin-dependent) diabetes mellitus prior to clinical diabetic nephropathy. Diabetes Res 1987; 4: 159–162.
Parving HH . Controlling hypertension in diabetes. Acta Diabetol 2002; 39: S35–S40.
Tarnow L, Rossing P, Gall MA, Nielsen FS, Parving HH . Prevalence of arterial hypertension in diabetic patients before and after the JNC-V. Diabetes Care 1994; 17: 1247–1251.
Stratton IM, Cull CA, Adler AI, Matthews DR, Neil HA, Holman RR . Additive effects of glycaemia and blood pressure exposure on risk of complications in type 2 diabetes: a prospective observational study (UKPDS 75). Diabetologia 2006; 49: 1761–1769.
The UKPDS Study Group. Tight blood pressure control and risk of macrovascular and microvascular complications in type 2 diabetes: UKPDS 38. UK Prospective Diabetes Study Group. BMJ 1998; 317: 703–713.
Zoungas S, de Galan BE, Ninomiya T, Grobbee D, Hamet P, Heller S, MacMahon S, Marre M, Neal B, Patel A, Woodward M, Chalmers J, Cass A, Glasziou P, Harrap S, Lisheng L, Mancia G, Pillai A, Poulter N, Perkovic V, Travert F, ADVANCE Collaborative Group. Combined effects of routine blood pressure lowering and intensive glucose control on macrovascular and microvascular outcomes in patients with type 2 diabetes: new results from the ADVANCE trial. Diabetes Care 2009; 32: 2068–2074.
Giunti S, Barit D, Cooper ME . Mechanisms of diabetic nephropathy: role of hypertension. Hypertension 2006; 48: 519–526.
Schrier RW, Estacio RO, Esler A, Mehler P . Effects of aggressive blood pressure control in normotensive type 2 diabetic patients on albuminuria, retinopathy and strokes. Kidney Int 2002; 61: 1086–1097.
Thomas MC, Atkins RC . Blood pressure lowering for the prevention and treatment of diabetic kidney disease. Drugs 2006; 66: 2213–2234.
Klein R, Knudtson MD, Lee KE, Gangnon R, Klein BE . The Wisconsin Epidemiologic Study of Diabetic Retinopathy: XXII the twenty-five-year progression of retinopathy in persons with type 1 diabetes. Ophthalmology 2008; 115: 1859–1868.
Klein R, Lee KE, Gangnon RE, Klein BE . The 25-year incidence of visual impairment in type 1 diabetes mellitus the wisconsin epidemiologic study of diabetic retinopathy. Ophthalmology 2010; 117: 63–70.
Rodrigues TC, Canani LH, Viatroski RS, Hoffmann LH, Esteves JF, Gross JL . Masked hypertension, nocturnal blood pressure and retinopathy in normotensive patients with type 1 diabetes. Diabetes Res Clin Pract 2010; 87: 240–245.
Körner A, Jaremko G, Eklöf AC, Aperia A . Rapid development of glomerulosclerosis in diabetic Dahl salt-sensitive rats. Diabetologia 1997; 40: 367–373.
Mauer SM, Steffes MW, Azar S, Sandberg SK, Brown DM . The effects of Goldblatt hypertension on development of the glomerular lesions of diabetes mellitus in the rat. Diabetes 1978; 27: 738–744.
Wakisaka M, Nunoi K, Iwase M, Kikuchi M, Maki Y, Yamamoto K, Sadoshima S, Fujishima M . Early development of nephropathy in a new model of spontaneously hypertensive rat with non-insulin-dependent diabetes mellitus. Diabetologia 1988; 31: 291–296.
Cooper ME, Allen TJ, O'Brien RC, Macmillan PA, Clarke B, Jerums G, Doyle AE . Effects of genetic hypertension on diabetic nephropathy in the rat--functional and structural characteristics. J Hypertens 1988; 6: 1009–1016.
Dosso AA, Leuenberger PM, Rungger-Brändle E . Remodeling of retinal capillaries in the diabetic hypertensive rat. Invest Ophthalmol Vis Sci 1999; 40: 2405–2410.
Hammes HP, Brownlee M, Edelstein D, Saleck M, Martin S, Federlin K . Aminoguanidine inhibits the development of accelerated diabetic retinopathy in the spontaneous hypertensive rat. Diabetologia 1994; 37: 32–35.
Gin T, Joon TL, Panagiotopoulos S, Cooper M, Taylor H, Jerums G . Organ specificity of antihypertensive therapy on ocular albumin vascular clearance and albuminuria in the hypertensive diabetic rat. Invest Ophthalmol Vis Sci 1996; 37: 281–289.
Righetti AE, Boer-Lima PA, Lopes de Faria JB . The presence of genetic hypertension stimulates early renal accumulation of fibronectin in experimental diabetes mellitus. Diabetologia 2001; 44: 2088–2091.
Lopes de Faria JM, Silva KC, Boer PA, Cavalcanti TC, Rosales MA, Ferrari AL, Lopes de Faria JB . A decrease in retinal progenitor cells is associated with early features of diabetic retinopathy in a model that combines diabetes and hypertension. Mol Vis 2008; 14: 1680–1691.
