Abstract
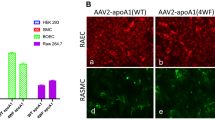
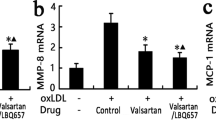
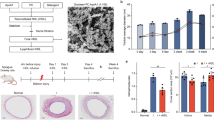
Paraoxonase-1 (PON-1) is a high-density lipoprotein (HDL)−associated enzyme that hydrolyzes oxidized phospholipids, thereby preventing the oxidative modification of low-density lipoproteins (LDL). A high-fat diet reduces PON-1 activity, enhancing LDL oxidation. Thus, PON-1 is a candidate for anti-atherogenic gene therapy. In the present study, we investigated the effect of local PON-1 overexpression on the development of atherosclerotic lesions using the Sendai virus–mediated transgenic technique. One-month-old rabbits (n=11) were fed a high-fat diet for 8 weeks and then subjected to balloon injury of the common iliac artery and simultaneous infection with a Sendai virus vector containing the PON-1 gene (n=7) or enhanced green fluorescence protein (EGFP) gene as a control (n=4). The arteries were examined 7–10 days after the operation. Local overexpression of PON-1 almost completely eliminated the immunohistochemical signals of the lectin-like oxidized LDL receptor-1 (LOX-1), thereby inhibiting macrophage accumulation, intimal thickening (by 63% compared with control), or atherosclerotic plaque formation in the vascular lumen (by 87.5%). Decreased levels of oxidative stress in the PON-1−treated arteries were confirmed by 4-hydroxy-2-nonenal (HNE) staining. Local overexpression of PON-1 in the arteries attenuated oxidative stress, thereby inhibiting the atherosclerotic process. Delivery of the PON-1 gene may be a possible therapeutic strategy for preventing atherosclerosis.
Similar content being viewed by others
Article PDF
References
James RW, Deakin SP : The importance of high density lipoproteins for paraoxonase 1 secretion, stability, and activity. Free Radic Biol Med 2004; 37: 1986–1994.
Durrington PN, Mackness B, Mackness MI : Paraoxonase and atherosclerosis. Arterioscler Thromb Vasc Biol 2001; 21: 473–480.
Clendenning JB, Humbert R, Green ED, Wood C, Traver D, Furlong CE : Structural organisation of the human PON1 gene. Genomics 1996; 35: 586–589.
Shih DM, Gu L, Xia YR, Navab M, Li WF, Hama S : Mice lacking serum paraoxonase are susceptible to organophosphate toxicity and atherosclerosis. Nature 1998; 394: 284–287.
Rozenberg O, Rosenblat M, Coleman R, Shih DM, Aviram M : Paraoxonase (PON1) deficiency is associated with increased macrophage oxidative stress: studies in PON1-knockout mice. Free Radic Biol Med 2003; 34: 774–784.
Shih DM, Xia YR, Wang XP, Miller E : Combined serum paraoxonase knockout/apolipoprotein E knockout mice exhibit increased lipoprotein oxidation and atherosclerosis. J Biol Chem 2000; 275: 17527–17535.
Rozenberga O, Shiha DM, Aviruma M : Paraoxonase 1 (PON1) attenuates macrophage oxidative status: studies PON1 transfected cells and in PON1 transgenic mice. Atherosclerosis 2005; 181: 9–18.
Shih DM, Gu L, Hama S, Xia YR, Navab M : Genetic-dietary regulation of serum paraoxonase expression and its role in atherogenesis in a mouse model. J Clin Invest 1996; 97: 1630–1639.
Ikeda Y, Suehiro T, Inoue M, Nakauchi Y, Morita T, Arii K : Serum paraoxonase activity and its relationship to diabetic complications in patients with non–insulin-dependent diabetes mellitus. Metabolism 1998; 47: 598–602.
