Abstract
We report a male patient with three copy-number variations (CNVs) and unique phenotype. He carried ~11.2 Mb terminal duplication on 4q, ~13.4 Mb terminal deletion on 7q and ~1.7 Mb interstitial duplication on Xp22.31, which were identified by array-based comparative genomic hybridization. He manifested mental retardation, mild brain anomalies and skeletal deformities ascribable to these CNVs, together with central precocious puberty and mild adrenocorticotropic hormone overproduction of unknown etiologies.
Similar content being viewed by others
Molecular cytogenetic technologies, including array-based comparative genomic hybridization, are useful to identify cryptic copy-number variations (CNVs) in individuals with an apparently normal karyotype.1–3 Recent cytogenetic studies have shown that cryptic CNVs account for a certain fraction of the etiologies of congenital disorders.1–3 Cryptic CNVs can underlie complex phenotypes that are not explainable by monogenic mutations, although they can also occur as benign polymorphisms.2,3
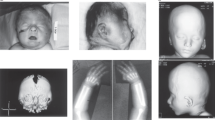
Here, we report a male patient with unique endocrine abnormalities and additional clinical features, in whom we identified three cryptic CNVs. This study was approved by the Institutional Review Board Committee at the National Center for Child Health and Development and performed after obtaining informed consent. The patient was born to non-consanguineous Japanese parents at 40 weeks of gestation. The patient’s parents were clinically normal, whereas two sisters and two of three maternal uncles of the patient had mild mental retardation. Endocrinological assessments were not performed for the family members. At birth, the patient showed mild genital skin pigmentation, mild joint contractures and muscle hypertonia. He had no episodes of hypoglycemia or seizure. His mental development was markedly delayed. He started walking on his own at 6 years of age. At 8 years of age, he was referred to our hospital for the evaluation of early sexual maturation. Physical assessment showed pubic hair of Tanner stage 2, penis of stage 5 and bilateral testes of 8 ml (+9.1 s.d.). He manifested skin pigmentation in the external genitalia, gingiva and fingers. In addition, he showed scoliosis and restricted joint extension of the limbs. He had no facial dysmorphism, except for strabismus, a broad nasal base and platycephaly. He spoke multiple words but no sentences, and was unable to run. His height and weight were 118.2 cm (−1.6 s.d.) and 20.8 kg (−1.1 s.d.), respectively. He had a significantly advanced bone age (14 years and 1 month); his predicted final height was around 130 cm (−7.0 s.d.). Skeletal radiography showed Madelung-like deformity of the forearm and scoliosis. Brain magnetic resonance imaging detected mild ventricular dilatation and mild atrophy of the anterior lobe. No abnormalities were observed in the hypothalamus or pituitary. Abdominal ultrasonography and computed tomography showed no abnormalities. Endocrine assessment revealed an elevated testosterone level and increased gonadotropin responses to gonadotropin-releasing hormone stimulation, which collectively indicated central precocious puberty (Table 1). He showed a mildly elevated level of insulin-like growth factor 1, which was consistent with the accelerated sexual maturation.4 Adrenocorticotropic hormone (ACTH) levels were elevated before and after corticotropin-releasing factor stimulation, while cortisol levels remained normal (Table 1). These data were consistent with a condition referred to as ‘overproduction of ACTH, not associated with Cushing disease’ (International Statistical Classification of Diseases and Related Health Problems 10th Revision). Blood levels of glucose and electrolytes were within the normal range. G-banding analysis showed a normal 46,XY karyotype. Array-based comparative genomic hybridization using a human catalog array (4×180 k format, Agilent Technologies, Santa Clara, CA, USA) delineated three heterozygous CNVs, i.e., ~11.2 Mb terminal duplication on 4q34.3-35.2, ~13.4 Mb terminal deletion on 7q35-36.3 and ~1.7 Mb interstitial duplication on Xp22.31 (Figure 1; Supplementary Table S1). These CNVs were not found in the database of Genomic Variants (http://dgv.tcag.ca/dgv/app/home?ref=GRCh37/hg19) or in the University of California Santa Cruz Genome Browser (https://genome.ucsc.edu/). The mother of the boy had the same duplication on Xp22.31, but no other CNV. DNA samples of other family members were not available for genetic testing.
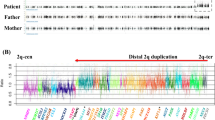
Cryptic copy-number variations (CNVs) in the patient. The black, red and green dots denote signals indicative of the normal, the increased (log ratio⩾+ 0.5) and the decreased (log ratio⩽−1.0) copy-numbers, respectively. Genomic positions correspond to the human genome reference assembly (UCSC Genome Browser, hg19, build 37). The red and green arrows indicate the duplicated and deleted regions, respectively. The names of the genes affected by the CNVs are shown in Supplementary Table S1.