Wolf G . Cell cycle regulation in diabetic nephropathy. Kidney Int 2000; 77: S59–S66.
Wolf G, Schroeder R, Thaiss F, Ziyadeh FN, Helmchen U, Stahl RA . Glomerular expression of p27Kip1 in diabetic db/db mouse: role of hyperglycemia. Kidney Int 1998; 53: 869–879.
Wolf G, Schanze A, Stahl RA, Shankland SJ, Amann K . p27(Kip1) knockout mice are protected from diabetic nephropathy: evidence for p27(Kip1) haplotype insufficiency. Kidney Int 2005; 68: 1583–1589.
Silveira LA, Bacchi CE, Pinto GA, De Faria JB . The genetics of hypertension modifies the renal cell replication response induced by experimental diabetes. Diabetes 2002; 51: 1529–1534.
Carmines PK . The renal vascular response to diabetes. Curr Opin Nephrol Hypertens 2010; 19: 85–90.
Gruden G, Zonca S, Hayward A, Thomas S, Maestrini S, Gnudi L, Viberti GC . Mechanical stretch-induced fibronectin and transforming growth factor-beta1 production in human mesangial cells is p38 mitogen-activated protein kinase-dependent. Diabetes 2000; 49: 655–661.
Rassam SM, Patel V, Kohner EM . The effect of experimental hypertension on retinal vascular autoregulation in humans: a mechanism for the progression of diabetic retinopathy. Exp Physiol 1995; 80: 53–68.
Aiello LP, Avery RL, Arrigg PG, Keyt BA, Jampel HD, Shah ST, Pasquale LR, Thieme H, Iwamoto MA, Park JE, Nguyen HV, Aiello LM, Ferrara N, King GL . Vascular endothelial growth factor in ocular fluid of patients with diabetic retinopathy and other retinal disorders. N Engl J Med 1994; 331: 1480–1487.
Seko Y, Seko Y, Fujikura H, Pang J, Tokoro T, Shimokawa H . Induction of vascular endothelial growth factor after application of mechanical stress to retinal pigment epithelium of the rat in vitro. Invest Ophthalmol Vis Sci 1999; 40: 3287–3291.
Suzuma I, Hata Y, Clermont A, Pokras F, Rook SL, Suzuma K, Feener EP, Aiello LP . Cyclic stretch and hypertension induce retinal expression of vascular endothelial growth factor and vascular endothelial growth factor receptor-2: potential mechanisms for exacerbation of diabetic retinopathy by hypertension. Diabetes 2001; 50: 444–454.
Suzuma I, Suzuma K, Ueki K, Hata Y, Feener EP, King GL, Aiello LP . Stretch-induced retinal vascular endothelial growth factor expression is mediated by phosphatidylinositol 3-kinase and protein kinase C (PKC)-zeta but not by stretch-induced ERK1/2, Akt, Ras, or classical/novel PKC pathways. J Biol Chem 2002; 277: 1047–1057.
Suzuma I, Murakami T, Suzuma K, Kaneto H, Watanabe D, Ojima T, Honda Y, Takagi H, Yoshimura N . Cyclic stretch-induced reactive oxygen species generation enhances apoptosis in retinal pericytes through c-jun NH2-terminal kinase activation. Hypertension 2007; 49: 347–354.
Beltramo E, Berrone E, Giunti S, Gruden G, Perin PC, Porta M . Effects of mechanical stress and high glucose on pericyte proliferation, apoptosis and contractile phenotype. Exp Eye Res 2006; 83: 989–994.
Mizutani M, Kern TS, Lorenzi M . Accelerated death of retinal microvascular cells in human and experimental diabetic retinopathy. J Clin Invest 1996; 97: 2883–2890.
Pfister F, Feng Y, vom Hagen F, Hoffmann S, Molema G, Hillebrands JL, Shani M, Deutsch U, Hammes HP . Pericyte migration: a novel mechanism of pericyte loss in experimental diabetic retinopathy. Diabetes 2008; 57: 2495–2502.
Yasuda T, Kondo S, Homma T, Harris RC . Regulation of extracellular matrix by mechanical stress in rat glomerular mesangial cells. J Clin Invest 1996; 98: 1991–2000.
Cortes P, Zhao X, Riser BL, Narins RG . Role of glomerular mechanical strain in the pathogenesis of diabetic nephropathy. Kidney Int 1997; 51: 57–68.
Riser BL, Ladson-Wofford S, Sharba A, Cortes P, Drake K, Guerin CJ, Yee J, Choi ME, Segarini PR, Narins RG . TGF-beta receptor expression and binding in rat mesangial cells: modulation by glucose and cyclic mechanical strain. Kidney Int 1999; 56: 428–439.
Riser BL, Denichilo M, Cortes P, Baker C, Grondin JM, Yee J, Narins RG . Regulation of connective tissue growth factor activity in cultured rat mesangial cells and its expression in experimental diabetic glomerulosclerosis. J Am Soc Nephrol 2000; 11: 25–38.
Giunti S, Pinach S, Arnaldi L, Viberti G, Perin PC, Camussi G, Gruden G . The MCP-1/CCR2 system has direct proinflammatory effects in human mesangial cells. Kidney Int 2006; 69: 856–863.