Paragh G, Balla P, Katona E, Seres I, Egerhazi A, Degrell I : Serum paraoxonase activity changes in patients with Alzheimer's disease and vascular dementia. Eur Arch Psychiatry Clin Neurosci 2002; 252: 63–67.
Paragh G, Asztalos L, Seres I, et al: Serum paraoxonase activity changes in uremic and kidney-transplanted patients. Nephron 1999; 83: 126–131.
Mackness MI, Durrington PN, Mackness B : The role of paraoxonase 1 activity in cardiovascular disease: potential for therapeutic intervention. Am J Cardiovasc Drugs 2004; 4: 211–217.
Deakin S, Leviev I, Guernier S, James RW : Simvastatin modulates expression of the PON1 gene and increases serum paraoxonase: a role for sterol regulatory element–binding protein-2. Arterioscler Thromb Vasc Biol 2003; 23: 2083–2089.
Bradshaw G, Gutierrez A, Miyake JH : Facilitated replacement of Kupffer cells expressing a paraoxonase-1 transgene is essential for ameliorating atherosclerosis in mice. Proc Natl Acad Sci U S A 2005; 102: 11029–11034.
Sakaguchi T, Uchiyama T, Fujii Y, et al: Double membrane vesicles released from mammalian cells infected with Sendai virus expressing the matrix protein of vesicular stomatitis virus. Virology 1999; 263: 230–243.
Nga CJ, Diana B, Shiha M : The paraoxonase gene family and atherosclerosis. Free Radic Biol Med 2005; 38: 153–163.
Watson AD, Berliner JA, Hama SY, et al: Protective effect of high density lipoprotein associated paraoxonase: inhibition of the biological activity of minimally oxidized low density lipoprotein. J Clin Invest 1995; 96: 2882–2891.
Kita T, Kume N, Minami M, Hayashida K : Role of oxidized LDL in atherosclerosis. Ann NY Acad Sci 2001; 947: 199–205.
Kume N, Murase T, Masaki T, Kita T : Inducible expression of lectin-like oxidized low density lipoprotein receptor-1 in vascular endothelial cells. Circ Res 1998; 83: 322–327.
Li D, Mehta JL : Upregulation of endothelial receptor for oxidized LDL (LOX-1) by oxidized LDL and implications in apoptosis of human coronary artery endothelial cells: evidence from use of antisense LOX-1 mRNA and chemical inhibitors. Arterioscler Thromb Vasc Biol 2000; 20: 1116–11122.
Tanigawa H, Miura S, Matsuo Y, Fujino M : Dominant-negative lox-1 blocks homodimerization of wild-type lox-1−induced cell proliferation through extracellular signal regulated kinase 1/2 activation. Hypertension 2006; 48: 294–300.
Vohra RS, Murphy JE, Walker JH, Ponnambalam S, Homer-Vanniasinkam S : Atherosclerosis and the lectin-like oxidized low-density lipoprotein scavenger receptor. Trends Cardiovasc Med 2006; 16: 60–64.
Li D, Liu L, Chen H : LOX-1 mediates oxidized low-density lipoprotein–induced expression of matrix metalloproteinases in human coronary artery endothelial cells. Circulation 2003; 107: 612–617.
Negre-Salvayre A, Vieira O, Escargueil-Blanc I : Oxidized LDL and 4-hydroxynonenal modulate tyrosine kinase receptor activity. Mol Aspects Med 2003; 24: 251–261.
Zarkovic N : 4-Hydroxynonenal as a bioactive marker of pathophysiological processes. Mol Aspects Med 2003; 24: 281–291.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Miyoshi, M., Nakano, Y., Sakaguchi, T. et al. Gene Delivery of Paraoxonase-1 Inhibits Neointimal Hyperplasia after Arterial Balloon-Injury in Rabbits Fed a High-Fat Diet. Hypertens Res 30, 85–91 (2007). https://doi.org/10.1291/hypres.30.85
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1291/hypres.30.85