These data suggest that the boy carried de novo or paternally inherited CNVs on 4q and 7q, and a maternally inherited duplication on Xp (Supplementary Table S1). CNVs on 4q and 7q appear to be pathogenic, because they affected more than 10 Mb regions and have been reported exclusively in individuals with developmental defects.5–14 Indeed, previous studies have shown that terminal duplications of 4q result in various clinical abnormalities, including mental retardation, short stature, microcephaly, facial dysmorphism and finger anomalies.5,6 Of these, mental retardation is likely to be associated with copy-number gain of GLRA3 or GPM6A,7,8 whereas craniofacial features and minor limb abnormalities can be ascribed to HAND2 duplication.9 Terminal deletions of 7q are known to cause haploinsufficiency of SHH that leads to holoprosencephaly, mental retardation, single incisor and limb anomalies.10 This 7q35-36 region also harbors several genes such as CNTNAP2, EZH2, KCNH2 and PRKAG2, whose haploinsufficiency has been associated with congenital malformations and/or neurological abnormalities.11–14 In contrast, the pathogenicity of the relatively small duplication on Xp22.3 remains unclear; Li et al.15 reported that CNVs at Xp22.3 are shared by 0.37% of individuals with neuropsychological disabilities and/or congenital anomalies and by 0.15% of healthy controls. Taken together, the mental retardation and mild brain anomalies, and probably skeletal deformities as well, of our patient are ascribable to one or more of these three CNVs. As molecular analysis was not performed for patient’s relatives except for the mother, it remains unknown whether these CNVs underlie mental retardation in his sisters and uncles. Furthermore, endocrinological evaluation was not performed for the patient’s mother with the same Xp22.3 duplication, and therefore the association between the CNV and hormonal abnormalities remained unknown. Although the boy lacked most of the characteristic features of 4q terminal duplications and 7q terminal deletions, this can be explained by a relatively incomplete penetrance or variable expressivity of these features.7,10 In particular, the lack of holoprosencephaly in this patient is consistent with the broad phenotypic spectrum of SHH haploinsufficiency.16 Alternatively, this patient may have somatic mosaicism.
The patient manifested additional clinical features that have not been reported in patients with 4q duplications, 7q deletions or Xp22.3 duplications. The most remarkable findings were central precocious puberty and mild ACTH overproduction. The origin of elevated ACTH levels in this case has yet to be studied. The hormone data are indicative of ACTH resistance; however, the patient showed no signs of glucocorticoid deficiency characteristic of ACTH resistance.17 Furthermore, it remains unclear whether precocious puberty and ACTH overproduction are independent events. Precocious puberty has not been described in patients with ACTH overproduction due to MC2R or MRAP mutations,17 although ACTH-dependent precocious pseudopuberty was observed in an infant with adrenal hypoplasia due to DAX1 mutations,18 and ACTH is known to stimulate testosterone production in the neonatal mouse testis.19 The CNVs in our patient included no known genes involved in the regulation of pituitary or adrenal hormones. However, since central precocious puberty and ACTH overproduction are genetically heterogeneous conditions,17,20 hitherto unidentified causative genes may reside within these CNVs. Further studies are necessary to clarify the etiology of the unique phenotype of the patient.
In summary, we identified a boy with three cryptic CNVs. The boy manifested mental retardation, mild brain anomalies and skeletal deformities that are ascribable to the CNVs on 4q, 7q and/or Xp, together with endocrine abnormalities of unknown genetic origin.
References
References
Di Gregorio E, Savin E, Biamino E, Belligni EF, Naretto VG, D’Alessandro G et al. Large cryptic genomic rearrangements with apparently normal karyotypes detected by array–CGH. Mol Cytogenet 2014; 7: 82.
Lee C, Iafrate AJ, Brothman AR . Copy number variations and clinical cytogenetic diagnosis of constitutional disorders. Nat Genet 2007; 39: S48–S54.
Miller DT, Adam MP, Aradhya S, Biesecker LG, Brothman AR, Carter NP et al. Consensus statement: chromosomal microarray is a first–tier clinical diagnostic test for individuals with developmental disabilities or congenital anomalies. Am J Hum Genet 2010; 86: 749–764.
Isojima T, Shimatsu A, Yokoya S, Chihara K, Tanaka T, Hizuka N et al. Standardized centile curves and reference intervals of serum insulin–like growth factor–I (IGF–I) levels in a normal Japanese population using the LMS method. Endocr J 2012; 59: 771–780.
Mikelsaar RV, Lurie IW, Ilus TE . "Pure" partial trisomy 4q25–qter owing to a de novo 4;22 translocation. J Med Genet 1996; 33: 344–345.