Gruden G, Setti G, Hayward A, Sugden D, Duggan S, Burt D, Buckingham RE, Gnudi L, Viberti G . Mechanical stretch induces monocyte chemoattractant activity via an NF-kappaB-dependent monocyte chemoattractant protein-1-mediated pathway in human mesangial cells: inhibition by rosiglitazone. J Am Soc Nephrol 2005; 16: 688–696.
Kuroiwa T, Lee EG, Danning CL, Illei GG, McInnes IB, Boumpas DT . CD40 ligand-activated human monocytes amplify glomerular inflammatory responses through soluble and cell-to-cell contact-dependent mechanisms. J Immunol 1999; 163: 2168–2175.
Mene P, Pugliese F, Cinotti GA . Adhesion of U-937 monocytes induces cytotoxic damage and subsequent proliferation of cultured human mesangial cells. Kidney Int 1996; 50: 417–423.
Gruden G, Thomas S, Burt D, Zhou W, Chusney G, Gnudi L, Viberti G . Interaction of angiotensin II and mechanical stretch on vascular endothelial growth factor production by human mesangial cells. J Am Soc Nephrol 1999; 10: 730–737.
Thomas S, Vanuystel J, Gruden G, Rodríguez V, Burt D, Gnudi L, Hartley B, Viberti G . Vascular endothelial growth factor receptors in human mesangium in vitro and in glomerular disease. J Am Soc Nephrol 2000; 11: 1236–1243.
Amemiya T, Sasamura H, Mifune M, Kitamura Y, Hirahashi J, Hayashi M, Saruta T . Vascular endothelial growth factor activates MAP kinase and enhances collagen synthesis in human mesangial cells. Kidney Int 1999; 56: 2055–2063.
Gnudi L, Viberti G, Raij L, Rodriguez V, Burt D, Cortes P, Hartley B, Thomas S, Maestrini S, Gruden G . GLUT-1 overexpression: link between hemodynamic and metabolic factors in glomerular injury? Hypertension 2003; 42: 19–24.
Heilig CW, Concepcion LA, Riser BL, Freytag SO, Zhu M, Cortes P . Overexpression of glucose transporters in rat mesangial cells cultured in a normal glucose milieu mimics the diabetic phenotype. J Clin Invest 1995; 96: 1802–1814.
Shankland SJ . The podocyte's response to injury: role in proteinuria and glomerulosclerosis. Kidney Int 2006; 69: 2131–2147.
White KE, Bilous RW . Structural alterations to the podocyte are related to proteinuria in type 2 diabetic patients. Nephrol Dial Transplant 2004; 19: 1437–1440.
Endlich N, Kress KR, Reiser J, Uttenweiler D, Kriz W, Mundel P, Endlich K . Podocytes respond to mechanical stress in vitro. J Am Soc Nephrol 2001; 12: 413–422.
Endlich N, Endlich K . Stretch, tension and adhesion- adaptive mechanisms of the actin cytoskeleton in podocytes. Eur J Cell Biol 2006; 85: 229–234.
Dessapt C, Baradez MO, Hayward A, Dei Cas A, Thomas SM, Viberti G, Gnudi L . Mechanical forces and TGFbeta1 reduce podocyte adhesion through alpha3beta1 integrin downregulation. Nephrol Dial Transplant 2009; 24: 2645–2655.
Patey N, Halbwachs-Mecarelli L, Droz D, Lesavre P, Noel LH . Distribution of integrin subunits in normal human kidney. Cell Adhes Commun 1994; 2: 159–167.
Ruoslahti E . Integrins. J Clin Invest 1991; 87: 1–5.
Durvasula RV, Shankland SJ . Activation of a local renin angiotensin system in podocytes by glucose. Am J Physiol Renal Physiol 2008; 294: F830–F839.
Durvasula RV, Petermann AT, Hiromura K, Blonski M, Pippin J, Mundel P, Pichler R, Griffin S, Couser WG, Shankland SJ . Activation of a local tissue angiotensin system in podocytes by mechanical strain. Kidney Int 2004; 65: 30–39.
Miceli I, Burt D, Tarabra E, Camussi G, Perin PC, Gruden G . Stretch reduces nephrin expression via an angiotensin II-AT(1)-dependent mechanism in human podocytes: effect of rosiglitazone. Am J Physiol Renal Physiol 2010; 298: F381–F390.
Doublier S, Salvidio G, Lupia E, Ruotsalainen V, Verzola D, Deferrari G, Camussi G . Nephrin expression is reduced in human diabetic nephropathy: evidence for a distinct role for glycated albumin and angiotensin II. Diabetes 2003; 52: 1023–1030.
Bonnet F, Cooper ME, Kawachi H, Allen TJ, Boner G, Cao Z . Irbesartan normalises the deficiency in glomerular nephrin expression in a model of diabetes and hypertension. Diabetologia 2001; 44: 874–877.
Amazonas RB, Sanita Rde A, Kawachi H, de Faria JB . Prevention of hypertension with or without renin-angiotensin system inhibition precludes nephrin loss in the early stage of experimental diabetes mellitus. Nephron Physiol 2007; 107: 57–64.
Gorin Y, Block K, Hernandez J, Bhandari B, Wagner B, Barnes JL, Abboud HE . Nox4 NAD(P)H oxidase mediates hypertrophy and fibronectin expression in the diabetic kidney. J Biol Chem 2005; 280: 39616–39626.
Susztak K, Raff AC, Schiffer M, Böttinger EP . Glucose-induced reactive oxygen species cause apoptosis of podocytes and podocyte depletion at the onset of diabetic nephropathy. Diabetes 2006; 55: 225–233.
Brownlee M . The pathobiology of diabetic complications: a unifying mechanism. Diabetes 2005; 54: 1615–1625.
Jones DP . Redefining oxidative stress. Antioxid Redox Signal 2006; 8: 1865–1879.
Halliwell B, Whiteman M . Measuring reactive species and oxidative damage in vivo and in cell culture: how should you do it and what do the results mean? Br J Pharmacol 2004; 142: 231–255.
Hinokio Y, Suzuki S, Hirai M, Chiba M, Hirai A, Toyota T . Oxidative DNA damage in diabetes mellitus: its association with diabetic complications. Diabetologia 1999; 42: 995–998.
Nishikawa T, Sasahara T, Kiritoshi S, Sonoda K, Senokuchi T, Matsuo T, Kukidome D, Wake N, Matsumura T, Miyamura N, Sakakida M, Kishikawa H, Araki E . Evaluation of urinary 8-hydroxydeoxy-guanosine as a novel biomarker of macrovascular complications in type 2 diabetes. Diabetes Care 2003; 26: 1507–1512.
Hinokio Y, Suzuki S, Hirai M, Suzuki C, Suzuki M, Toyota T . Urinary excretion of 8-oxo-7, 8-dihydro-2′-deoxyguanosine as a predictor of the development of diabetic nephropathy. Diabetologia 2002; 45: 877–882.
Wang Q, Pfister F, Dorn-Beineke A, vom Hagen F, Lin J, Feng Y, Hammes HP . Low-dose erythropoietin inhibits oxidative stress and early vascular changes in the experimental diabetic retina. Diabetologia 2010; 53: 1227–1238.
Furuta T, Saito T, Ootaka T, Soma J, Obara K, Abe K, Yoshinaga K . The role of macrophages in diabetic glomerulosclerosis. Am J Kidney Dis 1993; 21: 480–485.
Utimura R, Fujihara CK, Mattar AL, Malheiros DM, Noronha IL, Zatz R . Mycophenolate mofetil prevents the development of glomerular injury in experimental diabetes. Kidney Int 2003; 63: 209–216.
Tone A, Shikata K, Sasaki M, Ohga S, Yozai K, Nishishita S, Usui H, Nagase R, Ogawa D, Okada S, Shikata Y, Wada J, Makino H . Erythromycin ameliorates renal injury via anti-inflammatory effects in experimental diabetic rats. Diabetologia 2005; 48: 2402–2411.
Yozai K, Shikata K, Sasaki M, Tone A, Ohga S, Usui H, Okada S, Wada J, Nagase R, Ogawa D, Shikata Y, Makino H . Methotrexate prevents renal injury in experimental diabetic rats via anti-inflammatory actions. J Am Soc Nephrol 2005; 16: 3326–3338.
Okada S, Shikata K, Matsuda M, Ogawa D, Usui H, Kido Y, Nagase R, Wada J, Shikata Y, Makino H . Intercellular adhesion molecule-1-deficient mice are resistant against renal injury after induction of diabetes. Diabetes 2003; 52: 2586–2593.
Chow FY, Nikolic-Paterson DJ, Ozols E, Atkins RC, Rollin BJ, Tesch GH . Monocyte chemoattractant protein-1 promotes the development of diabetic renal injury in streptozotocin-treated mice. Kidney Int 2006; 69: 73–80.
Kern TS . Contributions of inflammatory processes to the development of the early stages of diabetic retinopathy. Exp Diabetes Res 2007; 2007: 95103.
McLeod DS, Lefer DJ, Merges C, Lutty GA . Enhanced expression of intracellular adhesion molecule-1 and P-selectin in the diabetic human retina and choroid. Am J Pathol 1995; 147: 642–653.
Limb GA, Hickman-Casey J, Hollifield RD, Chignell AH . Vascular adhesion molecules in vitreous from eyes with proliferative diabetic retinopathy. Invest Ophthalmol Vis Sci 1999; 40: 2453–2457.
Hernández C, Segura RM, Fonollosa A, Carrasco E, Francisco G, Simó R . Interleukin-8, monocyte chemoattractant protein-1 and IL-10 in the vitreous fluid of patients with proliferative diabetic retinopathy. Diabet Med 2005; 22: 719–722.
Silva KC, Pinto CC, Biswas SK, Souza DS, de Faria JB, de Faria JM . Prevention of hypertension abrogates early inflammatory events in the retina of diabetic hypertensive rats. Exp Eye Res 2007; 85: 123–129.
Calcutt NA, Cooper ME, Kern TS, Schmidt AM . Therapies for hyperglycaemia-induced diabetic complications: from animal models to clinical trials. Nat Rev Drug Discov 2009; 8: 417–429.