Egritas O, Cavdarli B, Dalgic B, Ergun MA, Percin F, Ziegler M et al. Duplication 4q associated with chronic cholestatic changes in liver biopsy. Eur J Med Genet 2010; 53: 411–414.
Thapa M, Asamoah A, Gowans GC, Platky KC, Barch MJ, Mouchrani P et al. Molecular characterization of distal 4q duplication in two patients using oligonucleotide array–based comparative genomic hybridization (oaCGH) analysis. Am J Med Genet A 2014; 164A: 1069–1074.
Gregor A, Kramer JM, van der Voet M, Schanze I, Uebe S, Donders R et al. Altered GPM6A/M6 dosage impairs cognition and causes phenotypes responsive to cholesterol in human and drosophila. Hum Mutat 2014; 35: 1495–1505.
Tamura M, Hosoya M, Fujita M, Iida T, Amano T, Maeno A et al. Overdosage of Hand2 causes limb and heart defects in the human chromosomal disorder partial trisomy distal 4q. Hum Mol Genet 2013; 22: 2471–2481.
Frints SG, Schoenmakers EF, Smeets E, Petit P, Fryns JP . De novo 7q36 deletion: breakpoint analysis and types of holoprosencephaly. Am J Med Genet 1998; 75: 153–158.
Mikhail FM, Lose EJ, Robin NH, Descartes MD, Rutledge KD, Rutledge SL et al. Clinically relevant single gene or intragenic deletions encompassing critical neurodevelopmental genes in patients with developmental delay, mental retardation, and/or autism spectrum disorders. Am J Med Genet A 2011; 155A: 2386–2396.
Tatton–Brown K, Rahman N . The NSD1 and EZH2 overgrowth genes, similarities and differences. Am J Med Genet C 2013; 163C: 86–91.
Zamorano–León JJ, Yañez R, Jaime G, Rodriguez–Sierra P, Calatrava–Ledrado L, Alvarez–Granada RR et al. KCNH2 gene mutation: a potential link between epilepsy and long QT–2 syndrome. J Neurogenet 2012; 26: 382–386.
Burwinkel B, Scott JW, Bührer C, van Landeghem FK, Cox GF, Wilson CJ et al. Fatal congenital heart glycogenosis caused by a recurrent activating R531Q mutation in the gamma 2–subunit of AMP–activated protein kinase (PRKAG2), not by phosphorylase kinase deficiency. Am J Hum Genet 2005; 76: 1034–1049.
Li F, Shen Y, Köhler U, Sharkey FH, Menon D, Coulleaux L et al. Interstitial microduplication of Xp22.31: causative of intellectual disability or benign copy number variant? Eur J Med Genet 2010; 53: 93–99.
Dubourg C, Lazaro L, Pasquier L, Bendavid C, Blayau M, Le Duff F et al. Molecular screening of SHH, ZIC2, SIX3, and TGIF genes in patients with features of holoprosencephaly spectrum: Mutation review and genotype–phenotype correlations. Hum Mutat 2004; 24: 43–51.
Metherell LA, Chan LF, Clark AJ . The genetics of ACTH resistance syndromes. Best Pract Res Clin Endocrinol Metab 2006; 20: 547–560.
Yeste D, González–Niño C, Pérez de Nanclares G, Pérez–Nanclares G, Audi L, Castaño L et al. ACTH–dependent precocious pseudopuberty in an infant with DAX1 gene mutation. Eur J Pediatr 2009; 168: 65–69.
O'Shaughnessy PJ, Fleming LM, Jackson G, Hochgeschwender U, Reed P, Baker PJ . Adrenocorticotropic hormone directly stimulates testosterone production by the fetal and neonatal mouse testis. Endocrinology 2003; 144: 3279–3284.
Silveira LF, Trarbach EB, Latronico AC . Genetics basis for GnRH–dependent pubertal disorders in humans. Mol Cell Endocrinol 2010; 324: 30–38.
Data Citations
Fukami, Maki HGV Database (2015) http://dx.doi.org/10.6084/m9.figshare.hgv.592
Fukami, Maki HGV Database (2015) http://dx.doi.org/10.6084/m9.figshare.hgv.594
Fukami, Maki HGV Database (2015) http://dx.doi.org/10.6084/m9.figshare.hgv.596
Acknowledgements
This work was supported by the Grants-in-Aid from the Ministry of Education, Culture, Sports, Science and Technology, from the Japan Society for the Promotion of Science and by the Grants from the Ministry of Health, Labor and Welfare, from National Center for Child Health and Development and from Takeda foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Supplementary information
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/4.0/
About this article
Cite this article
Okuno, M., Ogata, T., Nakabayashi, K. et al. Endocrinopathies in a boy with cryptic copy-number variations on 4q, 7q and Xp. Hum Genome Var 2, 15020 (2015). https://doi.org/10.1038/hgv.2015.20
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/hgv.2015.20