Vaziri ND, Rodriguez-Iturbe B . Mechanisms of disease: oxidative stress and inflammation in the pathogenesis of hypertension. Nat Clin Pract Nephrol 2006; 2: 582–593.
Biswas SK, Peixoto EB, Souza DS, de Faria JB . Hypertension increases pro-oxidant generation and decreases antioxidant defense in the kidney in early diabetes. Am J Nephrol 2008; 28: 133–142.
DeRubertis FR, Craven PA, Melhem MF, Salah EM . Attenuation of renal injury in db/db mice overexpressing superoxide dismutase: evidence for reduced superoxide-nitric oxide interaction. Diabetes 2004; 53: 762–768.
Fujita H, Fujishima H, Chida S, Takahashi K, Qi Z, Kanetsuna Y, Breyer MD, Harris RC, Yamada Y, Takahashi T . Reduction of renal superoxide dismutase in progressive diabetic nephropathy. J Am Soc Nephrol 2009; 20: 1303–1313.
Peixoto EB, Pessoa BS, Biswas SK, Lopes de Faria JB . Antioxidant SOD mimetic prevents NADPH oxidase-induced oxidative stress and renal damage in the early stage of experimental diabetes and hypertension. Am J Nephrol 2009; 29: 309–318.
Ribaldo PD, Souza DS, Biswas SK, Block K, Lopes de Faria JM, Lopes de Faria JB . Green tea (Camellia sinensis) attenuates nephropathy by downregulating Nox4 NADPH oxidase in diabetic spontaneously hypertensive rats. J Nutr 2009; 139: 96–100.
Silva KC, Pinto CC, Biswas SK, de Faria JB, de Faria JM . Hypertension increases retinal inflammation in experimental diabetes: a possible mechanism for aggravation of diabetic retinopathy by hypertension. Curr Eye Res 2007; 32: 533–541.
Madsen-Bouterse SA, Kowluru RA . Oxidative stress and diabetic retinopathy: pathophysiological mechanisms and treatment perspectives. Rev Endocr Metab Disord 2008; 9: 315–327.
Kowluru RA, Odenbach S . Effect of long-term administration of alpha-lipoic acid on retinal capillary cell death and the development of retinopathy in diabetic rats. Diabetes 2004; 53: 3233–3238.
Du Y, Smith MA, Miller CM, Kern TS . Diabetes-induced nitrative stress in the retina, and correction by aminoguanidine. J Neurochem 2002; 80: 771–779.
Pinto CC, Silva KC, Biswas SK, Martins N, De Faria JB, De Faria JM . Arterial hypertension exacerbates oxidative stress in early diabetic retinopathy. Free Radic Res 2007; 41: 1151–1158.
Rosales MA, Silva KC, Lopes de Faria JB, Lopes de Faria JM . Exogenous SOD mimetic tempol ameliorates the early retinal changes restablishing the redox status in diabetic hypertensive rats. Invest Ophthalmol Vis Sci 2010; 51: 4327–4336.
Silva KC, Rosales MA, de Faria JB, de Faria JM . Reduction of inducible nitric oxide synthase via angiotensin receptor blocker prevents the oxidative retinal damage in diabetic hypertensive rats. Curr Eye Res 2010; 35: 519–528.
Silva KC, Rosales MA, Biswas SK, Lopes de Faria JB, Lopes de Faria JM . Diabetic retinal neurodegeneration is associated with mitochondrial oxidative stress and is improved by an angiotensin receptor blocker in a model combining hypertension and diabetes. Diabetes 2009; 58: 1382–1390.
Jung KH, Chu K, Lee ST, Kim SJ, Song EC, Kim EH, Park DK, Sinn DI, Kim JM, Kim M, Roh JK . Blockade of AT1 receptor reduces apoptosis, inflammation, and oxidative stress in normotensive rats with intracerebral hemorrhage. J Pharmacol Exp Ther 2007; 322: 1051–1058.
Anderson S . Role of local and systemic angiotensin in diabetic renal disease. Kidney Int Suppl 1997; 63: S107–S110.
Cooper ME . The role of the renin-angiotensin-aldosterone system in diabetes and its vascular complications. Am J Hypertens 2004; 17: 16S–20S.
Lewis EJ, Hunsicker LG, Bain RP, Rohde RD . The effect of angiotensin converting-enzyme inhibition on diabetic nephropathy. The Collaborative Study Group. N Engl J Med 1993; 329: 1456–1462.
Mauer M, Zinman B, Gardiner R, Suissa S, Sinaiko A, Strand T, Drummond K, Donnelly S, Goodyer P, Gubler MC, Klein R . Renal and retinal effects of enalapril and losartan in type 1 diabetes. N Engl J Med 2009; 361: 40–51.
Chaturvedi N, Porta M, Klein R, Orchard T, Fuller J, Parving HH, Bilous R, Sjølie AK, DIRECT Programme Study Group. Effect of candesartan on prevention (DIRECT-Prevent 1) and progression (DIRECT-Protect 1) of retinopathy in type 1 diabetes: randomised, placebo-controlled trials. Lancet 2008; 372: 1394–1402.
Anonymous . Effects of ramipril on cardiovascular and microvascular outcomes in people with diabetes mellitus: results of the HOPE study and MICRO-HOPE substudy. Heart Outcomes Prevention Evaluation Study Investigators. Lancet 2000; 355: 253–259.
Ruggenenti P, Perna A, Ganeva M, Ene-Iordache B, Remuzzi G, BENEDICT Study Group. Impact of blood pressure control and angiotensin-converting enzyme inhibitor therapy on new-onset microalbuminuria in type 2 diabetes: a post hoc analysis of the BENEDICT trial. J Am Soc Nephrol 2006; 17: 3472–3481.
Parving HH, Lehnert H, Bröchner-Mortensen J, Gomis R, Andersen S, Arner P . Irbesartan in patients with type 2 diabetes and Microalbuminuria Study Group. The effect of irbesartan on the development of diabetic nephropathy in patients with type 2 diabetes. N Engl J Med 2001; 345: 870–878.
Brenner BM, Cooper ME, de Zeeuw D, Keane WF, Mitch WE, Parving HH, Remuzzi G, Snapinn SM, Shahinfar S, RENAAL Study Investigators. Effects of losartan on renal and cardiovascular outcomes in patients with type 2 diabetes and nephropathy. N Engl J Med 2001; 345: 861–869.
Lewis EJ, Hunsicker LG, Clarke WR, Berl T, Pohl MA, Lewis JB, Ritz E, Atkins RC, Rohde R, Raz I, Collaborative Study Group. Renoprotective effect of the angiotensin-receptor antagonist irbesartan in patients with nephropathy due to type 2 diabetes. N Engl J Med 2001; 345: 851–860.
Sjølie AK, Klein R, Porta M, Orchard T, Fuller J, Parving HH, Bilous R, Chaturvedi N . DIRECT programme study group: effect of candesartan on progression and regression of retinopathy in type 2 diabetes (DIRECT-Protect 2): a randomized placebo-controlled trial. Lancet 2008; 372: 1385–1393.
Mehdi UF, Adams-Huet B, Raskin P, Vega GL, Toto RD . Addition of angiotensin receptor blockade or mineralocorticoid antagonism to maximal angiotensin-converting enzyme inhibition in diabetic nephropathy. J Am Soc Nephrol 2009; 20: 2641–2650.
Kang YS, Ko GJ, Lee MH, Song HK, Han SY, Han KH, Kim HK, Han JY, Cha DR . Effect of eplerenone, enalapril and their combination treatment on diabetic nephropathy in type II diabetic rats. Nephrol Dial Transplant 2009; 24: 73–84.
Fujisawa G, Okada K, Muto S, Fujita N, Itabashi N, Kusano E, Ishibashi S . Spironolactone prevents early renal injury in streptozotocin-induced diabetic rats. Kidney Int 2004; 66: 1493–1502.
Han SY, Kim CH, Kim HS, Jee YH, Song HK, Lee MH, Han KH, Kim HK, Kang YS, Han JY, Kim YS, Cha DR . Spironolactone prevents diabetic nephropathy through an anti-inflammatory mechanism in type 2 diabetics rats. J Am Soc Nephrol 2006; 17: 1362–1372.
Takebayashi K, Matsumoto S, Aso Y, Inukai T . Aldosterone blockade attenuates urinary monocyte chemoattractant protein-1 and oxidative stress in patients with type 2 diabetes complicated by diabetic nephropathy. J Clin Endocrinol Metab 2006; 91: 2214–2217.
Xu Y, Osborne BW, Stanton RC . Diabetes causes inhibition of glucose-6-phosphate dehydrogenase via activation of PKA, which contributes to oxidative stress in rat kidney cortex. Am J Physiol Renal Physiol 2005; 289: 1040–1047.
Leopold JA, Cap A, Scribner AW, Stanton RC, Loscalzo J . Glucose-6-phosphate dehydrogenase deficiency promotes endothelial oxidant stress and decreases endothelial nitric oxide bioavailability. FASEB J 2001; 15: 1771–1773.
Leopold JA, Zhang YY, Scribner AW, Stanton RC, Loscalzo J . Glucose-6-phosphate dehydrogenase overexpression decreases endothelial cell oxidant stress and increases bioavailable nitric oxide. Arterioscler Thromb Vasc Biol 2003; 23: 411–417.
Xu Y, Zhang Z, Hu J, Stillman IE, Leopold JA, Handy DE, Loscalzo J, Stanton RC . Glucose-6-phosphate dehydrogenase-deficient mice have increased renal oxidative stress and increased albuminuria. FASEB J 2010; 24: 609–616.
Leopold JA, Dam A, Maron BA, Scribner AW, Liao R, Handy DE, Stanton RC, Pitt B, Loscalzo J . Aldosterone impairs vascular reactivity by decreasing glucose-6-phosphate dehydrogenase activity. Nat Med 2007; 13: 189–197.
Viengchareun S, Le Menuet D, Martinerie L, Munier M, Pascual-Le Tallec L, Lombès M . The mineralocorticoid receptor: insights into its molecular and (patho)physiological biology. Nucl Recept Signal 2007; 5: e012.
Wilkinson-Berka JL, Tan G, Jaworski K, Miller AG . Identification of a retinal aldosterone system and the protective effects of mineralocorticoid receptor antagonism on retinal vascular pathology. Circ Res 2009; 104: 124–133.
Castro-Chaves P, Cerqueira R, Pintalhao M, Leite-Moreira AF . New pathways of the renin-angiotensin system: the role of ACE2 in cardiovascular pathophysiology and therapy. Expert Opin Ther Targets 2010; 14: 485–496.
Oudit GY, Liu GC, Zhong J, Basu R, Chow FL, Zhou J, Loibner H, Janzek E, Schuster M, Penninger JM, Herzenberg AM, Kassiri Z, Scholey JW . Human recombinant ACE2 reduces the progression of diabetic nephropathy. Diabetes 2010; 59: 529–538.
Tikellis C, Johnston CI, Forbes JM, Burns WC, Thomas MC, Lew RA, Yarski M, Smith AI, Cooper ME . Identification of angiotensin converting enzyme 2 in the rodent retina. Curr Eye Res 2004; 29: 419–427.
Luhtala S, Vaajanen A, Oksala O, Valjakka J, Vapaatalo H . Activities of angiotensin-converting enzymes ACE1 and ACE2 and inhibition by bioactive peptides in porcine ocular tissues. J Ocul Pharmacol Ther 2009; 25: 23–28.
Burrell LM, Johnston CI, Tikellis C, Cooper ME . ACE2, a new regulator of the renin-angiotensin system. Trends Endocrinol Metab 2004; 15: 166–169.
Tikellis C, Johnston CI, Forbes JM, Burns WC, Burrell LM, Risvanis J, Cooper ME . Characterization of renal angiotensin converting enzyme 2 in diabetic nephropathy. Hypertension 2003; 41: 392–397.
Donoghue M, Hsieh F, Baronas E, Godbout K, Gosselin M, Stagliano N, Donovan M, Woolf B, Robison K, Jeyaseelan R, Breitbart RE, Acton S . A novel angiotensin-converting enzymerelated carboxypeptidase (ACE2) converts angiotensin I to angiotensin 1–9. Circ Res 2000; 87: E1–E9.
Reich HN, Oudit GY, Penninger JM, Scholey JW, Herzenberg AM . Decreased glomerular and tubular expression of ACE2 in patients with type 2 diabetes and kidney disease. Kidney Int 2008; 74: 1610–1616.
Wang G, Lai FM, Lai KB, Chow KM, Kwan CH, Li KT, Szeto CC . Discrepancy between intrarenal messenger RNA and protein expression of ACE and ACE2 in human diabetic nephropathy. Am J Nephrol 2009; 29: 524–531.
Wysocki J, Ye M, Soler MJ, Gurley SB, Xiao HD, Bernstein KE, Coffman TM, Chen S, Batlle D . ACE and ACE2 activity in diabetic mice. Diabetes 2006; 55: 2132–2139.
Shiota A, Yamamoto K, Ohishi M, Tatara Y, Ohnishi M, Maekawa Y, Iwamoto Y, Takeda M, Rakugi H . Loss of ACE2 accelerates time-dependent glomerular and tubulointerstitial damage in streptozotocin-induced diabetic mice. Hypertens Res 2010; 33: 298–307.
Nguyen G, Delarue F, Burckle C, Bouzhir L, Giller T, Sraer JD . Pivotal role of the renin/prorenin receptor in angiotensin II production and cellular responses to renin. J Clin Invest 2002; 109: 1417–1427.
Wilkinson-Berka JL, Heine R, Tan G, Cooper ME, Hatzopoulos KM, Fletcher EL, Binger KJ, Campbell DJ, Miller AG . RILLKKMPSV influences the vasculature, neurons and glia, and (Pro)renin receptor expression in the retina. Hypertension 2010; 55: 1454–1460.
Wilkinson-Berka JL, Campbell DJ . (Pro)renin receptor: a treatment target for diabetic retinopathy? Diabetes 2009; 58: 1485–1487.
Wilkinson-Berka JL . Prorenin and the (pro)renin receptor in ocular pathology. Am J Pathol 2008; 173: 1591–1594.
Feldman DL, Jin L, Xuan H, Contrepas A, Zhou Y, Webb RL, Mueller DN, Feldt S, Cumin F, Maniara W, Persohn E, Schuetz H, Jan Danser AH, Nguyen G . Effects of aliskiren on blood pressure, albuminuria, and (pro)renin receptor expression in diabetic TG(mRen-2)27 rats. Hypertension 2008; 52: 130–136.
Hsueh WA, Baxter JD . Human prorenin. Hypertension 1991; 17: 469–477.
Danser AH, van den Dorpel MA, Deinum J, Derkx FH, Franken AA, Peperkamp E, de Jong PT, Schalekamp MA . Renin, prorenin, and immunoreactive renin in vitreous fluid from eyes with and without diabetic retinopathy. J Clin Endocrinol Metab 1989; 68: 160–167.
Berka JL, Stubbs AJ, Wang DZ, DiNicolantonio R, Alcorn D, Campbell DJ, Skinner SL . Renin-containing Müller cells of the retina display endocrine features. Invest Ophthalmol Vis Sci 1995; 36: 1450–1458.
Campbell DJ . Critical review of prorenin and (pro)renin receptor research. Hypertension 2008; 51: 1259–1264.
Ichihara A, Hayashi M, Kaneshiro Y, Suzuki F, Nakagawa T, Tada Y, Koura Y, Nishiyama A, Okada H, Uddin MN, Nabi AH, Ishida Y, Inagami T, Saruta T . Inhibition of diabetic nephropathy by a decoy peptide corresponding to the ‘handle’ region for nonproteolytic activation of prorenin. J Clin Invest 2004; 114: 1128–1135.
Takahashi H, Ichihara A, Kaneshiro Y, Inomata K, Sakoda M, Takemitsu T, Nishiyama A, Itoh H . Regression of nephropathy developed in diabetes by (Pro)renin receptor blockade. J Am Soc Nephrol 2007; 18: 2054–2061.
Huang J, Siragy HM . Glucose promotes the production of interleukine-1beta and cyclooxygenase-2 in mesangial cells via enhanced (Pro)renin receptor expression. Endocrinology 2009; 150: 5557–5565.
Satofuka S, Ichihara A, Nagai N, Noda K, Ozawa Y, Fukamizu A, Tsubota K, Itoh H, Oike Y, Ishida S . (Pro)renin receptor-mediated signal transduction and tissue renin-angiotensin system contribute to diabetes-induced retinal inflammation. Diabetes 2009; 58: 1625–1633.
Tang Z, Shou I, Wang LN, Fukui M, Tomino Y . Effects of antihypertensive drugs or glycemic control on antioxidant enzyme activities in spontaneously hypertensive rats with diabetes. Nephron 1997; 76: 323–330.
Ogawa S, Kobori H, Ohashi N, Urushihara M, Nishiyama A, Mori T, Ishizuka T, Nako K, Ito S . Angiotensin II Type 1 receptor blockers reduce urinary angiotensinogen excretion and the levels of urinary markers of oxidative stress and inflammation in patients with Type 2 Diabetic nephropathy. Biomark Insights 2009; 4: 97–102.
United Kingdom Prospective Diabetes Study Group. Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). Lancet 1998; 352: 837–853.
Ruggenenti P, Cravedi P, Remuzzi G, Medscape. The RAAS in the pathogenesis and treatment of diabetic nephropathy. Nat Rev Nephrol 2010; 6: 319–330.
Wilkinson-Berka JL . Angiotensin and diabetic retinopathy. Int J Biochem Cell Biol 2006; 38: 752–765.
Silva PS, Cavallerano JD, Sun JK, Aiello LM, Aiello LP . Effect of systemic medications on onset and progression of diabetic retinopathy. Nat Rev Endocrinol 2010; 6: 494–508.
Gaede P, Lund-Andersen H, Parving HH, Pedersen O . Effect of a multifactorial intervention on mortality in type 2 diabetes. N Engl J Med 2008; 358: 580–591.
Nakamura T, Fujiwara N, Sugaya T, Ueda Y, Koide H . Effect of red wine on urinary protein, 8-hydroxydeoxyguanosine, and liver-type fatty acid-binding protein excretion in patients with diabetic nephropathy. Metabolism 2009; 58: 1185–1190.
Taubert D, Roesen R, Lehmann C, Jung N, Schömig E . Effects of low habitual cocoa intake on blood pressure and bioactive nitric oxide: a randomized controlled trial. JAMA 2007; 298: 49–60.
Mustata GT, Rosca M, Biemel KM, Reihl O, Smith MA, Viswanathan A, Strauch C, Du Y, Tang J, Kern TS, Lederer MO, Brownlee M, Weiss MF, Monnier VM . Paradoxical effects of green tea (Camellia sinensis) and antioxidant vitamins in diabetic rats: improved retinopathy and renal mitochondrial defects but deterioration of collagen matrix glycoxidation and cross-linking. Diabetes 2005; 54: 517–526.
Acknowledgements
This work was supported by the State of São Paulo Research Foundation (Fapesp) and National Council for Scientific and Technological Development (CNPq).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lopes de Faria, J., Silva, K. & Lopes de Faria, J. The contribution of hypertension to diabetic nephropathy and retinopathy: the role of inflammation and oxidative stress. Hypertens Res 34, 413–422 (2011). https://doi.org/10.1038/hr.2010.263
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/hr.2010.263
Keywords
This article is cited by
-
Retinal neurodegeneration in diabetic retinopathy with systemic hypertension
Acta Diabetologica (2024)
-
Burden and predictors of diabetic kidney disease in an adult Ugandan population with new-onset diabetes
BMC Research Notes (2023)
-
Diabetic nephropathy and hypertension in diabetes patients of sub-Saharan countries: a systematic review and meta-analysis
BMC Research Notes (2018)
-
Mitochondrial respiratory dysfunctions of blood mononuclear cells link with cardiac disturbance in patients with early-stage heart failure
Scientific Reports (2015)
-
Vitreous levels of oxidative stress biomarkers and the radical-scavenger α1-microglobulin/A1M in human rhegmatogenous retinal detachment
Graefe's Archive for Clinical and Experimental Ophthalmology (2013)